Shingles Vaccine Not Cost-Effective in Those Under 60 Save
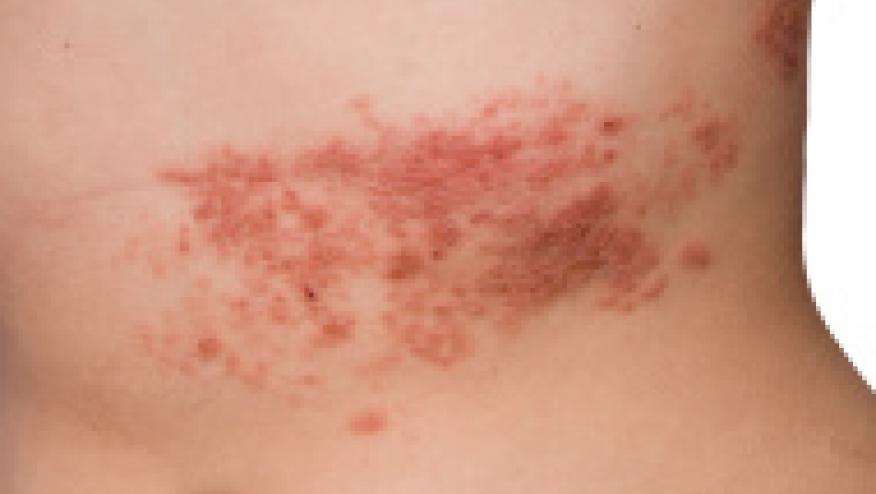
The herpes zoster (HZ- shingles) vaccine was FDA approved for use in individuals over age 50 years. However, the CDC Advisory Committee on Immunization Practices (ACIP) has recommended it only be used in those over age 60 years.
Annals of Internal Medicine reports an a cost-effectiveness analysis of of HZ vaccine versus no vaccination. For every 1000 persons receiving the vaccine at age 50 years, 25 HZ cases and 1 post-herpetic neuralgia case could be prevented. The incremental cost-effectiveness ratio (ICER) for HZ vaccine versus no vaccine was $323 456 per quality-adjusted life-year (QALY). Typically, preventive treatments are considered cost-effective when they cost at most $100,000 per QALY, that is, per year of healthy life gained.
The HZ vaccine costs over $200. To be cost effective in the 50-59 yr age group the cost would have to be less than $80. These findings support the ACIP recommendations to limit the vaccine to those over 60 yrs of age.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.