Psoriasis Risk Increased with TNF Inhibitors in Juveniles Save
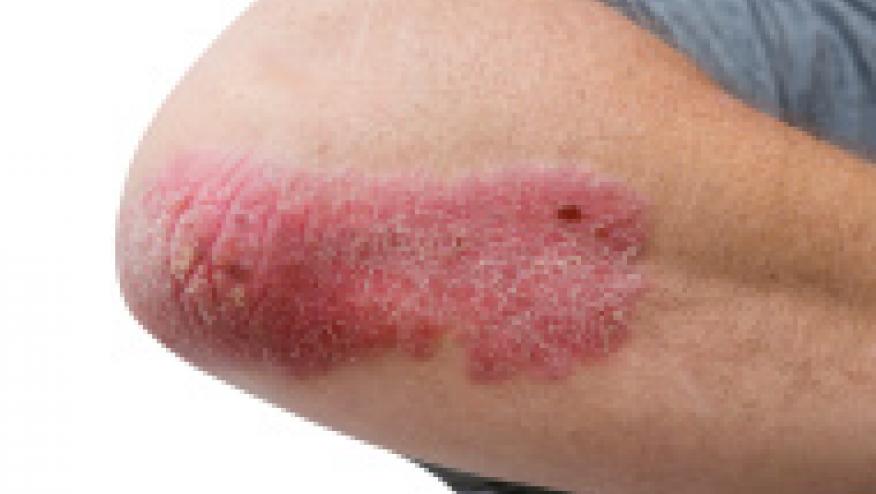
Children with inflammatory diseases who were treated with tumor necrosis factor (TNF) inhibitors had a higher rate of incident psoriasis than those not exposed to these biologics, a single-center retrospective study found.
In a cohort of more than 4,000 children with inflammatory disorders, the overall standardized incidence ratio for developing psoriasis was 18 (95% CI 15-22), but was 30 (95% CI 23-39) for those treated with TNF inhibitors compared with only 9.3 (95% CI 6.3-14) for those not using TNF inhibitors, according to Lisa H. Buckley, MD, of Children's Hospital of Philadelphia (CHOP), and colleagues.
In a multivariate analysis, after adjustment for factors such as sex, race, family history of psoriasis, obesity, underlying diagnosis, and methotrexate use, the risk for psoriasis among patients with TNF inhibitor exposure was increased almost fourfold (HR 3.84, 95% CI 2.28-6.47, P<0.001), the researchers reported online in Arthritis Care & Research.
The increasingly widespread use of TNF inhibitors for pediatric conditions such as inflammatory bowel disease (IBD) and juvenile idiopathic arthritis (JIA) has been accompanied by an increase in reports of cutaneous adverse events, including paradoxical incident psoriasis. But TNF inhibitors also are used in the treatment of psoriasis -- these agents "can both treat and trigger psoriasis," the authors explained.
Data thus far have primarily consisted of case reports and series. Therefore, to more fully explore this adverse event, Buckley and colleagues conducted a review of the CHOP electronic records, identifying 4,111 patients younger than age 19 with IBD, JIA, or chronic noninfectious osteomyelitis seen from 2008 to 2018. Those with a personal history of psoriasis were excluded.
Among the cohort, 1,614 had been treated with a TNF inhibitor -- for IBD in 74%, for JIA in 24%, and for chronic noninfectious osteomyelitis in 2%. Mean age was 12 years for those with TNF inhibitor exposure and 11 for the unexposed, and median follow-up was 2.47 years for the exposed and 1.94 years for the unexposed. More patients in the exposed group had been treated with methotrexate (37% vs 14%, P<0.001), but fewer of this group were obese (2% vs 4%, P=0.01).
The incidence rates for developing psoriasis were 7.3 per 1,000 person-years for the cohort overall, but 12.3 per 1,000 for those with TNF inhibitor exposure compared with 3.8 per 1,000 for those without exposure.
When the researchers broke the incidence rates down according to diagnosis, they found that for those with IBD, the incidence rates were 10.9 per 1,000 person-years among those with TNF inhibitor exposure and 2.6 per 1,000 for those without exposure. In the JIA group, the incidence rates were 14.7 per 1,000 among the exposed and 5.5 per 1,000 for the unexposed.
Standardized incidence ratios among those with IBD were 27 (95% CI 19-37) for those with TNF inhibitor exposure compared with 6.3 (95% CI 3.7-11) for those without exposure, and among those with JIA, the ratios were 36 (95% CI 23-57) and 13 (95% CI 6.7-27), respectively.
In the multivariate analysis, the only factor other than TNF inhibitor exposure in the overall cohort that was associated with an increased risk of incident psoriasis was a family history of psoriasis (HR 3.11, 95% CI 1.79-5.41, P<0.001). Risks were also increased with TNF inhibitor exposure according to diagnosis, with hazard ratios of 4.52 (95% CI 2.37-8.63, P<0.001) for those with IBD and 2.90 (95% CI 1.24-6.77, P=0.01) for those with JIA.
Limitations of the study included its single-center, retrospective design, the researchers said.
"There is a paucity of data regarding this adverse cutaneous event in children, and therefore this study contributes significantly to the current literature," Buckley and colleagues wrote. "Despite limitations, the results are highly significant and indicate the need for additional studies to include long-term data from large observational registries."
The authors reported no conflicts of interest.









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.