Drug Safety
The big news this week: the approval of deucravacitinib (a new class of drug?) for psoriasis; the 2022 ACR guidance on glucocorticoid-induced osteoporosis; a national poll of older adults over the age of 50 who claimed self-reported or doctor-diagnosed arthritis; and much more. Let's review these and other news, journal reports and announcements from this past week.

Dr. John Cush RheumNow
3 years 8 months ago
Open label, noninferiority study TNFi withdrawal in 64 PsA & 58 axSpA pts. 2/3 tapered by dz activity T2T strategy (N=81) & 1/3 continued TNFi. At 12 mos. LDA seen in 69% w/ Tapering (~avg 53% dose) vs 73% no-tapering group (91% dose) https://t.co/K6WFaesyRt https://t.co/zqo6nVGtqW

A systematic review suggests that low-dose methotrexate (MTX) use is associated with an increased melanoma risk, but the absolute risk increase could be considered negligible.
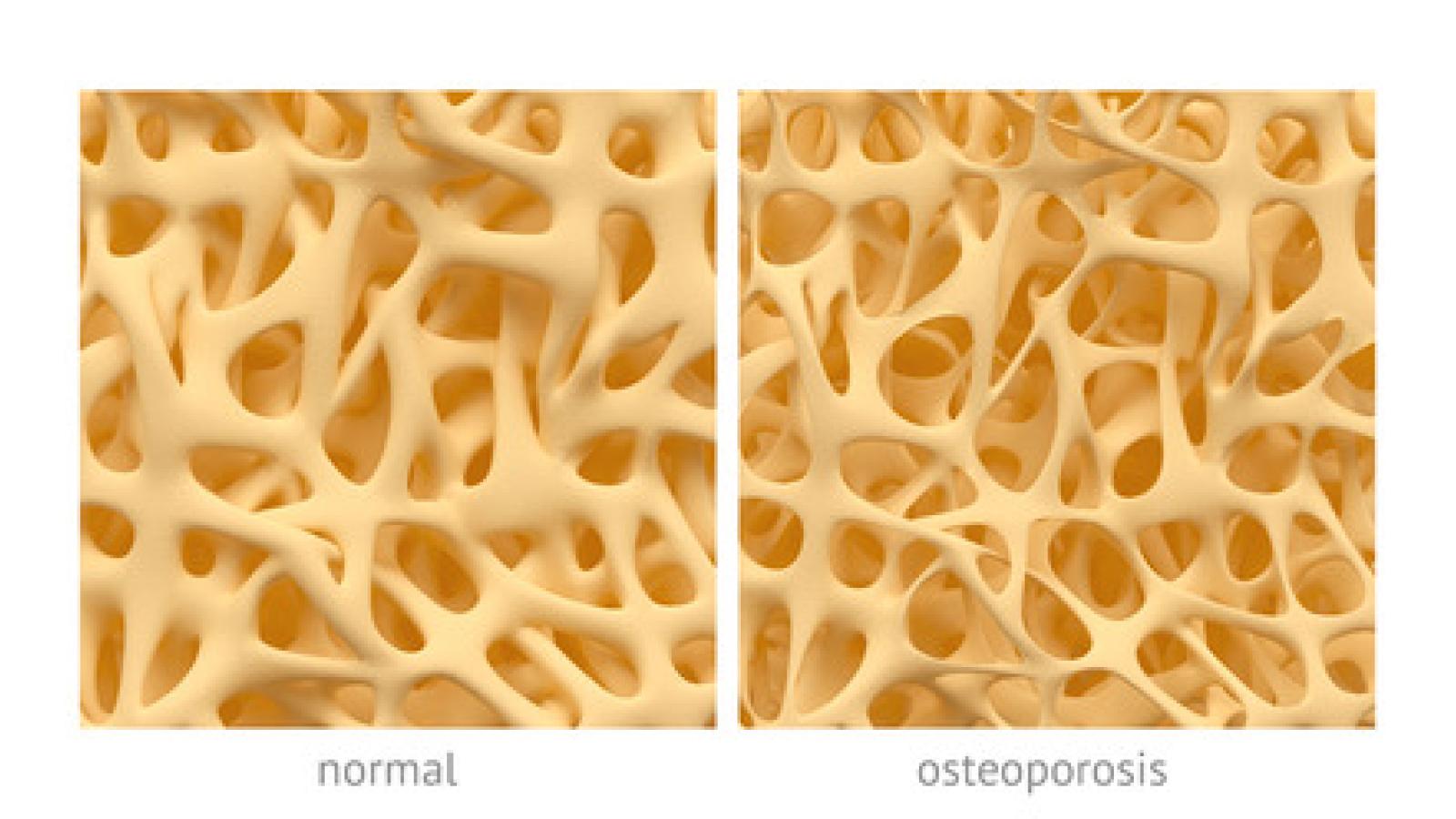
The ACR has updated this guideline and includes recommendations on abaloparatide (PTHrP) and romosozumab, which are newly available since the ACR’s 2017 GIOP guideline.

Dr. John Cush RheumNow
3 years 8 months ago
The 2022 ACR abstracts are published and posted for you. Let the Learning begin! https://t.co/ZqMAc8jV3U https://t.co/3iCGnzsPI5

Two large RA registries have shown that pregnancy outcomes in rheumatoid arthritis (RA) patients is more related to RA disease activity rather than treatments use to control RA.
Incidence of acute coronary syndrome (ACS) was significantly higher among gout patients than in the general Swedish population, researchers found, even when data were adjusted for common comorbidities.
Forbes reports that a recent Merritt Hawkins survey shows that patients are waiting an average of 26 days for a scheduled appointment with a doctor.
This is based on a 15 U.S. city survey of more than 1,000 physician offices - including family medicine, dermatology, obstetrics/gynecology, orthopedic surgery and cardiology practices.
The average wait time is up 8% from 24.1 days in 2017 (It was 21 days in 2004).
Deucravacitinib (Sotyktu), a first-in-class, oral, selective, allosteric tyrosine kinase 2 (TYK2) inhibitor, is the only approved TYK2 inhibitor worldwide and the first innovation in oral treatment for moderate-to-severe plaque psoriasis in nearly 10 years.
Can we predict the bad outcomes? Like when ITP evolves into SLE; or when psoriasis will develop arthritis; or if Sjogren's will develop lymphoma? Let's dive in and review these journal reports and this past week's news from RheumNow.com.

Dr. John Cush RheumNow
3 years 8 months ago
JAKi inhibit Vaccine responses some - study of 113 RA fully vaccinated pts assess 6wks post 2nd jab - 51 on JAKi and 62 not on JAKi. Pts not on JAK 77.4% had humoral response; 29% less (54.9%) in thos on JAK inhibitors; even more if on MTX + JAKi https://t.co/NtN4XEa2nE https://t.co/0Msto77iUK

Dr. John Cush RheumNow
3 years 8 months ago
Rheums! Have a Rheumatology question or case for Jack Cush? Record it here and we may feature it on an upcoming podcast. Tell us your name and where you practice rheumatology.
https://t.co/1aSrRsLkel https://t.co/gmh155ty6w


Dr. John Cush RheumNow
3 years 8 months ago
STARTER study looked at US in 256 RA pts completed the study & were Rx w/ biologics csDMARDS or combination DMARDs. Pts w/ US grey scal or power doppler tenosynovitis better controlled in combination DMARDs than csDMARDs alone (p:0.025) https://t.co/MXLVoIxzmw https://t.co/8N5aV3vAA0
Rheumatoid arthritis patients with existing interstitial lung disease (ILD) had less decline in lung function when receiving the antifibrotic agent pirfenidone (Esbriet) relative to placebo in a randomized trial, researchers reported here and in a simultaneous journal publication.
Among U.S. veterans receiving hydroxychloroquine (HCQ) as long-term therapy for rheumatoid arthritis, development of long QT syndrome was rare and not markedly more common compared with similar patients treated with other agents, researchers said.












 Poster Hall
Poster Hall