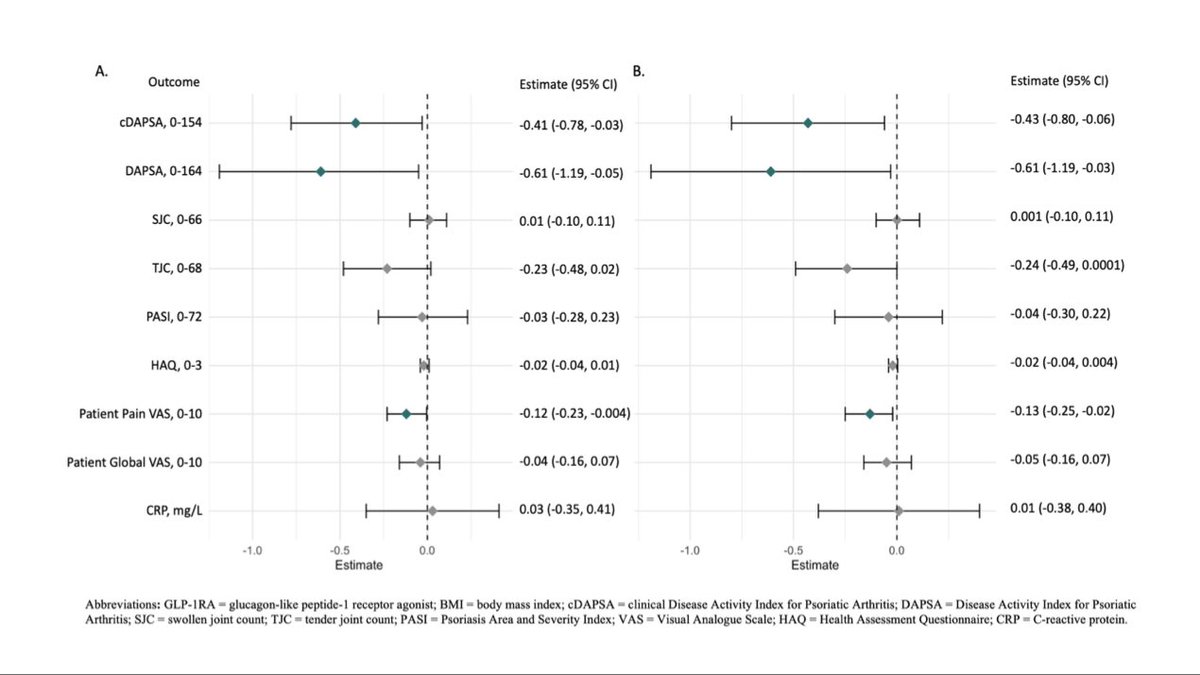
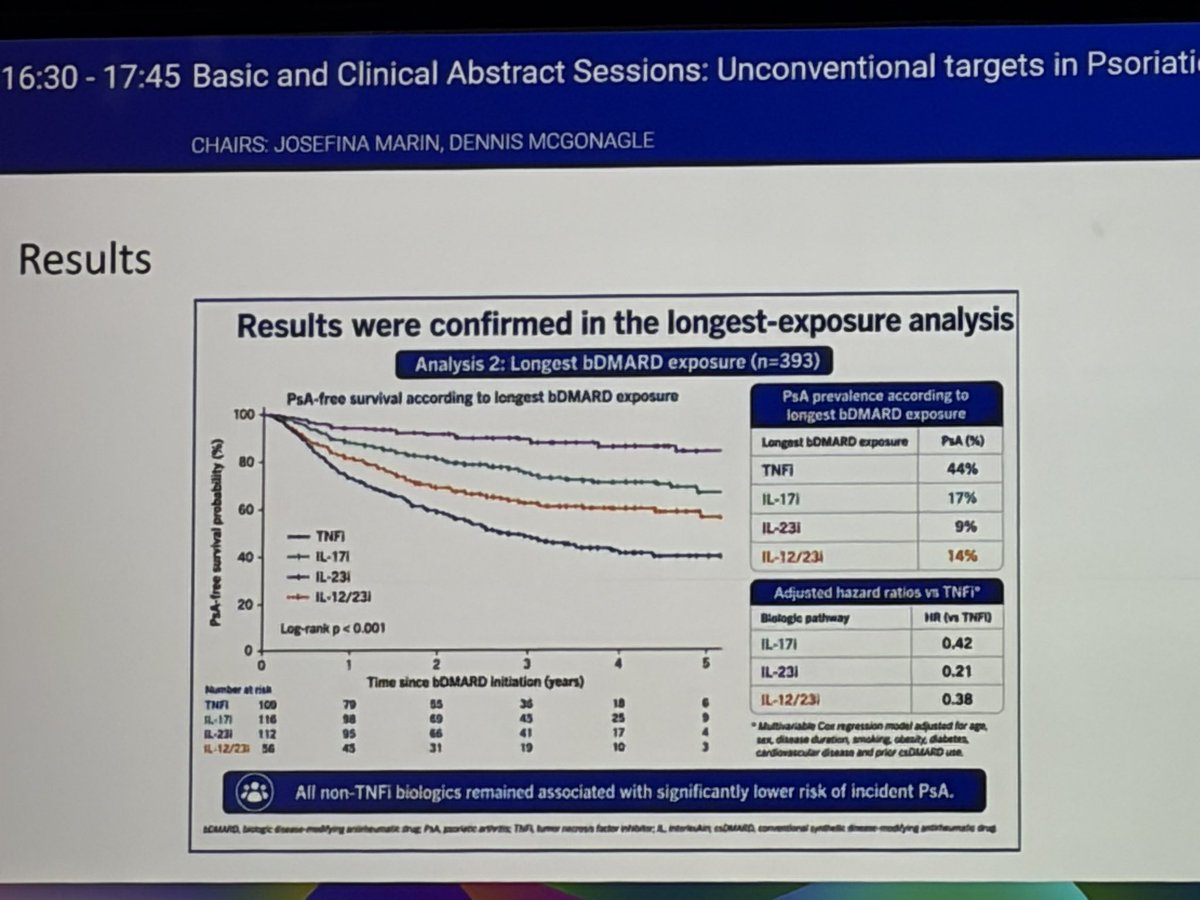
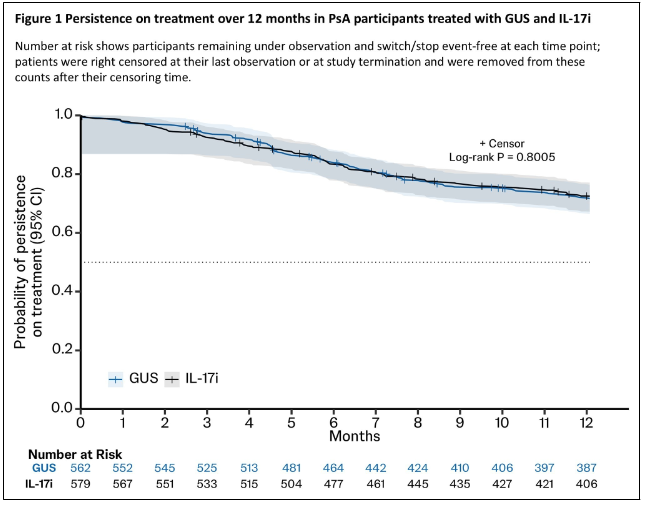
EULAR 2026: Industry Press Releases
Pharmaceutical companies are presenting pivotal clinical trial results and key data analyses at the EULAR 2026 Congress (June 3–6, London). Below is a summary of their most important studies — many of which are registration-enabling or will directly shape treatment decisions in rheumatic
Read Article