Upadacitinib Effective in Ankylosing Spondylitis Save
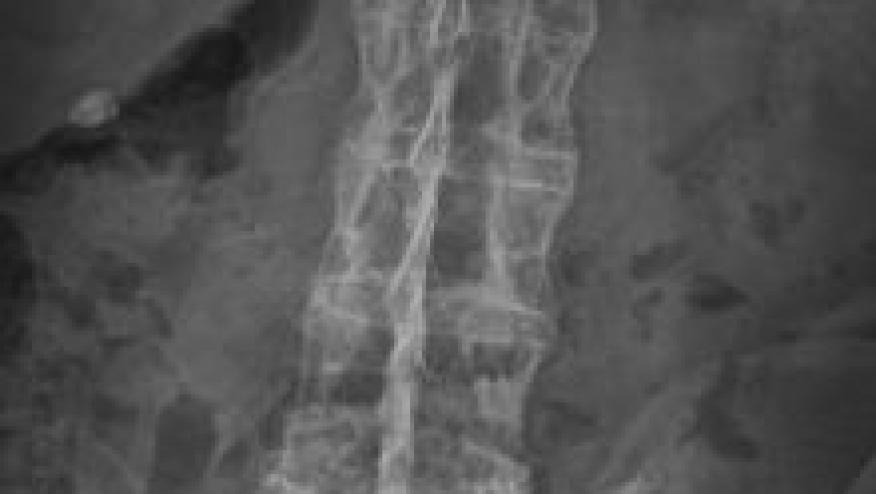
Lancet reports that the selective JAK1 inhibitor, upadacitinib, was effective when given to patients with active ankylosing spondylitis.
The SELECT-AXIS 1, enrolled adults with active ankylosing spondylitis patients who were biologics naive and were randomized to receive upadacitinib 15 mg once daily or placebo for the 14-weeks. The primary endpoint was the composite outcome measure of the Assessment of SpondyloArthritis international Society 40 (ASAS40) response at week 14.
A total of 187 patients were treated. UPAD had significantly more patients achieving an ASAS40 response at week 14 (52% vs 26%; p=0·0003).
Common adverse events with upadacitinib included increased creatine phosphokinase levels in 9% (2% with PBO). There were no serious infections, herpes zoster, malignancy, venous thromboembolic events, or deaths reported.
This is one of th first trials showing that JAK inhibition (with upadacitinib) is efficacious in active ankylosing spondylitis.






If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.