Cyclosporin and IVIG Effective in Kawasaki's Disease Save
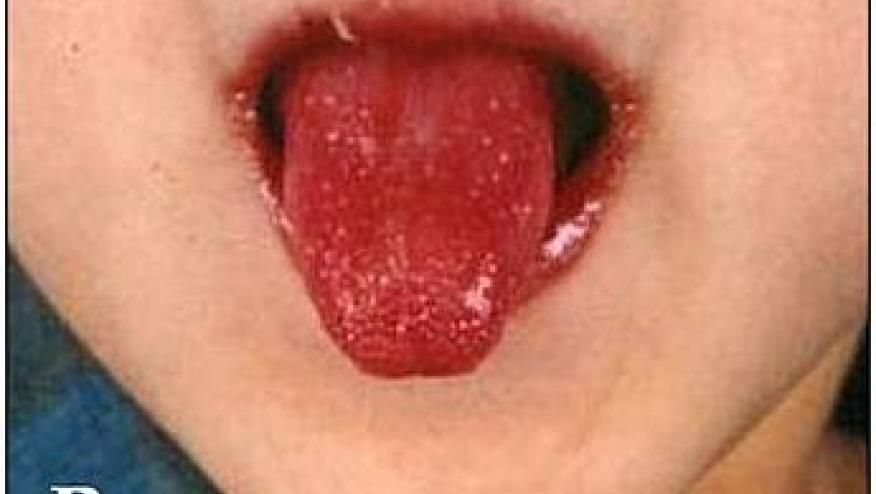
A Lancet study has shown that adding cyclosporin to intravenous immunoglobulin (IVIG) Kawasaki disease patients, who were predicted to be resistant to IVIG, was both safe and effective in averting severe coronary artery outcomes.
There are genetic studies suggesting upregulated calcium-nuclear factor of activated T cells pathway in the pathogenesis of Kawasaki disease. Moreover there are patients who are predicted to be resistent to IVIG therapy. Thus, this trial set out to assess if targeting these T cells with cyclosporin (CsA) would avert coronary artery abnormalities in Kawasaki patients.
This open-label, multicenter trial from Japan (2014 to 201) enrolled 175 Kawasaki patients predicted to be at higher risk for IVIG resistance and randomized them to receive either IVIG plus CsA (5 mg/kg/D for 5 days) or IVIG. The primary endpoint was incidence of coronary artery abnormalities at week 12.
Data pm 173 patients showed that coronary artery abnormalities:
- CsA + IVIG = 14% (Risk ratio 0·46; 95% CI 0·25–0·86; p=0·010)
- IVIG alone = 31%
Adverse events were not different between the groups (9% vs 7%).
The addition of CsA to IVIG yielded more favourable coronary artery outcomes in Kawasaki disease patients who were predicted to be unresponsive to IVIG.









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.