Drug-Induced Lupus is Rare with TNF inhibitor Therapy Save
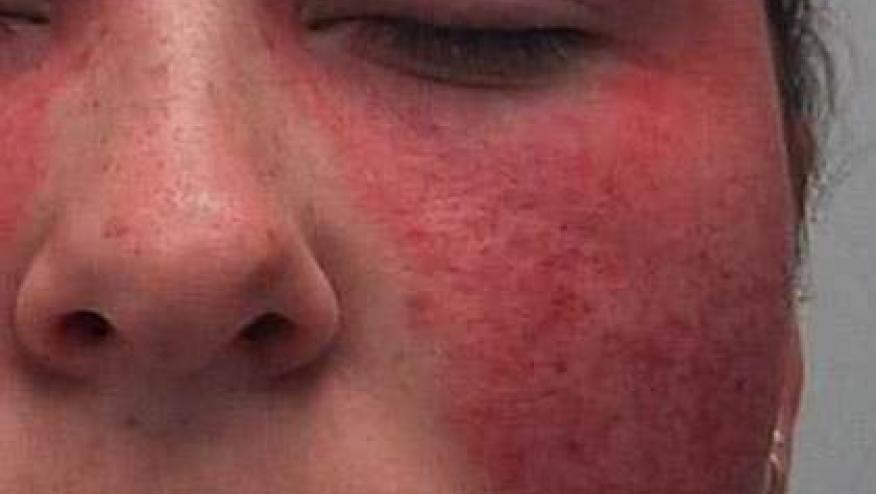
Drug-induced lupus (DIL) has been associated with all TNF inhibitors. Early rates suggested this may be between 0.04 - 0.06 events per 100 patient-years or roughtly 5 per 1000 patient-years. Jani and coworkers presented their analysis of the BSRBR biologics registry presented at the annual British Society of Rheumatology meeting. They found being female, non-white and having higher disease activity were predictive factors for DIL with TNFi. Cutaneous lupus manifestations were common. The found a 3-fold higher risk for patients on TNFi, with a crude event rate of 10 per 10,000 patient years (versus 3 per 10,000 PY on DMARDs alone).
Continue Reading
ADD THE FIRST COMMENT
Disclosures
The author has no conflicts of interest to disclose related to this subject








If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.