JAK/TYK2

Dr. Rachel Tate
2 years 5 months ago
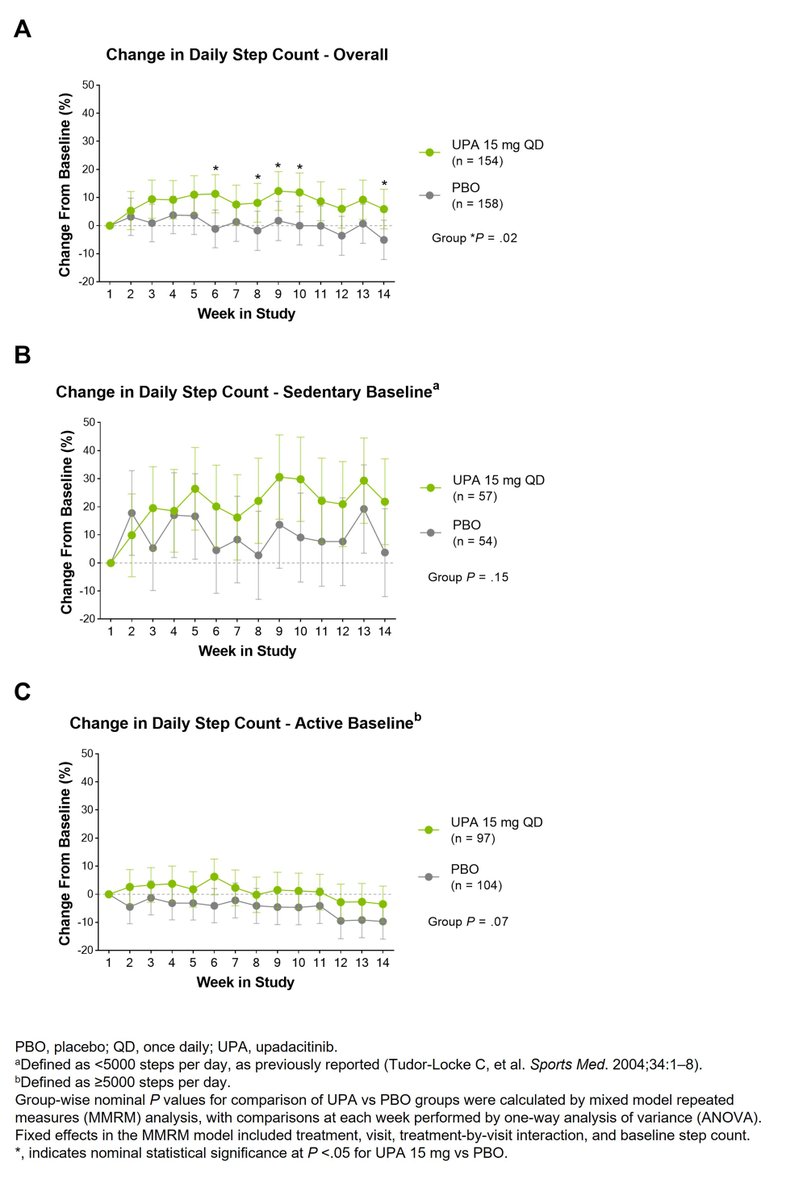
In bDMARD-IR AS patients, UPA led to numerically greater improvements vs PBO in physical activity as measured by a wearable device over 14 wks, especially in sedentary patients. #ACR23 Abs #0530 https://t.co/6Me2XctK6D @rheumnow https://t.co/c6E2GfAGt4

Dr. John Cush RheumNow
2 years 5 months ago
Join us tonight at 6pm PT/ 9pm ET for our daily #ACR23 recap. https://t.co/0RDwTdzhwS


Dr. John Cush RheumNow
2 years 5 months ago
Good Treatment review of biologics (IL-1, IL-6) in systemic JIA (Stills) including complications of #MAS and lung disease; also info on horizon therapies like JAK inhibitors and IL-18 targeting and novel biomarkers (calgranulins, IL-18 and CXCL9) https://t.co/ACe7nDDwop https://t.co/lV66WerkAp

Dr. John Cush RheumNow
2 years 5 months ago
Oral Surveillance Study Alters Practice at VA
Safety risks found in a postmarketing trial with the JAKi tofacitinib (Xeljanz) appear to have influenced prescribing patterns across the class for RA patients in the VA health system, researchers found.
https://t.co/6WktHCkrI1 https://t.co/8wpAeLsBbw


Dr. John Cush RheumNow
2 years 5 months ago
H. Zoster Subunit Vaccine Efficacy with JAK Inhibitor Therapy
Two recent reports suggest variable efficacy when JAK inhibitor (JAKi) treated patients are vaccinated with the recombinant herpes zoster subunit vaccine (RZV).
https://t.co/QEPQJkfSjE https://t.co/pFk2mF22ex

Dr. John Cush RheumNow
2 years 5 months ago
Analysis of German RABBIT registry did not show more MACE events in pts w/ JAK inhibitors compared to other DMARDs. 154 MACE w/ 14203 Rx episodes (21218 Pt-Yrs). IRs were 0.68 JAKi, 0.62 TNFi, 0.76 bdMARDs and 0.95 csDMARDs; higher in CV risk pts https://t.co/luUK9M9IM9 https://t.co/z8lxjWscCR


Dr. John Cush RheumNow
2 years 5 months ago
Metanalysis of 26 studies (2252) pts w/ HBsAg-/HBcAb+ RA treated with b/tsDMARDs, shows the pooled HBV reactivation rate was 2.0%
HBV reactivation higher w/ RTX (9%), ABA (6%), JAKi (1%); none w/ IL-6i or TNF-i
More reactivation if pt is HBsAb- (OR=4.56) https://t.co/V60nkUSpjj https://t.co/jnZKnY0Zuk

Dr. John Cush RheumNow
2 years 5 months ago
Cardiovascular and VTE Risk w/JAKi Treatment of Skin Disorders
A JAMA systematic review and meta-analysis has shown the use of JAKi in immune-mediated inflammatory skin diseases was not associated w/ increased risk of all-cause mortality, MACE, or VTE.
https://t.co/XYAeLnFvHr https://t.co/tGRNEUEZj9

Dr. Jack Cush reviews the news, journal reports and regulatory approvals from the past week on RheumNow.com.
Study of LATE-onset RA (>65y), shows DMARD initiation is low in LORA; despite current clinical practice guidelines recommending early aggressive Rx. Among 3,373 LORA pts (age77 yrs), only 29% initiated on a DMARD. https://bit.ly/47dT6x6
JAK inhibitors are known to increase the risk of herpes zoster infections between between 3-5 fold. Two recent reports suggest variable efficacy when JAK inhibitor (JAKi) treated patients are vaccinated with the recombinant herpes zoster subunit vaccine (RZV).
Safety risks found in a postmarketing trial with the Janus-associated kinase (JAK) inhibitor tofacitinib (Xeljanz) appear to have influenced prescribing patterns across the class for rheumatoid arthritis (RA) patients in the Veterans Affairs (VA) health system, researchers found.
A JAMA systematic review and meta-analysis has showed the use of JAK inhibitors (JAKi) in immune-mediated inflammatory skin diseases was not associated with increased risk of all-cause mortality, major adverse cardiovascular events (MACE), or venous thromboembolism (VTE) (compared to the placebo or active comparator cohorts).
Despite the official recognition of PMR as a distinct disease more than 60 years ago, patients with PMR are still largely treated with steroids (glucocorticoids, mostly prednisone). The persistent broad use of glucocorticoids in PMR is related to their quick initial efficacy in the majority of patients with PMR, their low price and the lack of alternative treatments and paucity of glucocorticoid-sparing treatments.














 Poster Hall
Poster Hall