Articles By Jack Cush, MD
Criteria for Early Referrals from Primary Care
Early diagnosis and early intervention has been the mantra of all who manage musculoskeletal (MSK) diseases for decades. While the rules for referral may be clear to some, the lack of uniformity and promotion amongst primary care providers is less certain.
Read Article
Mycophenolate and Steroids in Neuropsychiatric Lupus
Beyond diagnosis, management of neuropsychiatric lupus (NPSLE) can be challenging. Moreover, there are scant studies addressing optimal management. In this issue of Clinical Rheumatology, researchers show the efficacy of mycophenolate mofetil (MMF) and deflazacort in NPSLE.
Read Article
Therapeutic Update: 5 Questions on Sirukumab FDA Hearing
Drs. Cush and Gibofsky answer 5 questions about the August 2, 2017 FDA Arthritis Advisory Committee meeting that reviewed the NDA for sirukumab use in rheumatoid arthritis. The panel voted against (1-12) the approval of sirukumab.
Read Article
MSK Ultrasound Now Standard for Rheumatology Training
In the United States, musculoskeletal ultrasound (MSUS) in rheumatology has grown significantly in the last few decades. Ten years ago, more than half of rheumatology fellows had training or exposure to MSUS. A current survey of training programs shows that 94% provide MSUS training.
Read Article
FDA Arthritis Advisory Panel Endorses Tofacitinib Approval for Psoriatic Arthritis
On Thursday, August 3, the FDA Arthritis Advisory Committee (AAC) met to consider tofacitinib for use in patients with psoriatic arthritis. The panel voted 10-1 in favor of approval.
Read Article
The RheumNow Week in Review – 4 August 2017
Dr. Jack Cush reviews new FDA decisions on sirukumab and tofacitinib as well as other news items from the past week on RheumNow.com.
Read Article
Arthritis Advisory Panel Votes Down Sirukumab
On Wednesday, August 2, the FDA convened the Arthritis Advisory Committee (AAC) to evaluate the safety and efficacy of Janssen’s anti-IL-6 monoclonal antibody, sirukumab.
Read Article
Pediatric Consensus on Comorbidity Assessments
Comorbidity is a serious consequence or confounder in patients with inflammatory diseases. This has been well described in both psoriatic and rheumatoid disease.
Read Article
No Increase in Malformations with Etanercept Use During Pregnancy
While there are several reports detailing uncontrolled, observational results of biologic use during pregnancy, most are hampered by low numbers, incomplete data and too small a sample to make reliable judgements regarding the teratogenicity. A company sponsored claims data analysis has shown that etanercept exposure during pregnancy was not associated with an increase in major congenital malformations (MCMs).
Read Article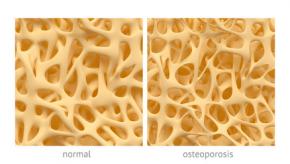
Romosuzumab Outperforms Teriparatide in Post-Bisphosphonate Osteoporosis
The STRUCTURE trial results have been reported in Lancet and have shown that after 12 months of therapy, romosozumab (ROMO) had superior gains in bone mineral density (BMD) compared to teriparatide (TER) in women with postmenopausal osteoporosis who have previously taken bisphosphonate therapy.
Read Article



