A Role for SGLT2 Inhibition in Gout? Save
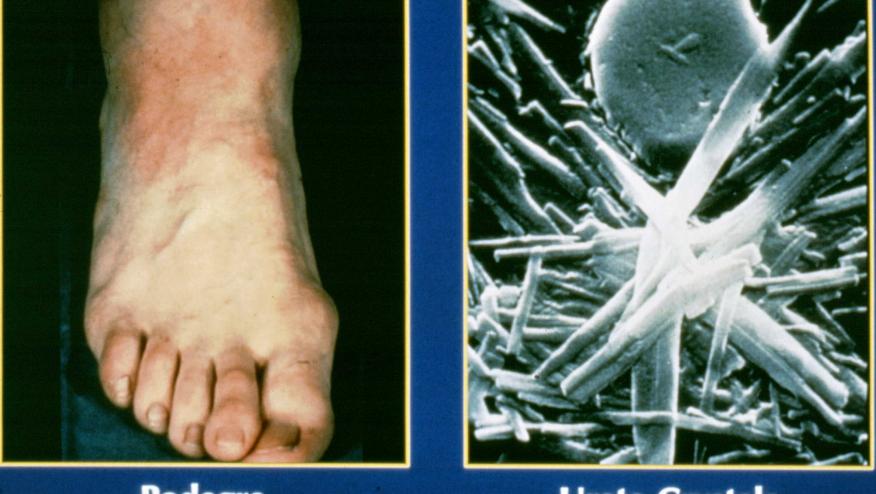
The antidiabetic medication canagliflozin (Invokana) lowered serum urate and reduced the risk of gout flare in a post-hoc analysis of data from two large clinical trials.
In the CANVAS clinical trial program, which was funded by Janssen Research & Development and included more than 10,000 patients, the percentage difference in serum urate reduction with canagliflozin versus placebo was 6.7% (95% CI -7.3 to -6.1), according to Bruce Neal, MBChB, PhD, of the University of Sydney in Australia, and colleagues.
In addition, the likelihood of a gout flare or the initiation of treatment for gout was halved in patients treated with canagliflozin, with a hazard ratio of 0.53 (95% CI 0.40 to 0.71, P<0.0001), the researchers reported online in The Lancet Rheumatology.
"This agent appears to show potential for being a dual-benefit gout therapy, lowering serum urate and providing potential flare prophylaxis and thereby potentially reducing the need for separate flare prophylaxis with agents that are often contraindicated or poorly tolerated in patients with gout," wrote Tuhina Neogi, MD, PhD, chief of rheumatology at Boston University School of Medicine, in an accompanying comment.
Canagliflozin is a sodium glucose co-transporter 2 (SGLT2) inhibitor that has varied effects including lowering blood glucose, weight, and blood pressure, as well as helping control albuminuria in individuals with type 2 diabetes. It also has been shown to decrease serum urate levels, as do other SGLT2 inhibitors.
However, whether this class of drugs or canagliflozin specifically can actually protect against gout has not been determined, so Neal and colleagues analyzed data from the Canagliflozin Cardiovascular Assessment Study program, which consisted of two randomized trials comparing the drug in daily doses of 100 mg or 300 mg with placebo in patients with type 2 diabetes and an increased risk of cardiovascular disease. The active treatment was given to 5,795 patients and placebo to 4,347, and mean follow-up was 3.6 years.
The majority of patients were white men, mean age was 63, and mean duration of diabetes was 13.5 years. At baseline, mean serum urate concentration was 349 μmol/L, and more than 40% had serum urate levels above 360 μmol/L, or 6 mg/dL. A history of clinical gout was present in 5% of patients.
At the first follow-up visit (week 6), serum urate levels had already fallen in the canagliflozin group and remained lower throughout the study, with mean concentrations -23.3 μmol/L (95% CI -25.4 to -21.3) lower than in the placebo group.
Reductions in serum urate were greater among the following groups:
- Individuals older than 65 (-7.4% vs -6.1%, P=0.019)
- Women (-9.1% vs -5.4%, P<0.0001)
- Patients who were obese (-7.3% vs -5.8%, P=0.017)
- Those whose duration of diabetes was shorter than 12 years (-7.7% vs -5.7%, P-0.0010)
- Those whose glycated hemoglobin was below 8% (-9.1% vs -4.9%, P<0.0001)
- Those whose estimated glomerular filtration rate was above 60 mL/min/1.73 m2 (-7.3 vs -4, P<0.0001)
The rate of gout flare or initiating gout treatment was 4.1 per 1,000 patient-years with canagliflozin versus 6.6 per 1,000 patient-years with placebo.
For gout flare specifically, the rates were 2.0 per 1,000 with canagliflozin versus 2.6 per 1,000 with placebo (HR 0.64, 95% CI 0.41 to 0.99, P=0.046), while for starting new gout treatment, the rates were 3.3 per 1,000 versus 5.4, respectively (HR 0.52, 95% CI 0.38 to 0.72, P<0.0001).
"These data suggest a possible protective effect for gout from an agent used for the management of diabetes and, if confirmed in dedicated prospective studies, would provide a rationale for the selective use of canagliflozin among people with type 2 diabetes who have gout or are at high risk for gout," Neal and colleagues wrote.
They also noted that the 6.7% mean lowering of serum urate was small, compared with reductions typically seen with allopurinol or febuxostat, which average 24% to 27%.
Nonetheless, the protective effect against gout was large, they observed, suggesting that the mechanisms involved may extend beyond urate lowering. This could involve the activation of the anti-inflammatory mediator AMPK in endothelial cells, which has been shown in vitro to result in the suppression of crystal-induced inflammation, Neogi explained.
"Thus, canagliflozin might have unique anti-inflammatory effects that are not seen class-wide, and might therefore have potential as another novel urate-lowering antiflare therapy," she wrote.
"Well conducted, appropriately powered phase III trials are warranted for careful benefit-risk evaluation and assessment of canagliflozin's potential role as a urate-lowering, antiflare therapy among the handful of therapies that are currently available for the management of gout," she concluded.
A limitation of the study, the researchers said, was its post-hoc design.
The study was funded by Janssen Research & Development.
The authors reported financial ties with Janssen and multiple other companies, including AbbVie, Astellas, AstraZeneca, Bayer, Baxter, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Gilead, GlaxoSmithKline, Merck, Novartis, Novo Nordisk, Pfizer, Sanofi, Servier, Mitsubishi Tanabe, Johnson & Johnson, and Regeneron.
Primary Source










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.