Is Anifrolumab Good Enough for Systemic Lupus Erythematosus? Save
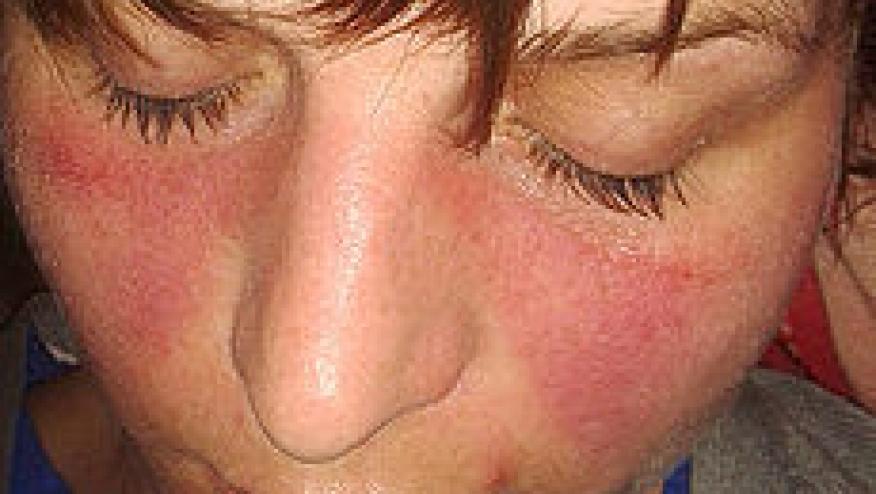
The NEJM published favorable results of a phase 3 trial of the alpha interferon inhibitor, anifrolumab, showing it to be superior to placebo in active systemic lupus erythematosus (SLE) patients studied for 48 weeks.
Anifrolumab, a human monoclonal antibody to type I interferon receptor subunit 1, has had split results in SLE studies previously. Furie et al reported significant benefits in a large phase 2 trial in active SLE (SRI-4: 34.3% vs 17.6%, p=-14). At the 2019 ACR Annual, meeting Furie et al presented the results of the TULIP 1 study; a large phase 3 trial of anifrolumab 300 mg vs. placebo in active SLE patients. Also using an SRI-4 primary outcome this study failed to meet its primary endpoint when compared to placebo (placebo 40% vs. anifrolumab 36%.
TULIP 2 is the name of the current phase 3 trial, similar in design to the previous trial but differed in using a different primary end point - the BICLA.
A total of 362 patients were randomized to receive intravenous anifrolumab (300 mg) or placebo every 4 weeks for 48 weeks. The primary end point of this trial was a response at week 52 defined with the use of the British Isles Lupus Assessment Group (BILAG)–based Composite Lupus Assessment (BICLA). A BICLA response requires reduction in any moderate-to-severe baseline disease activity and no worsening in any of nine organ systems in the BILAG index, no worsening on the Systemic Lupus Erythematosus Disease Activity Index, no increase of 0.3 points or more in the score on the Physician Global Assessment of disease activity (on a scale from 0 [no disease activity] to 3 [severe disease]), no discontinuation of the trial intervention, and no use of medications restricted by the protocol.
Whereas the TULIP 1 (similar numbers and inclusion criteria) failed to show a response, this TULIP 2 trial shows significantly better BICLA responses (48% vs. 31.5%; P=0.001). Interestingly the SRI-4 response was also higher in the anifrolumab treated patients.
A high interferon gene signature was seen in >80% of patients, but the response rates were similar for those with this signature (48% vs. 31%) or those without the aIFN signature (47% vs. 35%).
Primary safety concerns included herpes zoster and bronchitis in 7.2% and 12.2% of the anifrolumab patients, respectively.
While these results are encouraging, the positive TULIP 2 results are contrasted the by the negative TULIP 1 results. Moreover, the mechanistic plausibility (alpha interferon targeting) of anifrolumab is confused by the lack of augmented responses in those possessing high aIFN expression.








If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.