Minimal Disease Activity: The Goal in PsA Save
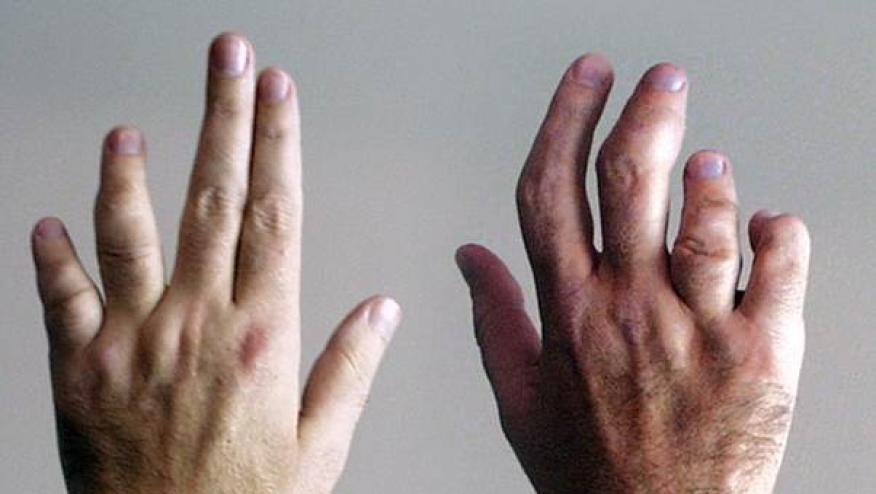
The majority of patients with psoriatic arthritis (PsA) who achieved a state of minimal disease activity (MDA) after 6 months of treatment with adalimumab (Humira) maintained that response through 24 months, a large observational study found.
At month 6, 35.5% of patients with longstanding, active PsA receiving adalimumab had reached MDA, and by month 24, 75% of those patients still had that response, according to Harald Burkhardt, MD, of Johann Wolfgang Goethe University in Frankfurt, Germany.
Minimal disease activity in PsA has been defined as swollen and tender joint counts of 1 or less, Psoriasis Area and Severity Index of 1 or less or body surface area of 3% or less, patient pain visual analog score of 15 or lower, patient global disease activity score of 20 or lower, Health Assessment Questionnaire Disability Index (HAQ-DI) no higher than 0.5, and tender entheseal points of 1 or less.
"Although MDA has been proposed as a means of guiding treatment decisions during daily clinical practice, the use of these criteria in a large nonselected multicenter population with longstanding disease has not been fully analyzed," Burkhardt and colleagues wrote in Rheumatology.
They therefore conducted a study from 2005 to 2013 of patients with PsA receiving routine medical care by approximately 300 clinicians in Germany. All patients had active disease, with a Disease Activity Score in 28 joints of 3.2 or higher, and completed at least 6 months of treatment.
A total of 1,684 patients were included. The sexes were equally represented, mean age was 50, and mean body mass index was 28.6.
The duration of psoriasis and arthritis symptoms was 18.1 and 9.5 years, respectively. Mean tender and swollen joint counts were 16.3 and 9.3, respectively, and pain scores averaged 6.3 on a scale of 10.
The mean time to reaching MDA among those who did so by month 6 was 4.4 months. Among patients who had not reached that goal by month 6 and remained on therapy, an additional 13.2% did so by month 9, as did another 26% by month 24.
Among the individual components of the MDA, the most often met were absence of enthesitis, achieved by 90% of patients; swollen joint counts of 1 or less, seen in 70% to 80%; and psoriasis body surface area of 3% or less, reached by 65% to 70%. The pain goal was the least often achieved, in only 21% to 25%.
For swollen and tender joints, 10% of patients who had not achieved the goal by 6 months did so by month 24, "suggesting that achievement of these outcomes might require a longer duration of therapy in some patients," the authors noted.
Responses on the criteria of the American College of Rheumatology (ACR20%, 50%, and 70% improvements) also were calculated. At 6 months, 59.9% had ACR20 responses, 37.3% had ACR50 responses, and 19% had ACR70 responses. At month 6, 51.5% of those with ACR20 responses also had reached MDA, as did 70.6% of those with ACR50 responses and 93.2% of those with ACR70 responses.
"Our findings indicate that MDA is a stable and valid assessment of disease activity during 'real-world' care; patients with PsA who achieved MDA at month 6 after initiation of adalimumab tended to maintain this status for at least 24 months," Burkhardt and colleagues observed.
Baseline characteristics that were predictive of achieving MDA were better function (assessed on the Funktionsfragebogen Hannover, similar to the HAQ-DI), with an odds ratio of 1.042 (95% CI 1.035 to 1.050, P<0.0001) and lower tender joint counts, with an odds ratio of 0.974 (95% CI 0.965 to 0.984, P<0.0001).
Other components of the MDA -- psoriasis body surface area, pain, swollen joint counts, patient global assessment, and enthesitis -- were not significant predictors. "The fact that only two of the seven MDA characteristics are significant predictors of MDA at month 6 suggests that baseline characteristics do not have an insurmountable influence on treatment outcome as assessed by MDA," the researchers noted.
They concluded that MDA can be a useful tool for guiding treatment decisions, but also that some patients appeared to need more than 6 months to achieve this goal.
"For patients who are tolerating therapy and showing signs of improvement but have not yet achieved MDA, it may be appropriate to consider continuing treatment for longer than 6 months before suggesting a change in therapy, particularly if pain and skin parameters have improved but joint counts have not yet reached MDA status," they wrote.
A limitation of the study, the researchers said, was the loss of some patients to follow-up during the 24 months of the study.
The study was funded by AbbVie, and several of the authors reported financial relationships with the company.







If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.