All News
Biologic Safety Guidelines from the British Society for Rheumatology
In the United Kingdom, NICE has looked to the British Society of Rheumatology (BSR) to develop evidence based guidance on the safe use of biologic DMARDs in patients with inflammatory arthritis.
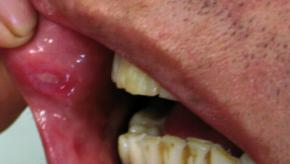
Read ArticleApremilast Reduces Oral Ulcers in Behcet's Syndrome
A multinational trial finds a statistically significant number of patients with Behçet's syndrome showed a greater reduction in the number of oral ulcers with apremilast compared with placebo.
Read ArticleVoluntary Recall of Zantac by Sanofi/FDA
Sanofi has initiated a voluntary recall of all Zantac OTC (over-the-counter) products in the United States. This includes Zantac 150®, Zantac 150® Cool Mint, and Zantac 75®.
Read ArticleFDA Approves Ustekinumab for Ulcerative Colitis
Ustekinumab (Stelara) is now FDA-approved to treat moderately to severely active ulcerative colitis, said drugmaker Janssen on Monday, in addition to its indications for psoriasis, psoriatic arthritis, and Crohn's disease.
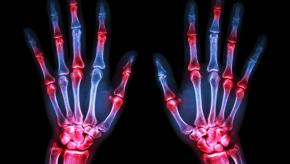
Read ArticleComorbidity Worsens Axial Spondyloarthritis
Comorbidities are common in patients with axial spondyloarthropathy (axSpA), and a recent study has shown that multimorbidity, the coexistence of 2 or more conditions, is associated with more severe disease than those without comorbidities.
Read ArticleUstekinumab Efficacy in Ulcerative Colitis
The NEJM reports the results of a one year trial wherein ustekinumab (UST) was shown to be effective at inducing and maintaining remission in patients with moderate-to-severe ulcerative colitis. Ustekinumab, currently approved for use in psoriasis, psoriatic arthritis and Crohn's disease, is a monoclonal antibody against the p40 subunit of interleukin-12 and interleukin-23.
Read ArticlePersistent Inflammatory Arthritis After Immune Checkpoint Inhibitors
Braaten and colleagues from Johns Hopkins School of Medicine have reported their experience with chronic inflammatory arthritis induced by immune checkpoint inhibitor therapies, showing that in some, inflammatory arthritis persists after the immunotherapy has been discontinued.
Parenteral Out-Performs Oral Weekly Methotrexate
A systematic review in PLOS suggests that parenteral MTX therapy is more successful than oral MTX in achieving optimal disease activity control.
Read ArticleRise in Fatty Liver Disease Linked to Obesity and Diabetes
Gut has published population results from NHANES population study showing that the prevalence of fatty liver disease is rising in the US and is driven by obesity and diabetes.
Read ArticleFDA AAC Splits Vote in Favor of Nintedanib for Scleroderma Interstitial Lung Disease
The FDA convened Arthritis Advisory Committee to consider nintedanib for the treatment of systemic sclerosis-associated interstitial lung disease (SSc-ILD) and voted 10-7 in favor of approving the drug for this new indication.
Read ArticleRisk of Major Gastrointestinal Bleeding With New vs Conventional Oral Anticoagulants
The comparative risk for gastrointestinal bleeding (GIB) with non-vitamin K antagonist oral anticoagulants (NOACs) was compared to vitamin K antagonists or anti-platelet agents in a recent metanalysis, which showed no significant difference in major GIB events between these agents.
Read ArticleEMA Restricts Tofacitinib Dosing
The Pharmacovigilance Risk Assessment Committee of the European Medicines Agency issued recommendations limiting the use of Xeljanz (tofacitinib) 10 mg twice daily in patients with ulcerative colitis in the EU.
The new recommendations are temporary while PRAC undertakes a review of all available evidence on the safety and efficacy of tofacitinib. The review follows warnings of an increased risk of pulmonary embolism (PE) and death from the U.S. Food and Drug Administration based on Pfizer's large post-marketing safety study (in high risk rheumatoid arthritis patients with one or more underlying cardiovascular risk factors) wherein those receiving in tofacitinib 10 mg twice daily in study A3921133 had more PE and mortalities than comparator groups (tofacitinib 5 mg bid or adalimumab).
FDA Final Guidance on Interchangeability with Biosimilars
The FDA has published an industry guidance document to define interchangeability as regards to biosimilar use in the United States under section 351(k) of the Public Health Service Act (PHS Act) (42 U.S.C. 262(k)).
Read ArticleDermatology Guidelines for Psoriatic Comorbidities
The JAAD has published joint guidelines from the American Academy of Dermatology (AAD) and National Psoriasis Foundation (NPF) on the approach, management and dermatolgist roles for select comorbid conditions.
Read ArticleRheumNow Podcast - Death and Surgery (2-22-19)
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com:
Read ArticleLupus Microbiome May Drive Disease Activity
Silverman and colleagues have published their study of the fecal microbiome of systemic lupus erythematosus (SLE) patients and found roughly five times more gut bacteria known as Ruminococcus gnavus, and that these abnormalities in microbiota can correlate with measures of disease severity i
Read ArticleSafety Warnings for Higher Dose Tofacitinib
Pfizer has issued a press release regarding the safety of tofacitinib, citing higher rates of pulmonary embolism and death associated with the use of higher dose tofacitinib 10mg bid in rheumatoid arthritis (RA) patients treated in a long-term safety trial.
Read ArticleRheumNow Podcast - Scurvy and Mechanics (2-1-19)
Dr. Jack Cush reviews new journal articles, news reports, and more from the past week on RheumNow.com.
Read ArticleLow IBD Risk with Secukinumab
The interleukin-23 (IL-23)/IL-17 axis plays an important role in inflammation and infection. Some of these chronic immune-mediated inflammatory disorders include PsO, PsA, AS, and IBD. Patients with PsO, PsA, and AS have up to a four-fold risk of developing IBD versus the general population.
Read ArticleHepatic Safety of Urate Lowering Therapies in Gout
Lee and a group of Korean investigators have analyzed the comparative safety of allopurinol and febuxostat in a small cohort of gout patients with fatty liver disease (FLD) and found febuxostat had a lower risk for hepatotoxicity (defined as AST or ALT at least 3× the upper limit of normal)
Read Article