All News
ACR 2025 – Day 2 Report
Here are 3 abstracts that caught my eye on Day 2 at ACR25. Notably these have takeaway messages that should support your current practices.
Read Article
Can AI predict who stops their RA meds?
In older adults on b/tsDMARDs, interpretable ML found frailty, comorbidity & age top the list for nonadherence risk.
@RheumNow #ACR25 Abstract#2287
Jiha Lee JihaRheum ( View Tweet)

Girolami et al. VA study. Safety of DMARDs in RA following melanoma. 644 patients. 3 year all cause mortality. No significant difference, but graph sure looks like b/tsDMARDs are better. No melanoma specific mortality/recurrence data however. @RheumNow #ACR25 Abstr#2237 #ACRBest https://t.co/IN4LDkioT7
Richard Conway RichardPAConway ( View Tweet)
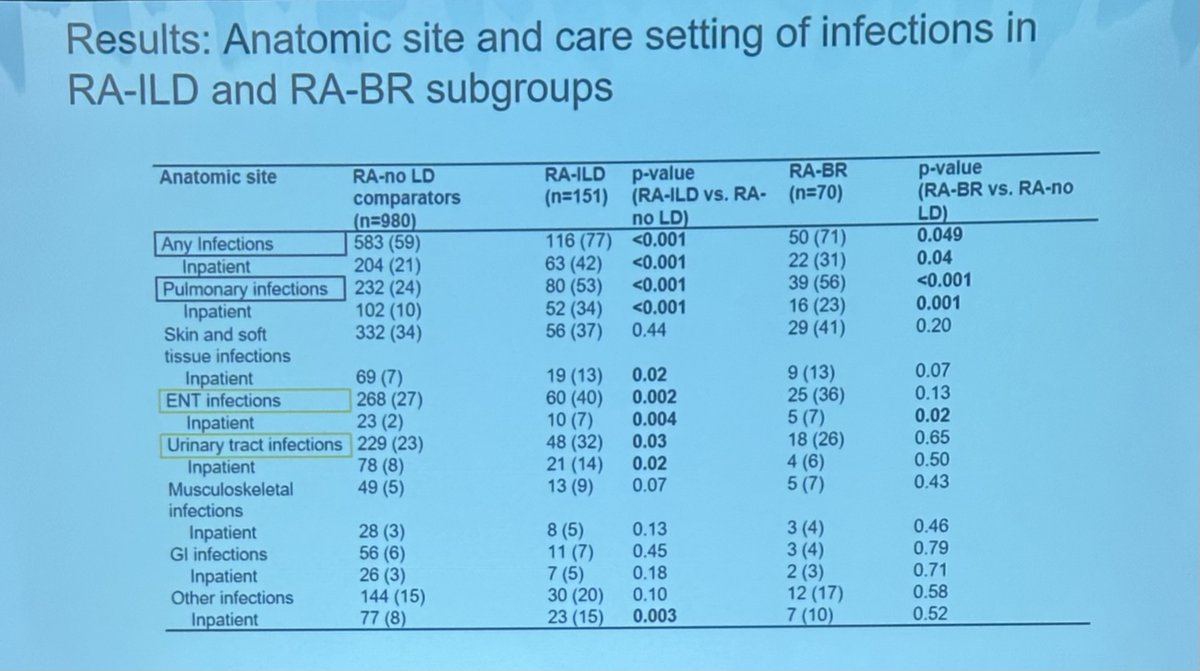
Is RA-ILD associated with/ higher risk of infection?
Retrospective US cohort study
151 RA-ILD
980 matched controls
79% higher risk of any and pulmonary infections in RA-ILD
Pathogen type: bacterial, viral and fungi
Some of these infections are preventable!
Take Home: https://t.co/fPmRjLL7Po
Links:
Aurelie Najm AurelieRheumo ( View Tweet)

In DISCOVER-2, male PsA patients had greater radiographic progression than females despite similar baseline scores. Early joint response (W8 cDAPSA LDA) was linked to less progression more strongly in men. Highlights the sex differences in PsA outcomes. Abstract#2345 @RheumNow https://t.co/zIo59FYPVg
Antoni Chan MD (Prof) synovialjoints ( View Tweet)

Does specific ANCA subtype inform infectious risk?
Retrospective review of 270 AAV patients revealed that about 1/3 pt developed severe infections... of these:
- 64.1% MPO+
- 35.9% PR3+
Most infx during year 1 of tx
No difference between CYC vs. RTX
@RheumNow #ACR25 abst 2510
Brian Jaros, MD Dr_Brian_MD ( View Tweet)

Simulation modeling by Patel et al helps generate probability of remission, relapse, and adverse events in newly dx AAV
Projected risk by model were similar to observed rates in RCTs
@RheumNow #ACR25 Abst 1764 https://t.co/xevkJ2Ih0M
Links:
Brian Jaros, MD Dr_Brian_MD ( View Tweet)

HCQ weight-based dosing: out
HCQ whole blood monitoring: in
Whole blood levels more precisely balance risks of SLE flare (under-dosing) vs. toxicitiy (over-dosing)
Many pt on <5 mg/kg had supra-therapeutic blood levels with risk for toxicity
@RheumNow #ACR25 #ACRBest Abst 1722 https://t.co/qIbACxAW39
Brian Jaros, MD Dr_Brian_MD ( View Tweet)

Passive transfer of Ab can occur w #IVIg
#Ab can occur from IVIg passive transfer incl HepB
IVIg - if pt has #cryoglobulins
👇
Can precipitate severe #cryo flare
#ClinicalPearl
Secrets & Pearls session
#ACR25 @RheumNow @ACRheum https://t.co/tTaSuVy1yQ
Janet Pope Janetbirdope ( View Tweet)

#1725 Functional NOTCH4 variants contribute to vasculopathy & fibrosis in SSc, particularly in African-American patients
NOTCH4 hyperactivation disrupts angiogenesis & drives EndoMT; inhibition (including with FDA-approved Nirogacestat) restores vascular repair @RheumNow #ACR25
Mrinalini Dey DrMiniDey ( View Tweet)

#1724 MAIT cells central in RA pathogenesis. These innate-like T cells accumulate in synovial fluid, driving fibroblast activation, cytokine release & joint damage. Blocking MAIT activation or deleting MR1 reduces arthritis in mice- a potential new target in RA. @RheumNow #ACR25
Mrinalini Dey DrMiniDey ( View Tweet)

Pooled data fr diff SLE cohorts by Dr SGarg et al were evaluated to determine an upper threshold tx range of HCQ
750-1150 ng/mL: safe & effective HCQ levels
>1150ng/mL-supratx, no added tx benefit
CKD st >/=3: 2x ⬆️odds of toxic hcq levels
#ACR25 @RheumNow Abs1722 #ACRBest https://t.co/RFx1VO0bov
Links:
sheila RHEUMarampa ( View Tweet)

#1723 Why is systemic autoimmunity more common in females?
Loss of X-chromosome inactivation maintenance in B cells (not T cells) amplifies interferon-driven disease, with increased autoantibodies, inflammation and renal injury. Key mechanistic advance. @RheumNow #ACR25
Mrinalini Dey DrMiniDey ( View Tweet)

3rd plenary session!
#1722 Defining safe HCQ levels in SLE: whole-blood 750-1150ng/mL= therapeutic range; >1150ng/mL ➡️ ~2× toxicity risk; <750ng/mL ➡️ higher flare risk
CKD ≥3 increases odds of supratherapeutic levels
New era of precision monitoring in SLE?
@RheumNow #ACR25
Mrinalini Dey DrMiniDey ( View Tweet)

Plenary 3, HCQ blood levels in SLE
HCQ level > 1150: 1.9x risk of HCQ toxicity
HCQ level < 750: 1.4x risk of active SLE
HCQ dose < 5mg/kg: 1.9x risk active SLE
My take home? Blindly reducing dose to <5mg/kg is BAD; use levels instead!
@RheumNow #ACRBest #ACR25 Abstr1722 https://t.co/UCBPAKAyon
Links:
Mike Putman EBRheum ( View Tweet)

#ACR25 Please find my video interview with Dr Kate Rochlin on a novel Bi-specific T-cell engager (BiTE) in development which binds to pan-gamma delta T-cell instead of CD3 to minimise risk of cytokine release syndrome side effect. Abstr#1535 @RheumNow
https://t.co/MOmUm41cyA https://t.co/TgIUGgWGw5
Links:
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)

PET and histologic discordance in aortitis
Of 5 aortitis patients who had PET/CT prior to aortic surgery with path, only 1 showed concordance (neg PET, neg biopsy)
4/5 patients had inflammatory biopsy with neg PET
Small # but PET is not perfect!
@RheumNow #ACR25 Abst 1620
Brian Jaros, MD Dr_Brian_MD ( View Tweet)

Abstract 1718: Immune checkpoint inhibitors (ICI) in pre-existing #SLE
Compared to non-SLE pts, those with SLE receiving ICIs had:
🔹 ↑ risk of thrombocytopenia & pancytopenia
🔹 No significant difference in all-cause mortality
@RheumNow #ACR25 https://t.co/N1iOvxbsAZ
Akhil Sood MD, MS AkhilSoodMD ( View Tweet)

IL-5 agents may in fact work for severe EGPA manifestations
Although severe pt were excluded from enrollment in MANDARA, post-hoc and OLE data suggest development of severe manifestations is rare in benra treated patients
@RheumNow #ACR25 https://t.co/slUpAT9mWo
Brian Jaros, MD Dr_Brian_MD ( View Tweet)

ANCA status does not inform IL-5 agent response in EGPA patients
Terrier et al. analyzed the OLE of MANDARA and compared ANCA+ and ANCA- subgroups:
- similar rates of remission
- similar rates of GC withdrawal
- comparable relapse rates
@RheumNow #ACR25 Abst 1594
Brian Jaros, MD Dr_Brian_MD ( View Tweet)




