Updates on Psoriatic Arthritis at ACR 2018 Save
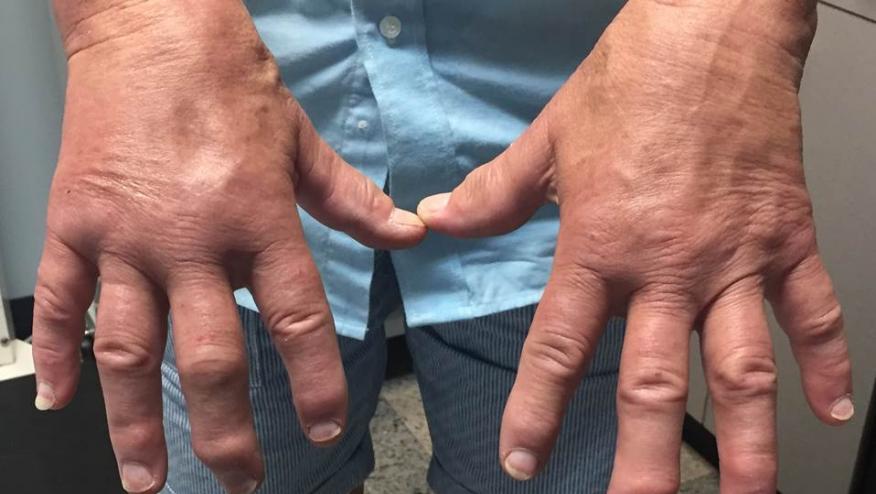
Memorable annual conferences are usually marked by a key focus on an emerging area of interest. I think 2018 was the year that belonged to psoriatic arthritis (although some may argue in favour of immune conditions induced by Checkpoint inhibitors). Many years ago the late Professor Verna Wright in Leeds (UK) was a lone voice for many years describing the clinical subtypes of psoriatic arthritis as something distinctive from rheumatoid arthritis. For a long time we thought that the treatments for PsA were just the same as RA since we were treating the same problem of synovitis.
One of the sessions this year was aptly entitled ‘RA and PsA – the great divorce’. The last few years have seen a paradigm change which was strongly reflected in many of the presentations this year. We saw evidence presented to support the hypothesis put forward by Professor Denis McGonagle (who also works in Leeds, but with Irish roots!) that the core pathology underpinning PsA is actually enthesitis. We heard about pathological and immunological studies that back this up, with a particular focus on IL-17 and the gut-enthesis axis.
So how Important are the Entheses?
Dr Lihi Eder (Abstract 1591) carried out a systematic ultrasound study of the entheses in 64 patients with PsA and found that sonographic enthesitis was noted in a similar number of patients to the clinical assessment of enthesitis (58 vs 52%) – and the presence of enthesitis was strongly linked to the presence of nail disease and disability (HAQ) score, less so to disease activity. Another interesting paper (Abstract 1614) screened for enthesitis using ultrasound in 180 patients with psoriasis but without arthritis. In this group, only 3% had tenderness over an entheseal site. They found a high Madrid enthesis index (MASEI) score in just under 10%. Perhaps not as common as I had thought it would be - not quite ready for a screening programme to help with early diagnosis in the skin clinic!
How many joints should we count? The GRAPPA group (international special interest group in PsA) has recommended the use of the SJC66/TJC68 count as the best performing measure (Abstract 1606). The GRAPPA group are also responsible for the CASPAR guidelines and treatment recommendations that are based on assessing each of the different aspects of the disease.
Apremilast
Abstract 1644 Dr Orbai Reported improvements in morning stiffness along with pain, physical function and response in a group of 219 PsA patients treated with Placebo or Apremilast 30mg bd for 16 weeks. As early as 2 weeks after starting treatment 43% of the active treatment group experienced a significant improvement in stiffness compared to 21% in the placebo group.
Filgotinib
Abstract 1821 Philip Mease presented the results of a Phase II study of Filgotinib, a selective JAK-1 inhibitor. This 16 week double blind, placebo controlled multicentre study involved 131 patients with active PsA. The endpoint of an ACR20 was achieved by 80% of those taking Filgotinib vs. 33% of those taking placebo. Minimal disease activity was achieved by 22% and 9% respectively. Disability (HAQ DI) scores decreased by an average of 0.57 compared to 0.28 in the control group. A PASI75 was achieved by 45 vs 15%. There were no new safety signals (cholesterol increased by 0.45 mmol/l)s. This drug looks like it will be another treatment option for PsA patients in the future, although it is not clear what it can offer that is better than existing drugs.
Abstract 1018 Tofacitinib has recently been approved by the FDA for the treatment of PsA. Dr Marzaioli (Dublin) presented an interesting study suggesting that the mode of action in these patients may be by inhibiting the differentiation of monocytes into dendritic cells, an important step in innate immunity.
Abstract 2565 Dr Nash reviewed the results of OPAL Balance, a 36 month extension study involving 468 patients. Notably, 13% of patients had serious adverse events including four patients who developed tuberculosis in spite of having negative QuantiFERON tests before the study.
Ixekizumab (IL-17A inhibitor)
Abstract 662 Desiree van der Heijde presented results from the SPIRIT-1 study – a phase I clinical trial investigating IXE in biologic naïve pts with active PsA. Over a 3 year period there were minimal changes in radiological scores in the majority of patients.
Abstract 1866 Dr Genovese presented data on the incidence of IBD in patients treated with Ixekizumab. Out of around 6000 patients who have received this drug in Psoriasis trials, 26 patients developed confirmed IBD (19 UC, 7 CD) i.e. 1.6 per 1000 patient years. The risk of developing IBD has been know for some time, but it is good to know that this is not a common event.
Abstract 2555 Dr Sunkureddi presented evidence that disease activity (as measured with DAPSA) improved rapidly and significantly in a group of PsA patients treated with Ixekizumab for 24 weeks, and that improvements were sustained through 52 weeks. The improvements were similar whether or not the patients were anti-TNF naive.
Abstract 2559 Dr Orbai presented the 2 year data from the SPIRIT-P2 trial in PsA.Overall 54% of patients completed 104 weeks of treatment. The ACR20 response rates were 60 and 48% for the monthly/fortnightly dosing regimens and responses were sustained over the 104 weeks. No significant new safety issues were identified.
Secukinumab (IL-17A inhibitor)
Abstract 2553. Dr Vibeke Strand presented results from the FUTURE trials in PsA patients. In over 2000 patients studied, the 300mg and 150mg doses resulted in significant and sustained improvements in quality of life, regardless of whether or not they had previously been treated with anti-TNF.
Guselkumab
Plenary Abstract 633 Dafna Gladman presented the results of a phase II study of 149 patients with PsA; 81 of whom had dactylitis. This study demonstrated a significant improvement in dactylitis which correlated with improved physical function. Three quarters of the group treated with GUS from the outset achieved remission of dactylitis. Dactylitis can be very difficult to treat using existing drugs, and this study suggests that it is very effective for this manifestation.
Abstract 2560 Dr Orbai presented results from the VOYAGE trial of GUS vs Adalimumab in patients with psoriasis (18% of whom self reported arthritis symptoms). Both drugs resulted in improvement but Guselkumab was significantly more successful in clearing scalp psoriasis.
Abstract 2566 Dr Puig presented results from ‘VOYAGE 2’, a phase III double blind study comparing Guselkumab with adalimumab and placebo in patients with moderate to severe psoriasis. Changes in patient reported outcomes were significantly better for Guselkumab than placebo in the PsA subgroup as well as the whole group. In the second half of the study, some of the PROs improved significantly better than those given Adalimumab although the design of the trial makes this difficult to assess (the GUS group had already been on the drug for 16 weeks whereas the ADA group had been on the drug for a shorter time).
Abatacept
Abstract 2554. Vibeke Strand presented a subgroup analysis from the Phase III ASTRAEA double blind placebo controlled study involving 424 patients with PsA: 168 were on monotherapy (no MTX). At week 24, the ACR20 response rates were 32% vs 12% in the placebo group. The mean changes in DAS28 CRP were -1.5 vs -0.68. These results do confirm that this drug could be an option in PsA, although to me those response rates are disappointing.
Other Topics
Abstract 1390 Dr Laura Coates presented an online global survey using the PsAID – looking at the impact of psoriatic arthritis from the patients perspective. This work (and others presented at the meeting) highlighted that the life impact of this condition is significant, and often reflects other issues that doctors might not focus on such as the extent or visibility of the skin disease or fatigue. Fatigue was the focus of Abstract 1393 by Dr Orbai (John Hopkins). Another abstract (1660) focused on Depression, which is commoner in PsA than RA. This study confirmed that depression was over 50% more likely in PsA than RA, and also found that this was independent of their joint score.
These brief reviews reflect my own take on some of the great work presented at this meeting. Of course, you should also read the abstracts online at the ACR website and check out some of the other reviews presented here on RheumNow.





If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.