All News
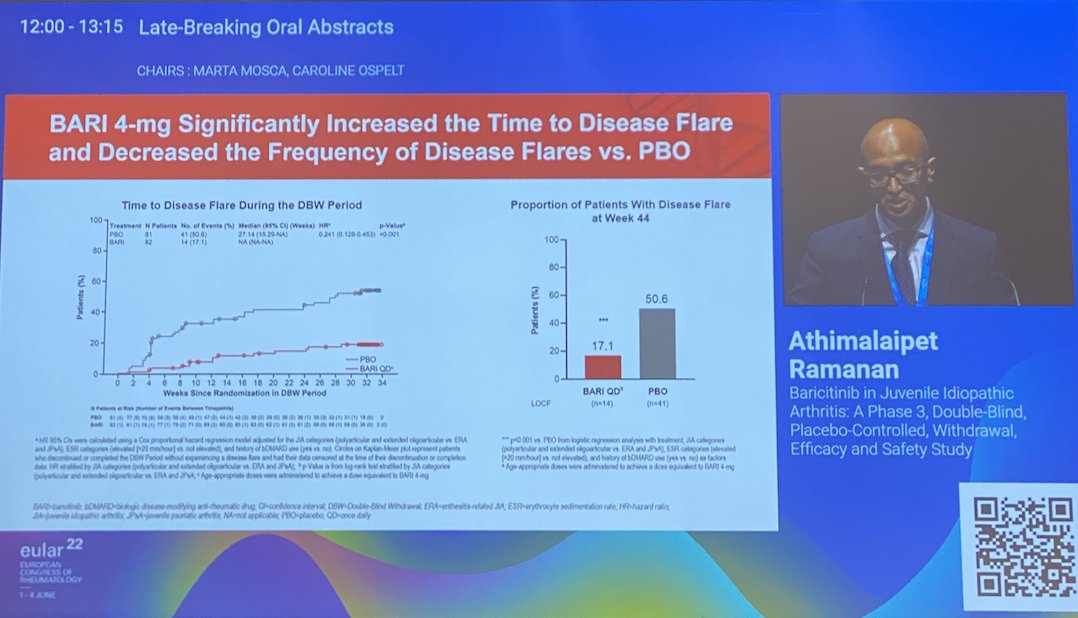
JUVE-BASIS Baricitinib in JIA
Phase 3 RCT wk12
Cs or BioDMARDs IR
⚡️Time to disease flare HR 0.24
⚡️JIA-ACR50% 63%
⚡️% flares 17% vs. 50% in PBO
Safety profile similar than adults
@RheumNow
#EULAR2022 LB0002 https://t.co/dX3DpB4QHT
Aurelie Najm AurelieRheumo ( View Tweet)
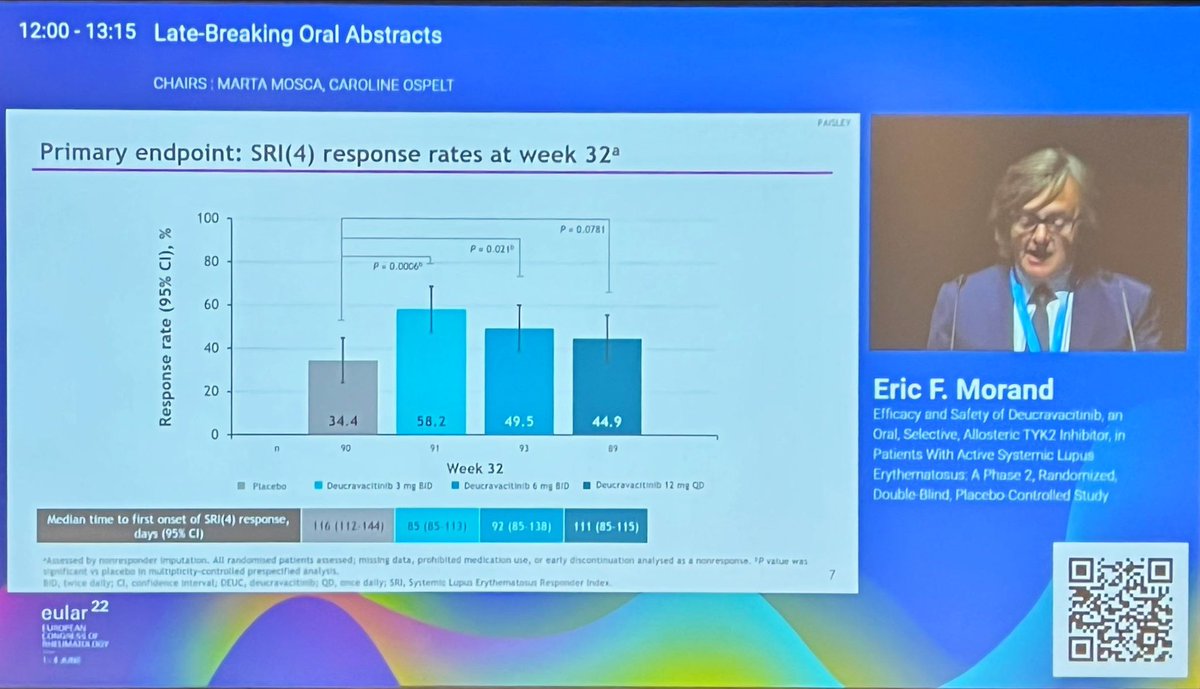
@EricFMorand up first in late-breakings
PAISLEY: deucravacitinib (TYK2i) ph2 in SLE - add-on, steroid wean
Good outcomes:
primary: SRI(4) at 32w
skin/joint/LLDAS & dsDNA/C4
Safety good (like PsO/PsA) but higher dose less so
Watch for: other clinical, ph3
#EULAR2022 @RheumNow https://t.co/XOQWuiHkTx
David Liew drdavidliew ( View Tweet)
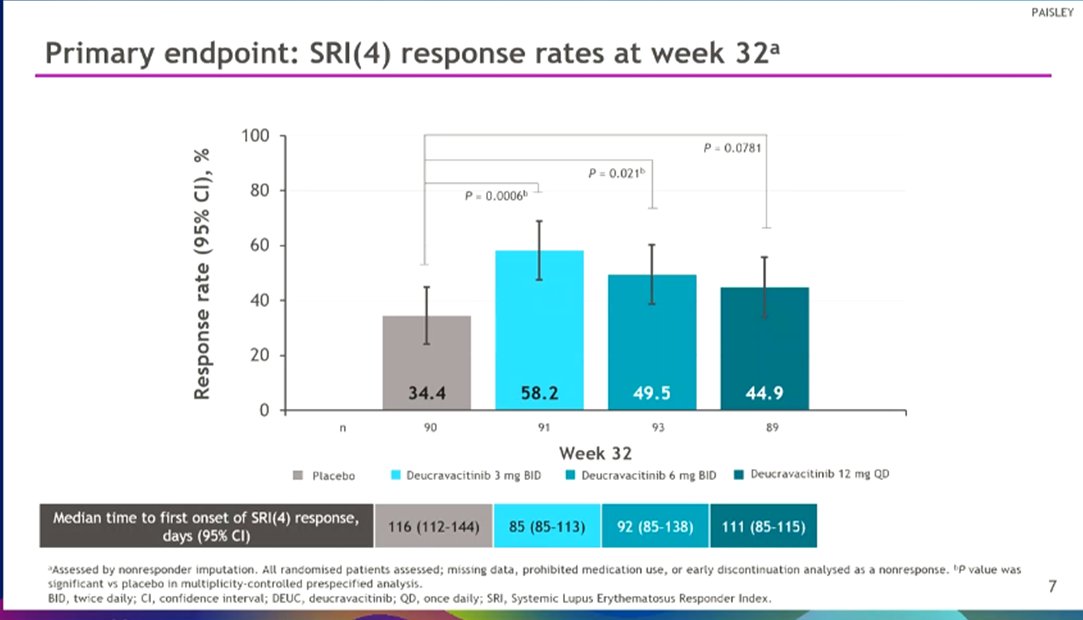
Wow! Phase 2 #deucravacitinib data for #SLE met primary & secondary endpoints: SRI(4) response, BICLA, LLDAS, CLASI-50. AEs include: skin related events and UTIs but no increase in SIE, HZ, MACE/VTE @bmsnews LB0004 #EULAR022 @rheumnow https://t.co/bsKI0mixVT
TheDaoIndex KDAO2011 ( View Tweet)
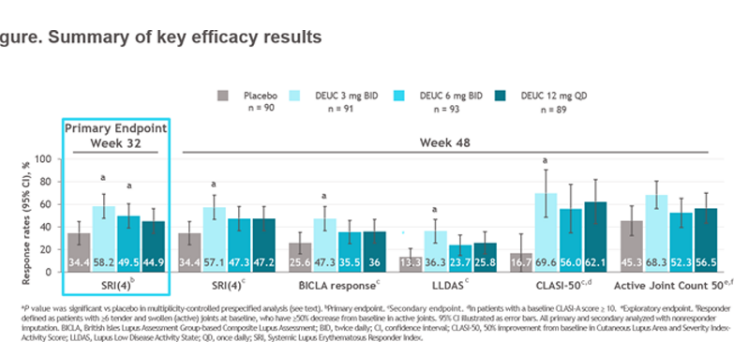
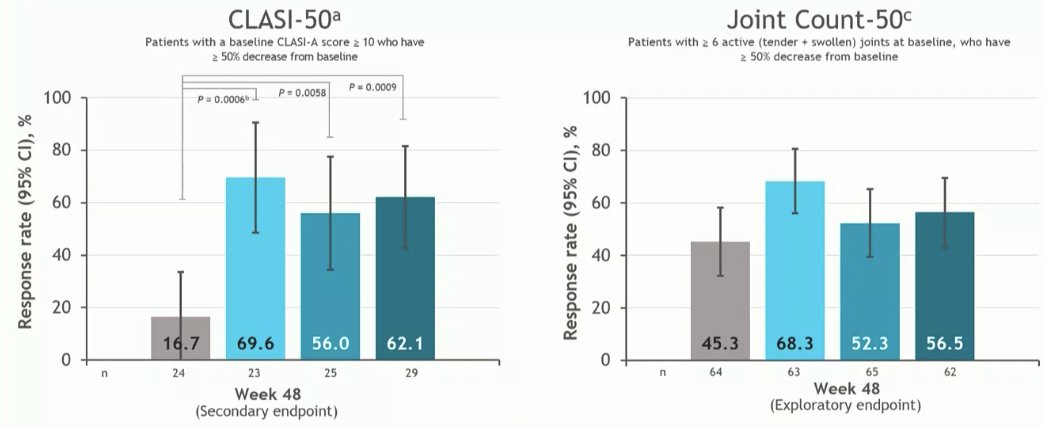
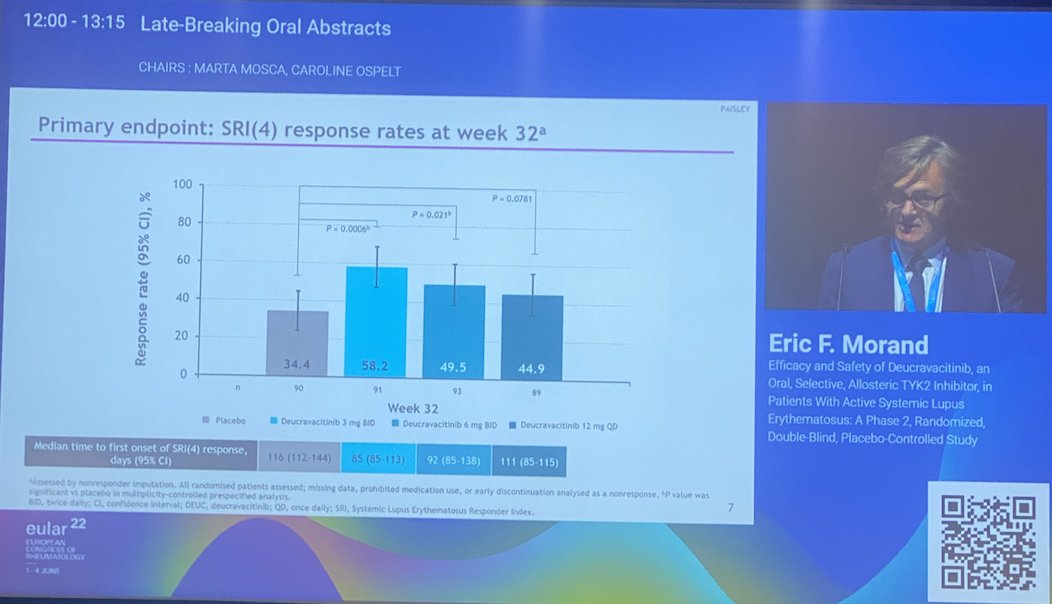
Morand et al Tyk2i deucravacitinib in SLE. 48-week phase 2 RCT. SRI(4) PBO: 34.4%; DEUC 3 mg BID: 58.2%, P =0.0006; DEUC 6 mg BID: 49.5%, P =0.021; DEUC 12 mg QD: 44.9%, P =0.078. BICLA, LLDAS, CLASI-50, active joint count also+ @RheumNow #EULAR2022 LB0004 https://t.co/MBPc9xQFvG https://t.co/n3GUwl0RtD
Links:
Richard Conway RichardPAConway ( View Tweet)
PAISLEY LB004
@EricFMorand on deucravacitinib phase 2 for SLE
⭐️Meets primary endpt: SRI(4)
⭐️Secondary endpts: BICLA, LLD, CLASI, jt count, biomarkers improved
⭐️Safety data wo VTE, CVD events
Earlier at #EULAR2022, BRAVE trial phase 3 baricitinib: No benefit in SLE
@RheumNow https://t.co/3YpPSJcenF
Eric Dein ericdeinmd ( View Tweet)
PAISLEY study Phase 2 RCT
Deucravacitinib in SLE meets primary endpoint wk 32
Results for dose 3mg BID:
*SRI(4) response 58.2% vs.
PBO 34%
*LLDAS 36%
*CLASI 70%
*⬇️ SJC
No new safety signal
Now waiting for Phase 3 👀
@RheumNow LB0004 #EULAR2022 #Lupus https://t.co/2KYSvfq4h7
Aurelie Najm AurelieRheumo ( View Tweet)
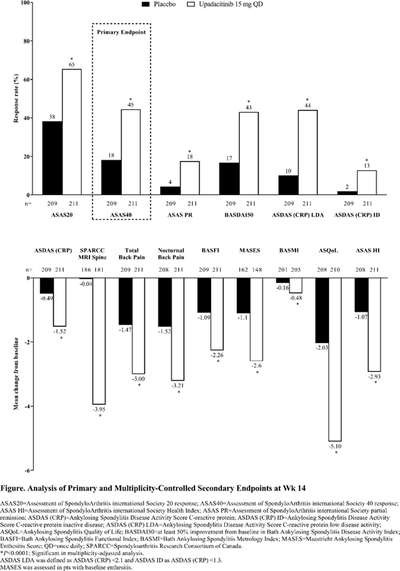
New in AS. Upadacitinib 15mg od was significantly more effective than placebo over 14 weeks in AS bDMARD-IR (ASAS40 45% vs 18% and the onset of effect seen by week 4), no new safety risks identified in the SELECT-AXIS 2 study by Van de Heijde et al #EULAR2022 @RheumNow POS0306 https://t.co/LCQBf38GWe
Dr. Antoni Chan synovialjoints ( View Tweet)
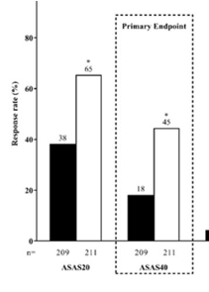
SELECT-AXIS 1 trial of upadacitinib in active #AS with an inadequate resp to prior biologic - pivotal phase 2/3 RCT of 420 AS pts (Dz dur 7.7 yrs; 83% B27+) showed better ASAS40 at wk 14 w/ UPA vs PBO (45% vs 18%; P<0.0001) #EULAR2022 POS0306 https://t.co/6ZBEvjRjqc https://t.co/GL0vlB34mE
Links:
Dr. John Cush RheumNow ( View Tweet)

Please see my short video on Promising Safety Outlook for Rituximab and Baricitinib in Vaccinated Patients #EULAR2022 @RheumNow https://t.co/bqzdWIzoOg
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)
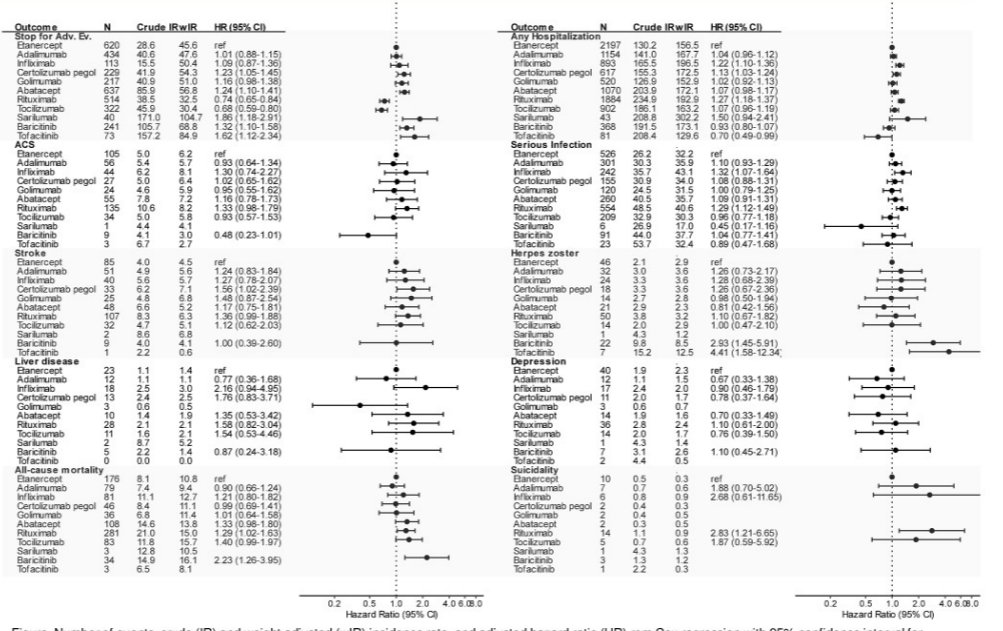
Frisell et al. Safety b/tsDMARDs from 10 years ARTIS. There is a mass of interesting data here, look at that figure! Higher rates of discontinuation due to AEs for tofa, bari, sari, rituximab. May be explained by chanelling and residual confounding @RheumNow #EULAR2022 POS0637 https://t.co/KrspU6c9Cs
Richard Conway RichardPAConway ( View Tweet)

What drives residual pain improvement in #JAKi treated pts? Dunno but #Baricitinib and #Sarilimab showed better pain decrease vs placebo and #Adalimumab. #OP0052 showed both #Tofacitinib & Adalimumab reduced pain more than placebo in PsA & RA if in remission @RheumNow @eular_org
Janet Pope Janetbirdope ( View Tweet)
Are JAKi fulfilling their promises in RA? H Schulz-Koops said a survey showed rheumatologists wanted effective fast acting Rx that could be used as monotherapy. @eular_org #EULAR2022 @RheumNow https://t.co/Gdel9YnAzA
Janet Pope Janetbirdope ( View Tweet)
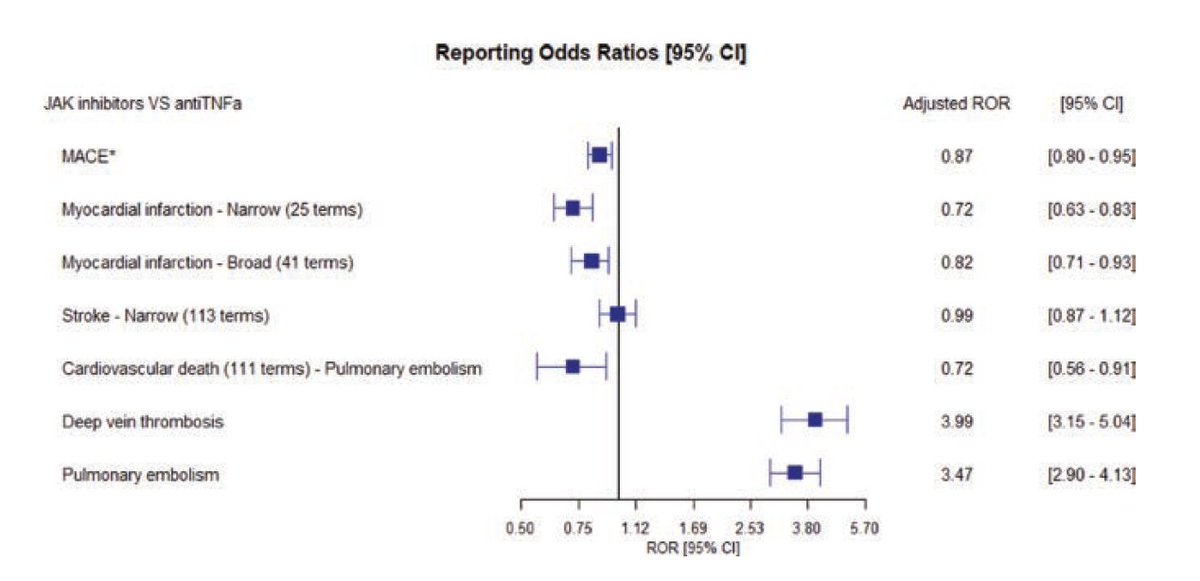
VIGIBASE registry RA
39000+ pts JAKi and 231000+ pts TNFi
*No increase in MACEs with JAKi 1.4% vs. 0.9%
*JAKi Increase in DVT RR 3.99
and PE RR 3.5
adjusted on age and sex
@RheumNow #OP0268 #EULAR2022 #Lupus https://t.co/hiAvKctgky
Aurelie Najm AurelieRheumo ( View Tweet)

Some / most of promises of #JAKi are fulfilled. Efficacy, adherence = on mono vs combination Rx with MTX. But some emerging safety issues. NK cells reduce inflammation of NK cells on fibroblasts re other MOA. @eular #EULAR2022 @RheumNow The promise of #JAKi I RA https://t.co/8Ry3vVMyzO
Janet Pope Janetbirdope ( View Tweet)
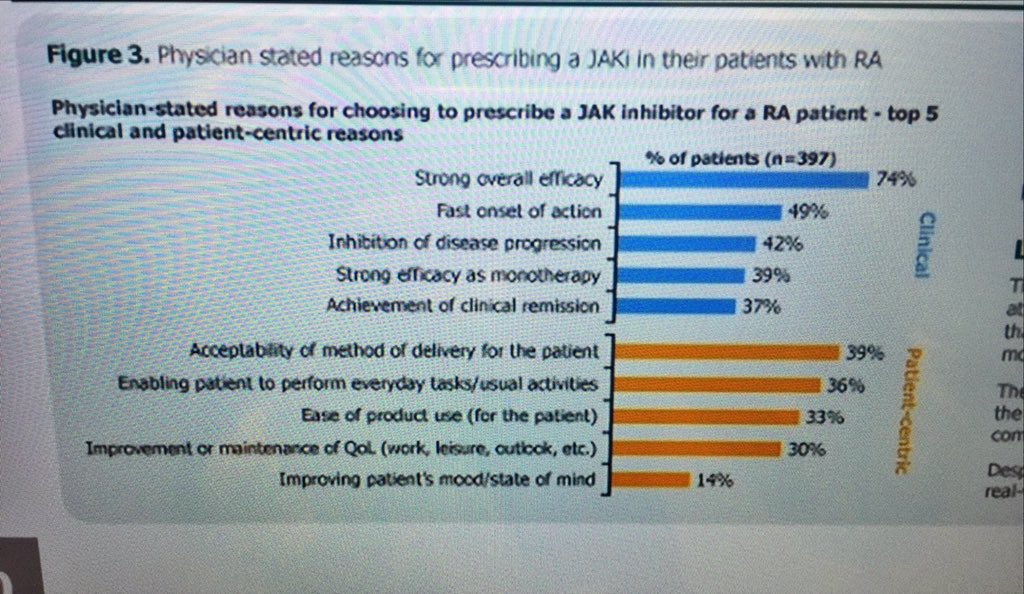
#ClinicalPearl. Why do doctors prescribe #JAKi in RA? Efficacy, fast onset, X-ray inhibition, can work as monotherapy, remission - in order of diminishing frequency BUT I will add access and oral (convenience). POS0680 @RheumNow @eular_org #EULAR2022 https://t.co/XHtNqguqvh
Janet Pope Janetbirdope ( View Tweet)
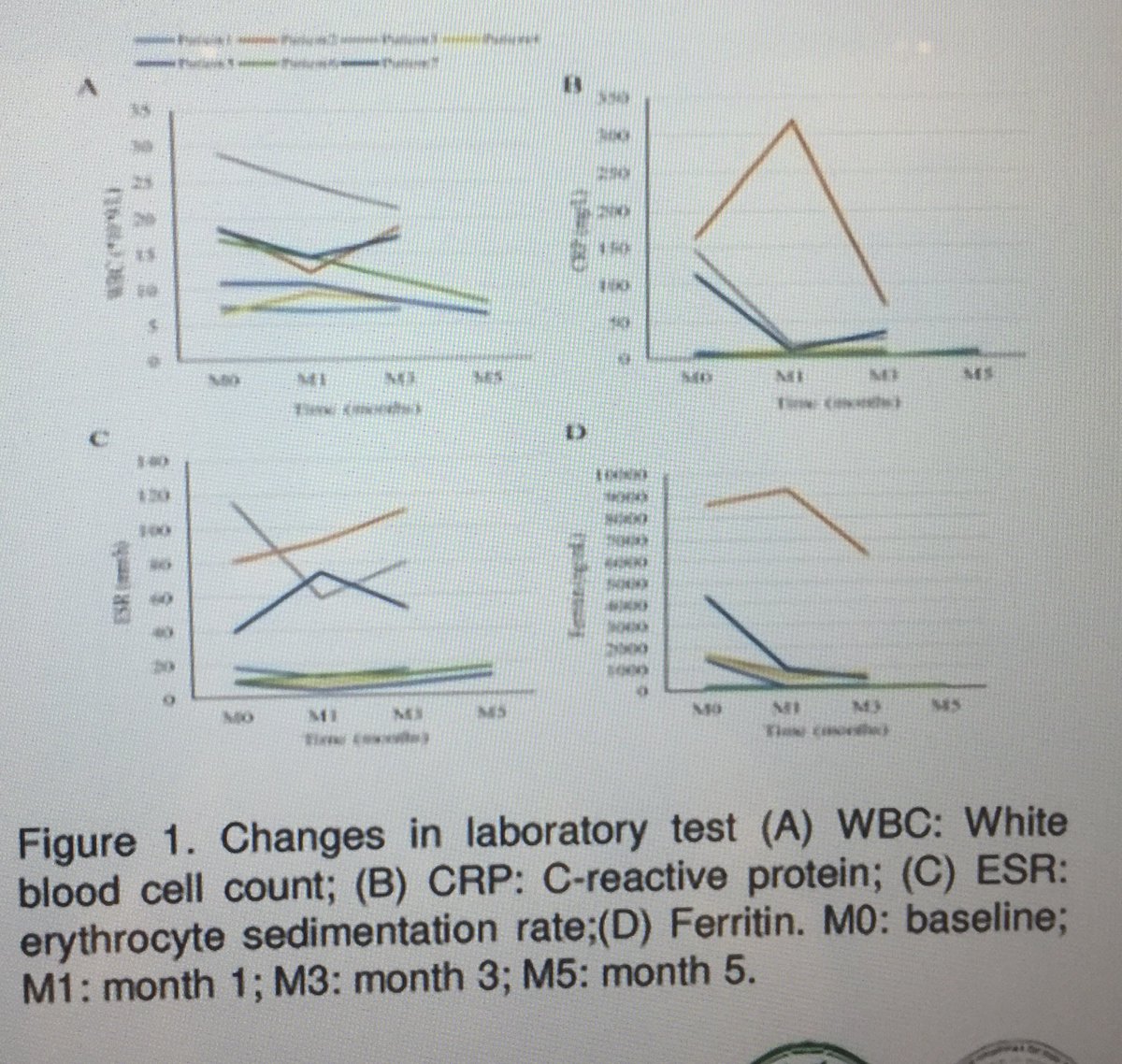
JAK will quiet Still’s disease? 7 w Still’s 4 refractory disease had benefit w #JAKi POS0014. Needs more data but looks promising most w partial response . 5 on #Tofacitinib and 2 w #Baricitinib. @eular_org #EULAR2022 POS0014 @RheumNow https://t.co/j9IRgT9XJf
Janet Pope Janetbirdope ( View Tweet)

Insightful session with H Schulze-Koops: JAKi-Are all promises fulfilled?
"JAKi have not shown us sufficient data to prove that they can induce remission or inhibit disease progression"
@RheumNow #EULAR2022 https://t.co/SKKAS1ViIU
Aurelie Najm AurelieRheumo ( View Tweet)
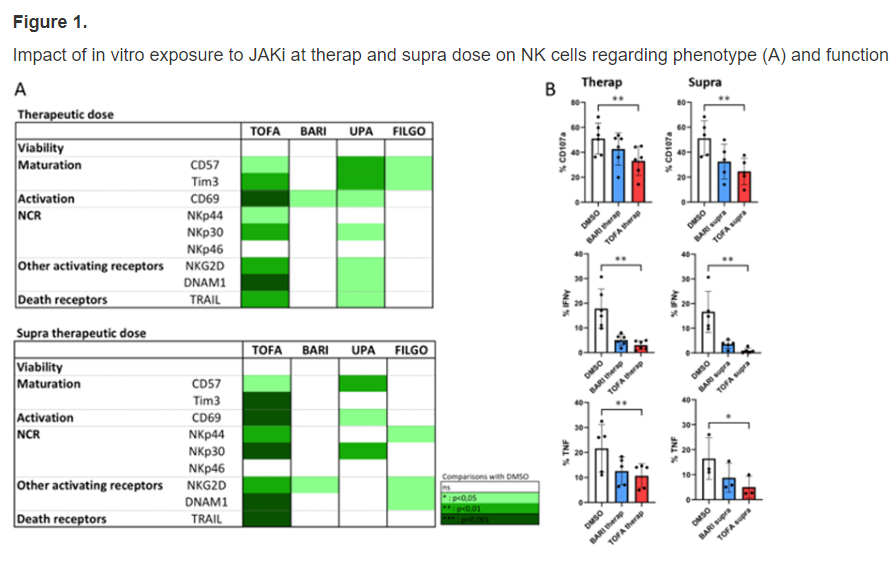
Meudec et al. Comparative effects of JAKi on NK cell function on cancer cells (lymphoma and lung cancer). Tofacitinib had most negative impact. Interesting given blanket black box based on tofa data! @RheumNow #EULAR2022 OP0265 https://t.co/XSE7hP60Ui https://t.co/MjtAJcvWAD
Links:
Richard Conway RichardPAConway ( View Tweet)
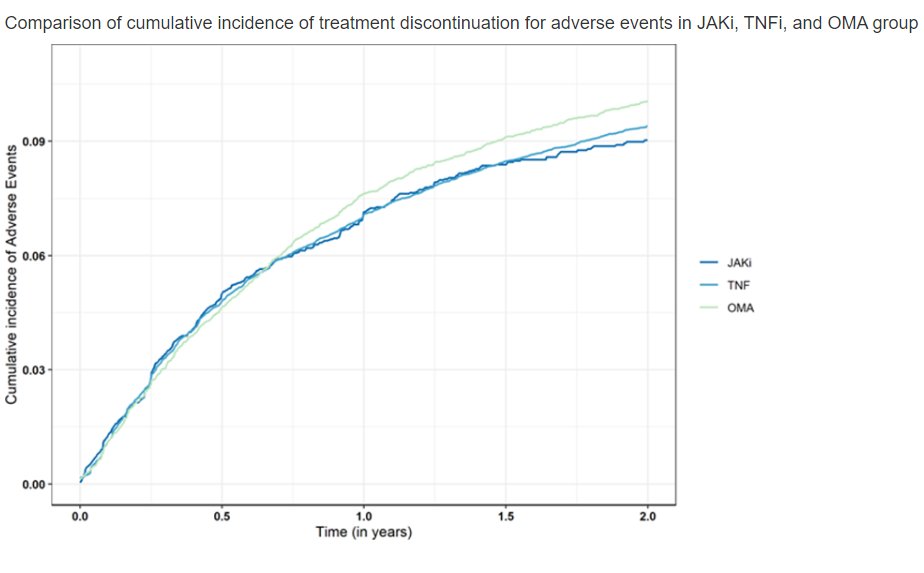
Nyam et al. JAK-pot study. >90000 treatment courses. No difference in aHR for treatment stop due to adverse events in JAKi vs TNFi or non-TNF bDMARD. @RheumNow #EULAR2022 OP0266 https://t.co/3tgADvH1Bq https://t.co/2qLRzfMB9c
Links:
Richard Conway RichardPAConway ( View Tweet)
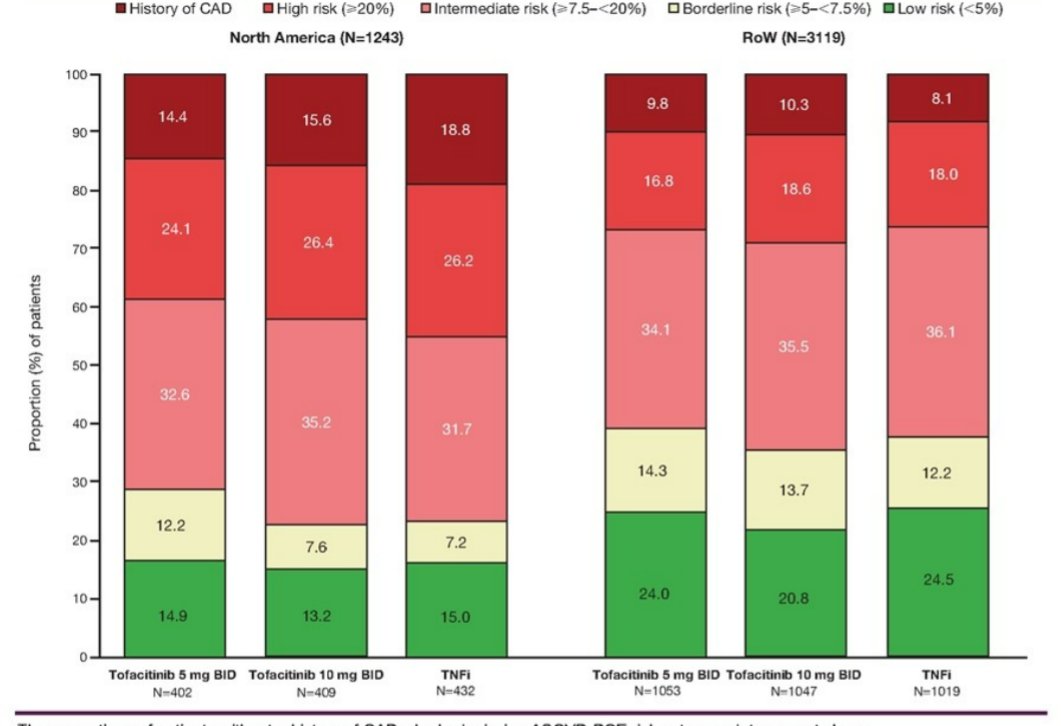
Szekanecz et al. ORAL Surveillance. Geographic differences in MACE largely driven by history CVD and high baseline risk in North America and ROW. @RheumNow #EULAR2022 POS0110 https://t.co/cR6a7N2KP5
Richard Conway RichardPAConway ( View Tweet)



