All News
EULAR 2022 – Day 2 Report
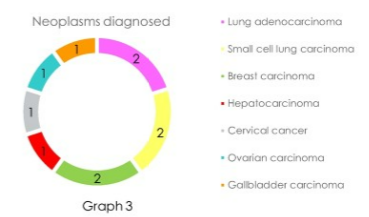
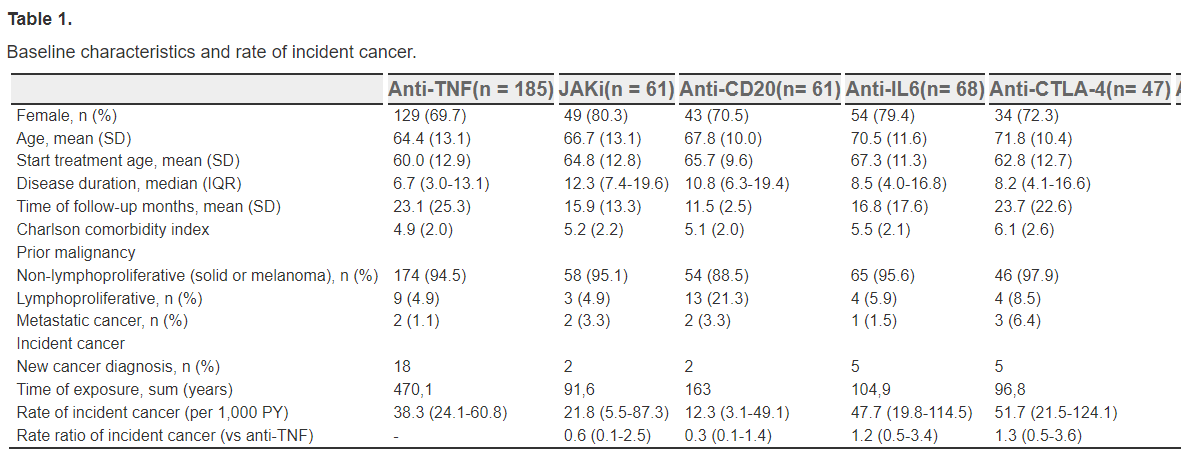
A full day at EULAR with oral and poster presentations of abstracts in the morning and a plethora of scientific (review) sessions in the afternoon. The latter covering topics like sarcoidosis, Still’s disease, fibromyalgia, back pain, sarcopenia, APS, IgG4 and imaging in vasculitis.
Read Article
Links:

Immune Checkpoint Inhibitor Associated Arthritis
Immune checkpoint inhibitors (ICIs) have dramatically improved outcomes in numerous cancers but have been hampered by a growing spectrum of immune-related adverse events (irAEs). Rheumatic complaints may be less frequent than other irAEs associated with ICI in cancer patients.
Read ArticleThe Inflammation Reflex (5.20.2022)
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com. This week, neuroimmunology leading to therapy, the cold shoulder approach and Regulatory updates from the FDA.
Read Article
Links:
Increased Cancer Risk with Autoimmune Disorders
The incidence and mortality of cancer among autoimmune diseases (AID) was studied in a large Chinese cohort, demonstrating that while onset time and risks vary, there is an increased risk of malignancy in AID.
Read Article
Risk vs. Benefit Lessons from the ORAL Surveillance Study
In an editorial in the current edition of the NEJM, Dr. Jasvinder Singh comments on the Oral Surveillance trial (Ytterberg et al.), a 4-year randomized, open-label, noninferiority, postauthorization, safety end-point trial, in high risk active rheumatoid arthritis patients over age 50 years.
Read Article2021 Rheumatology Year in Review
Our top 10 list of advances, game-changers, worries and those better medical practices that evolved during 2021.
Read Article
Links:
Dr. John Cush RheumNow ( View Tweet)
Best of 2021: FDA Puts Boxed Warnings on JAK Inhibitors
Based on the safety review of tofacitinib in Pfizer's Oral Surveillance (1133) study, the FDA has added serious boxed warnings to all three marketed JAK inhibitors (for inflammatory diseases) and formalized the recommendation that patients should be started on a TNF inhibitor (TNFi) before trying a JAK inhibitor. These recommendations apply to tofacitinib, baricitinib and upadacitinib.
Read ArticleBest of 2021: Tofacitinib Safety Concerns
Dr. Jack Cush reviews and discusses the news and journal reports from the past week on RheumNow.com.
Read ArticleCertain Cancers Increased with Autoimmune Diseases
Rheumatologists are well aware that inflammation and immune dysregulation are prime contributors to cancer risk, but do oncologists carry the same impressions? A large study from JAMA Oncology shows that patients with immune-mediated diseases (IMD) have an overall increased risk of cancer, especially in organ-specific immune-mediated diseases.
Read ArticleFDA Puts Boxed Warnings on JAK Inhibitors
Based on the safety review of tofacitinib in Pfizer's Oral Surveillance (1133) study, the FDA has added serious boxed warnings to all three marketed JAK inhibitors (for inflammatory diseases) and formalized the recommendation that patients should be started on a TNF inhibitor (TNFi) before trying a JAK inhibitor. These recommendations apply to tofacitinib, baricitinib and upadacitinib.
Read Article
Links: