Primary Sjögren's Syndrome Guideline from the British Society for Rheumatology Save
The British Society of Rheumatology (BSR) has been accredited by NICE to develop a guidance document for the management of primary Sjögren's Syndrome intended for rheumatologists, general physicians, general practitioners, specialist nurses and other specialists (e.g. ophthalmologists, dental practitioners and ENT specialists),
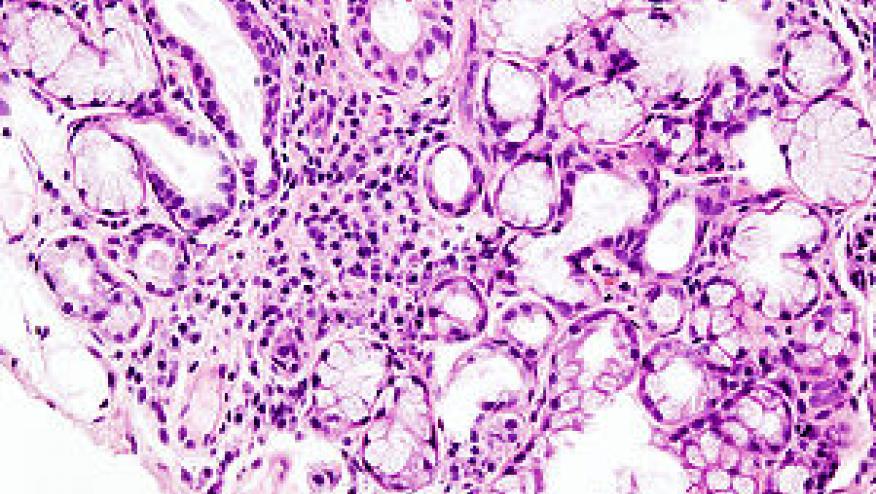
Primary SS (pSS) is a classic, immune-mediated, condition [1] typically presenting in women in their fifth or sixth decade, although up to 10% of cases occur in men. The prevalence in women in the UK is 0.1–0.4% [2]. Patients characteristically present with dryness of the eyes and mouth but systemic features are common and B-cell lymphoma affects 5–10% [3, 4].
Guidance was based on a literature reivew from 1990 to Jan 2016 and recommendations were only included where the mean strength of agreement (SOA) was ⩾7 and ⩾75% respondents scored ⩾7.
The guideline focus on several key areas. The following are key concepts taken from the guidance document.
Eyes
- Humidification and avoidance of systemic medication that exacerbates dryness. LOE Ib/A; SOA 8.9 (97%).
- Early referral to an ophthalmologist for review and consideration of insertion of punctual plugs or cauterization. LOE II/B; SOA 9.27 (100%).
- Start with simple lubricating drops using the most cost-effective options (see Table 1 for preparations). LOE IV/D; SOA 9.3 (93.3%).
- Avoid preservative-containing preparations. LOE I/A; SOA 9.21 (92.9%).
- Refer patients with severe dry eye, not responding to conventional treatment, to specialist commissioned centres for consideration of serum eye drops. LOE IIb/B; SOA 9.5 (100%).
- Low dose steroid-containing eye drops for short term use under ophthalmic supervision only. LOE Ib/A; SOA 9.64 (100%).
- Avoid long term use of topical steroids. LOE Ib/A; SOA 9.64 (100%).
- Ciclosporin eye drops or ointments under ophthalmic supervision for chronic inflammation. LOE Ib/A; SOA 9.38 (100%).
- Use topical NSAIDs with caution under ophthalmic supervision only. LOE IIb/B; SOA 9.86 (100%).
- A trial of pilocarpine 5 mg once daily increasing stepwise to 5 mg qds is recommended for patients with significant sicca symptoms. LOE IIb/B; SOA 9.08 (92.3%).
Mouth
- Advise excellent oral hygiene, limit sugar intake and avoid food and drinks other than plain water between meals and from 1 h before bedtime and through the night. LOE IV/D; SOA 9.8 (100%).
- Advise assessment by an oral medicine specialist and/or regular visits to a general dental practitioner. LOE IV/D; SOA 9.8 (100%).
- Brush teeth at least twice daily (but not immediately after eating) including before bed using a pea sized amount of high fluoride toothpaste and use fluoride-containing oral gel on teeth twice daily. LOE I/A; SOA 9.8 (100%).
- Chew xylitol-containing sugar-free gum. LOE IIb/B; SOA 9.67 (100%).
- A trial of pilocarpine 5 mg once daily increasing stepwise to 5 mg qds if significant sicca symptoms. LOE IIb/B; SOA 9.77 (100%).
Management of salivary gland enlargement
- Consider baseline US to assess for active inflammation, infection and stones. LOE IV/D; SOA 9.7 (100%).
- If acute inflammation, in the absence of infection and stones, consider short course of oral prednisolone or intra-muscular depomedrone. LOE IV/D; SOA 9.01 (100%).
Systemic dryness
- A trial of pilocarpine 5 mg once daily increasing stepwise to 5 mg qds if systemic dryness. LOE IV/D; SOA 9.08 (92.3%).
- A non-hormonal vaginal moisturizer ± topical oestrogen if vaginal dryness. LOE I/A; SOA 9.8 (100%).
Treatment of systemic disease
- HCQ (6 mg/kg) for those with skin, joint disease or fatigue. LOE IIa/B; SOA 9.64 (100%).
- Ciclosporin A may be helpful in patients with significant joint involvement. LOE III/C; SOA 8.58 (85.7%).
- AZA may be considered in patients with systemic complications. LOE III/C; SOA 9.09 (100%).
- MTX is useful for patients with an associated inflammatory arthritis. LOE IV/D SOA 9.54 (100%).
- Mycophenolate may be considered in patients with systemic complications. LOE III/C; SOA 9.1 (100%).
Corticosteroids
- Intermittent short courses of oral or intramuscular steroid for systemic flares and significant organ manifestations with or without additional immunosuppressive treatment. LOE III/C; SOA 9.2 (100%).
- Low dose oral prednisolone for persistent constitutional symptoms in patients with inadequate response to other immunosuppresants. LOE IIb/B; SOA 8.92 (100%).
- Rituximab for specialist use in patients with significant systemic manifestations refractory to other immunosuppresives and those with lymphoma, immune thrombocytopenia, vasculitic neuropathy or cryoglobulinaemia. LOE IIb/B; SOA 9.43 (100%).
Assessment and management of lymphoma
- Review high risk patients regularly and warn patients to report firm, painless glandular swelling that does not settle. LOA IV/D; SOA 9.86 (100%).
- Investigate suspicious lesions with local US, biopsy and CT chest, abdomen and pelvis for staging. LOA III/C; SOA9.77 (100%).









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.