All News
Psoriatic Arthritis: Hit hard and Early
EurekAlert!
Two recent studies suggest there is no significant benefit of early biologics over standard step-up care with methotrexate2,3, but these did not select for poor prognosis.
Read ArticleEULAR 2025 - Day 1 Report
Wednesday was Day One at EULAR 2025 in Barcelona. Thousands from around the world gathered, eager to reunite at this international educational forum. Below are a few of my favorites from Day 1.
Read Article
Fascinating to see JAKi uptake in European JAK-pot centres
- tofa was already on the decline pre-ORAL Surveillance
- JAKi use (driven by upadacitinib) continues to grow, although maybe it’s slowed
Hopefully rational JAKi has its place #EULAR2025 POS0150 @RheumNow https://t.co/XRyHJVw821
David Liew drdavidliew ( View Tweet)

In #SLE, #HCQ levels >1150 ng/ml ↑toxicity risk w/o added benefit. Levels 750-1150 ng/ml linked to ↓disease activity.
CKD stage ≥3 doubled HCQ levels at same dose.
Supports monitoring & dose adjustment in CKD to balance safety & efficacy.
@RheumNow #EULAR2025 #OP0199
Mrinalini Dey DrMiniDey ( View Tweet)

PHOENYCS GO🏃: Dapirolizumab pegol (DZP) led to higher rates of LLDAS (41% vs 20%) & DORIS remission (19% vs 8%) at wk48 vs PBO+SOC. DZP showed earlier & more sustained disease control, advancing T2T in SLE.
@RheumNow #EULAR2025 #OP0201 https://t.co/zuxBELxTDd
Mrinalini Dey DrMiniDey ( View Tweet)

Ph 2 RCT CPL'116 dual inhibition JAK/ROCK
106 MTX IR pts
wk12 primary endpoint met (DAS28-CRP decrease) in 240mg arm vs. PBO
75% >= AEs, SAE 2: 1 MI, 1 bladder cancer
Authors suggest absence of lipids/LFTs disruption compared to other JAKs
Ph3 is awaited!!
#OP0193 #EULAR2025 https://t.co/TnnM410LfM
Links:
Aurelie Najm AurelieRheumo ( View Tweet)

OP0195: MTX+LEF isn’t commonly used—but could it be a viable option after all?
In >1,100 RA pts, no link to liver fibrosis (FIB-4/APRI), modest ALT ↑, and fewer hospitalizations.
real-world safety data worth a second look.
@RheumNow #EULAR2025
Jiha Lee JihaRheum ( View Tweet)

ACQUIVAS: RTX-treated AAV patients had poor response to #pneumococcal vaccine PCV-13 (8% vs 45% in non-RTX). Boosting w/ PPV-23 helped a few but 48% failed to mount robust response. Time to rethink vaccine strategies in RTX-treated patients?
@RheumNow #EULAR2025 #OP0173 https://t.co/soU5qfwRib
Mrinalini Dey DrMiniDey ( View Tweet)

#MITIGATE subgroup analysis: Inebilizumab reduced flares & boosted remission across all #IgG4-RD subgroups. HR for flare vs PBO: 0.12-0.16 across sex/race. Similar results for all subgroups in achieving flare-free, treatment-free complete remission.
@RheumNow #EULAR2025 #OP0189
Mrinalini Dey DrMiniDey ( View Tweet)

There is still hope for vagal nerve stimulation in RA!
RCT RESET-RA 240+pts multibioIR
Implantable cervical device
Meets Primary endpoint ACR 20 wk12
Subgroup 1 prior bioDMARD ACR 20 wk12 46% vs 19% sham
EULAR Good response 61% vs 42% sham Wk 12
SAE rate 1.7%
#OP0190 #EULAR2025 https://t.co/DCON01XXgd
Links:
Aurelie Najm AurelieRheumo ( View Tweet)

2-year #MANDARA data: Benralizumab shows durable remission, eosinophil suppression & OGC-sparing in EGPA. ~62-68% in remission at wk104. Low relapse rate, no loss of asthma control or decline of lung function. No new safety signals.
@RheumNow #EULAR2025 #OP0166 https://t.co/kXh2q2ilJF
Mrinalini Dey DrMiniDey ( View Tweet)
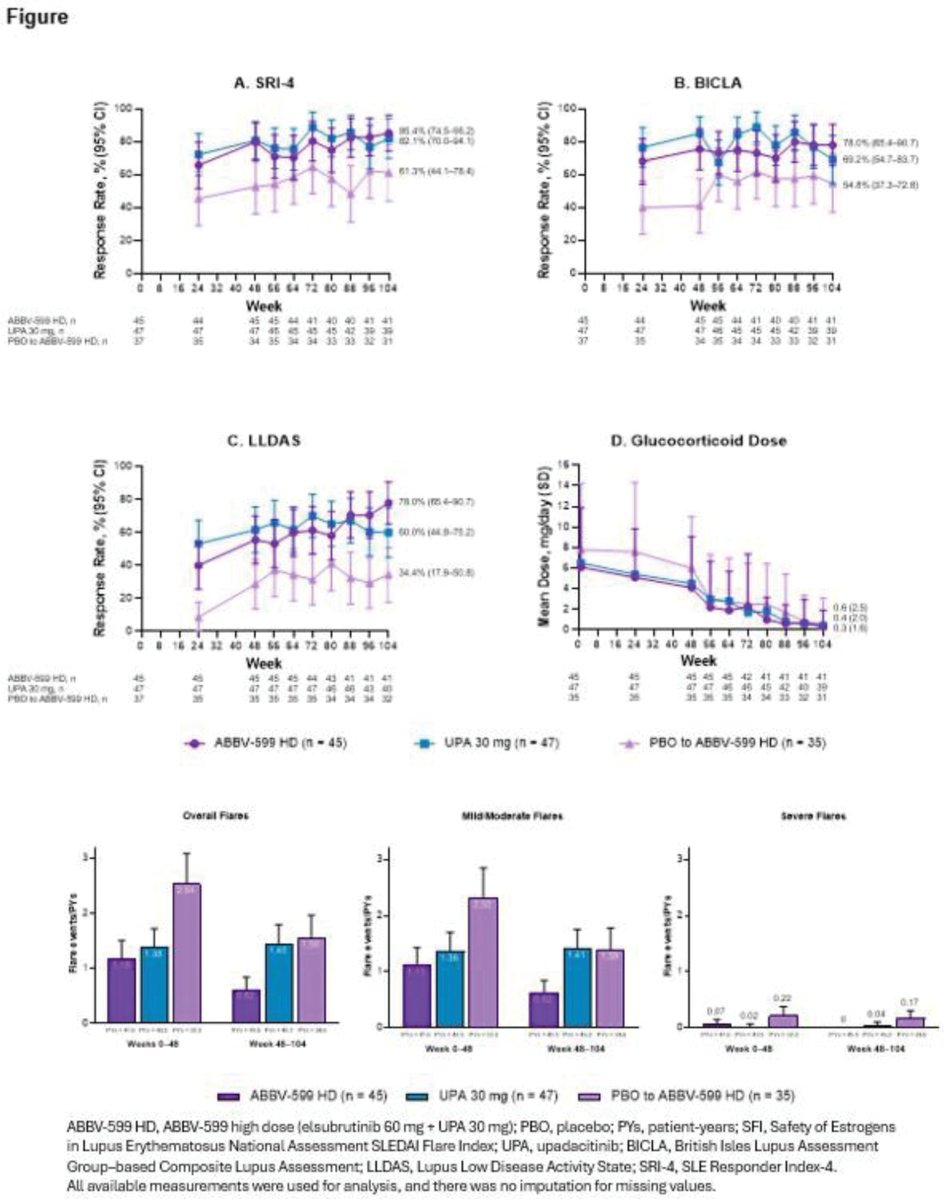
In SLEek LTE, ABBV-599 HD (BTKi+JAKi) & UPA 30mg sustained or improved disease control through 104wks: SRI-4 ≥82%, ↓flares, near steroid-free, no new safety signals. PBO-switchers improved too. Targeted oral combos look promising.
@RheumNow #EULAR2025 #OP0198 https://t.co/IHXC2iD0h6
Mrinalini Dey DrMiniDey ( View Tweet)
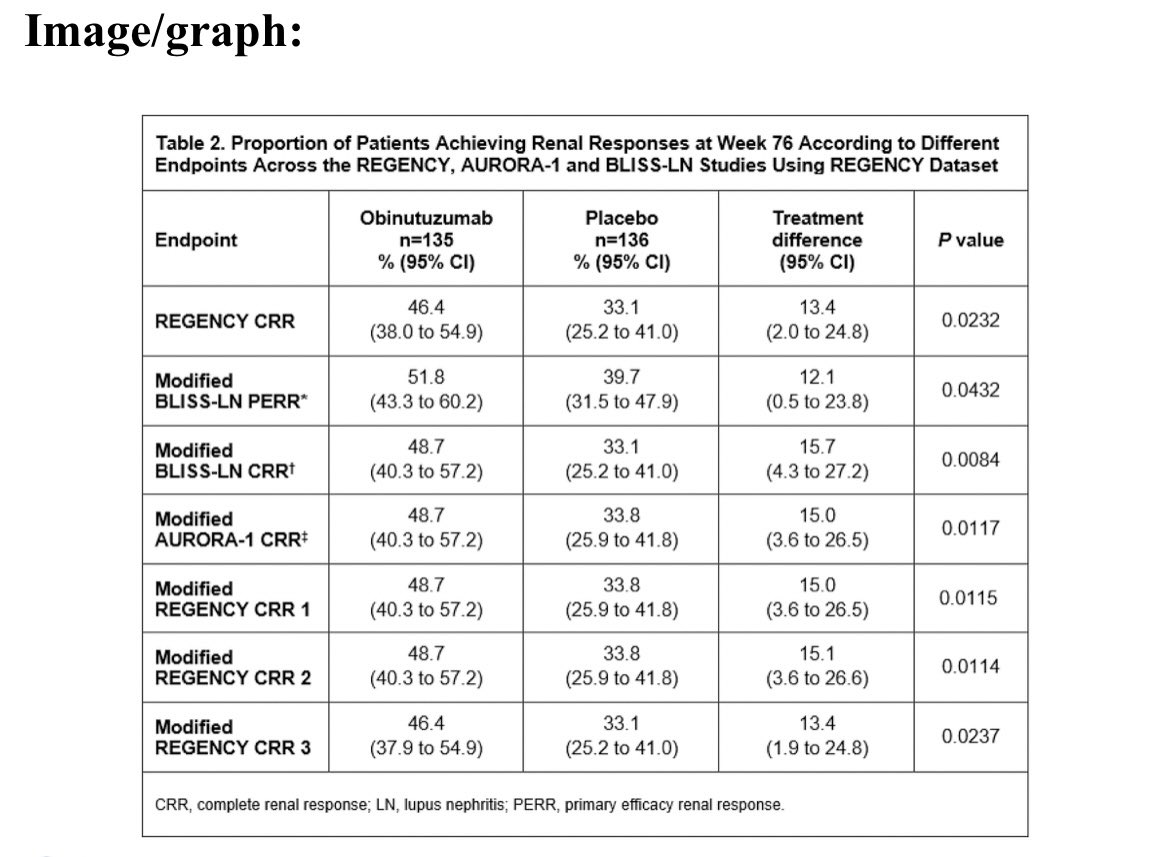
#EULAR2025 Abstr#OP0006. There is no standardised definition for complete renal response in #lupus nephritis. Post-hoc analysis of REGENCY showed Obinutuzumab + SOC was superior to PBO + SOC if endpoints for voclosporin and belimumab RCTs were used. Effect size ~13-16% @RheumNow https://t.co/q8MNAV8XF3
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)

T2T in #gout w serum #uric #acid / #urate target
SAVES LIVES
Study of achievement of target 🎯
⬇️CVE and death
If not achieving Urate <360
T2T also reduced baseline #SUA
#EULAR2025 @rheumnow abst#OP0005 https://t.co/ePchJ4vqnU
Janet Pope Janetbirdope ( View Tweet)

Secukinumab for PMR?
-post hoc analysis of the TitAIN study (phase 2 RCT on new onset/relapsing GCA) showed:
-Numerical reduction in patients experiencing PMR symptoms when treated with secukinumab compared to placebo.
-Safety profile was similar to the overall GCA study
Adela Castro AdelaCastro222 ( View Tweet)

SPEED RCT: In early PsA with poor prognostic factors, PASDAS at 24wks:
•Early TNFi: 3.7
•Combo csDMARDs: 4.1
•Step-up csDMARDs: 4.7
Early TNFi beat step-up by -1.09 (p<0.001); combo csDMARDs also superior (-0.69, p=0.02). Early TNFi benefit sustained at 48wks. Abstract#OP0089 https://t.co/MpazF8BIr2
Antoni Chan MD (Prof) synovialjoints ( View Tweet)

In vitro, balinatunfib (TNFR1-selective inhibitor) preserved Treg expansion in CD4+ T cells co-cultured with IL-2 and memTNF (Treg 8.99 percent, p<0.0001), unlike adalimumab and etanercept which reduced Tregs by 27.5 to 41 percent. Confirms TNFR2 sparing with selective TNFR1 https://t.co/xaczz87Xrd
Antoni Chan MD (Prof) synovialjoints ( View Tweet)

#EULAR2025 Abstr#OP0002 Promising new mode of action therapy for #myositis. Phase 2 RCT of efgartigimod (FcRn-i; coformulated with recombinant PH20) showed improvement in TIS & other key endpoints vs PBO at Wk24. Injection reaction common (23%). Will proceed to Phase 3 @RheumNow https://t.co/SuMolfcDkS
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)

Phase 3 of POETYK-PsA-2:
-Deucravacitinib (oral TyK2i) met the primary endpoint of ACR20 at week 16 and was superior to placebo in multiple PsA domains.
-Clinical efficacy maintained until week 52.
-No major safety signals reported.
-Notably high placebo response rates.
Abstract
Adela Castro AdelaCastro222 ( View Tweet)

📊 In a 40-yr cohort (n=2818), RA pts had 2.3x ↑ risk of DVT (HR 2.26) & 1.6x ↑ risk of PE (HR 1.61) vs non-RA.
Risk ↑ with RF/CCP+, nodules (HR 2.9), BMI ≥30 (HR 1.9), & biologic use in year 1 (HR 2.4–2.9).
Remission = ↓ PE risk (HR 0.47)
📍OP0070
@RheumNow #EULAR2025
Jiha Lee JihaRheum ( View Tweet)