Ixekizumab COAST-V Trial Wins in Axial Spondyloarthritis Save
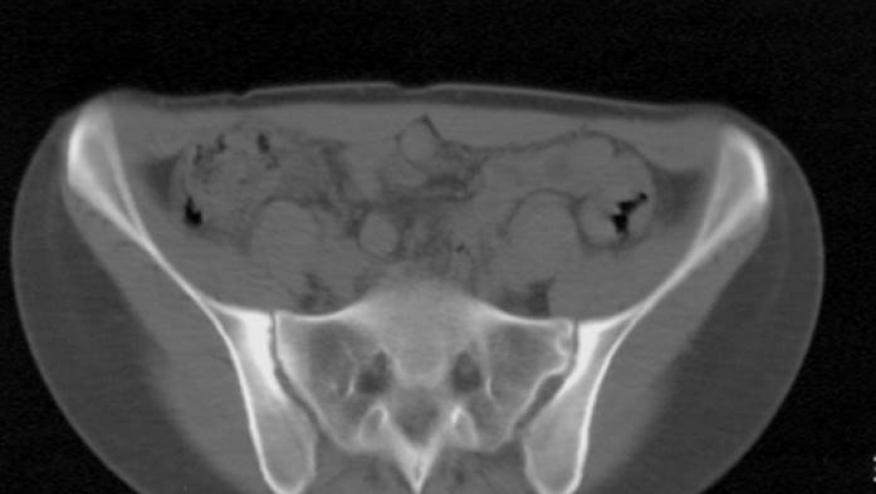
Lancet has published the results of a study showing that ixekizumab (an IL-17A inhibitor) yielded significant clinical benefit and radiographic protection when given to NSAID treated patients with radiographic axial spondyloarthritis (AxSpA).
The COAST-V trial was a phase 3, randomised, double-blind, placebo-controlled superiority study of ixekizumab, adult patients with inadequate response or intolerance to non-steroidal anti-inflammatory drugs.
Patients (n=341) were randomly assigned (1:1:1:1) to 80 mg subcutaneous ixekizumab every two (Q2W) or four (Q4W) weeks, 40 mg adalimumab Q2W (active reference group), or placebo (16%). The primary endpoint was the ASAS40 response at week 16.
ASAS40 responses were higher with ixekizumab Q2W (52%; p<0·0001), ixekizumab Q4W (48%; p<0·0001) than that seen with adalimumab (36%) or placebo. Adalimumab results were also significantly better than placebo responses.
MRI assessment of the spine and SI joints showed adalimumab and both doses of ixekizumab to be signficantly less SPARCC score changes from baseline assessments.
Serious infections were seen in 1% and there was one Candida infection in the adalimumab group.
IL-17A inhibition appears to be a safe and effective treatment option for patients with AxSpA and ankylosing spondylitis.





If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.