All News
No Increase in Cardiovascular Events with JAK Inhibitors in IBD Patients
UMKC researchers have compared the use of JAK inhibitors versus tumor necrosis factor (TNF) blockers in inflammatory bowel disease (IBD) patients, but failed to see an increase in major adverse cardiovascular events (MACE) in JAKi vs TNFi use in IBD.
Read ArticleWeight Loss from Anti-Obesity Medications Prevents Gout
A population based study looked at the effect of the weight loss drug, Orlistat, in obese individuals. Compared to those untreated, orlistat over 1 year was associated with significant weight loss and significantly lower risks of incident gout and recurrent gout flares among overweight or obese people.
Read Article
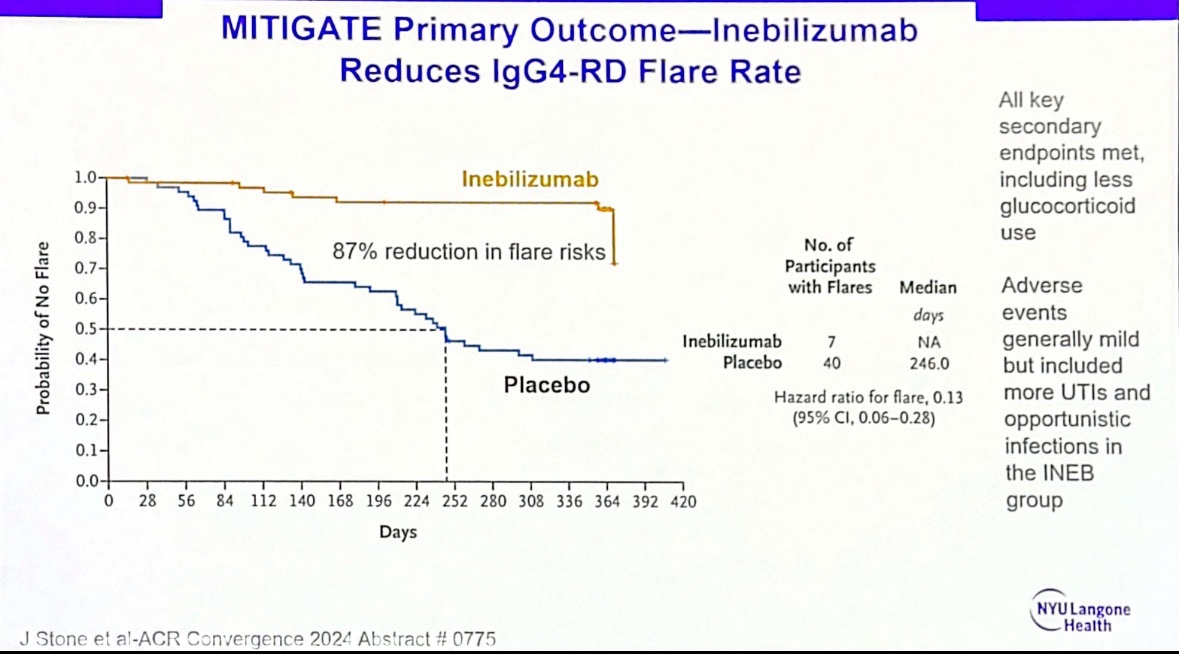
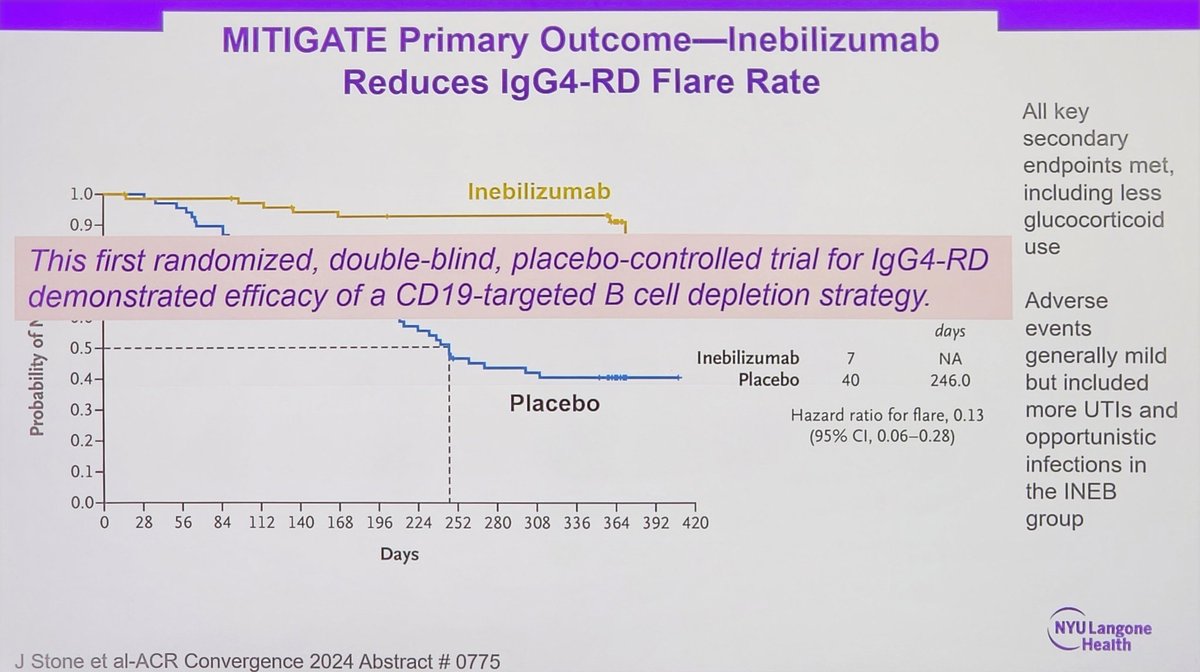
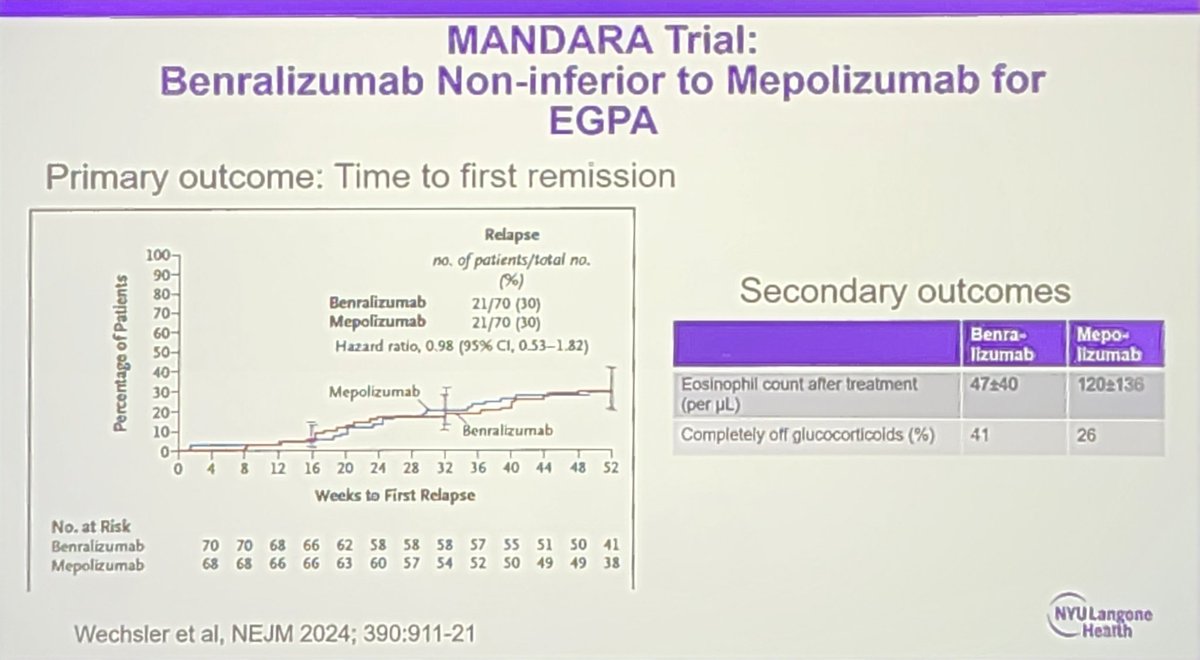
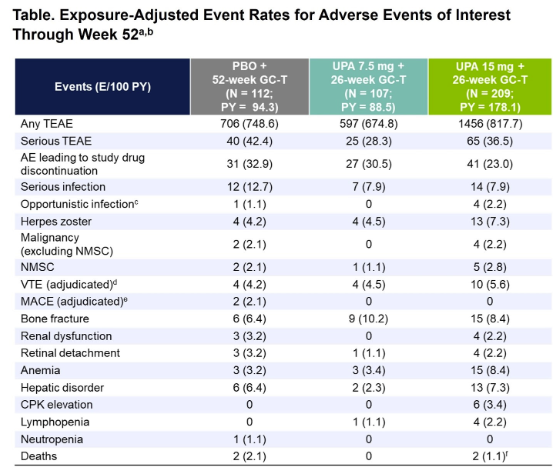
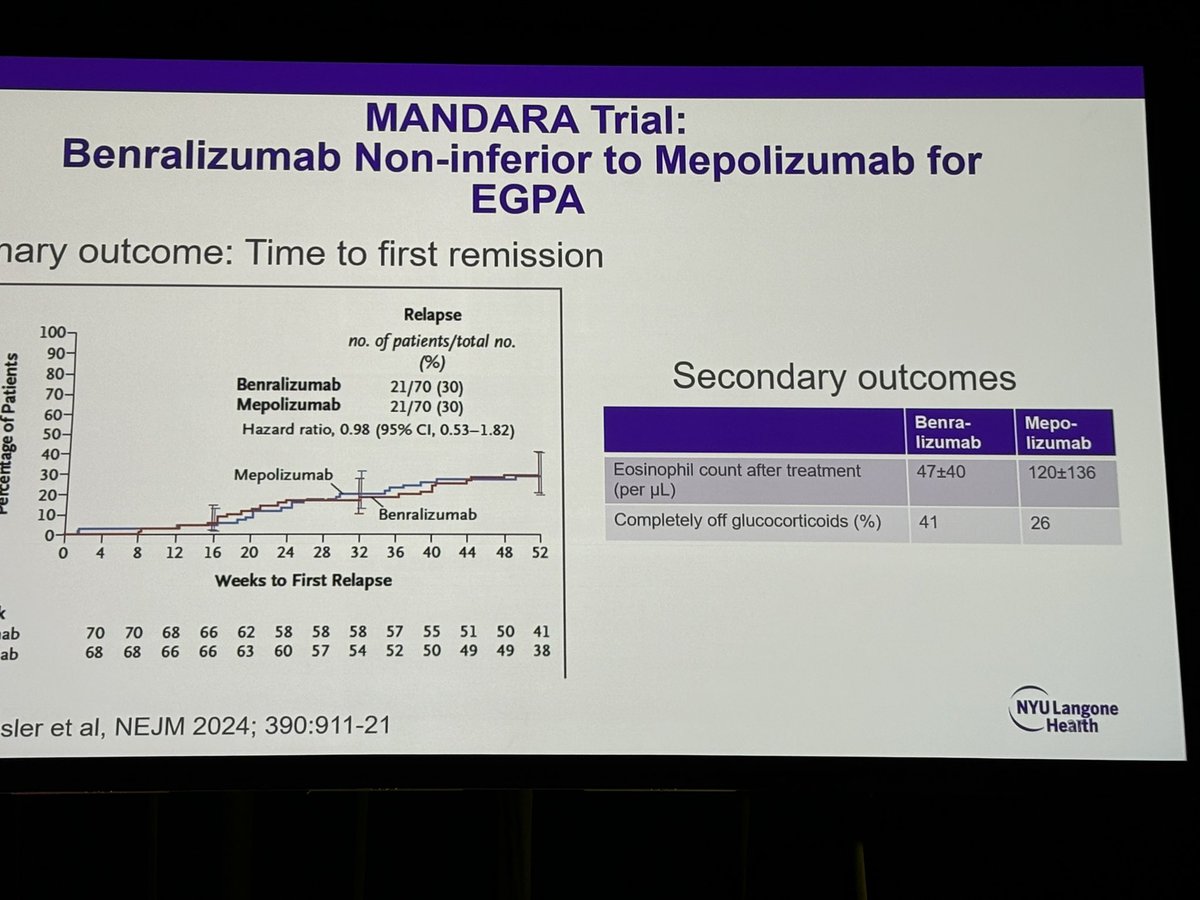
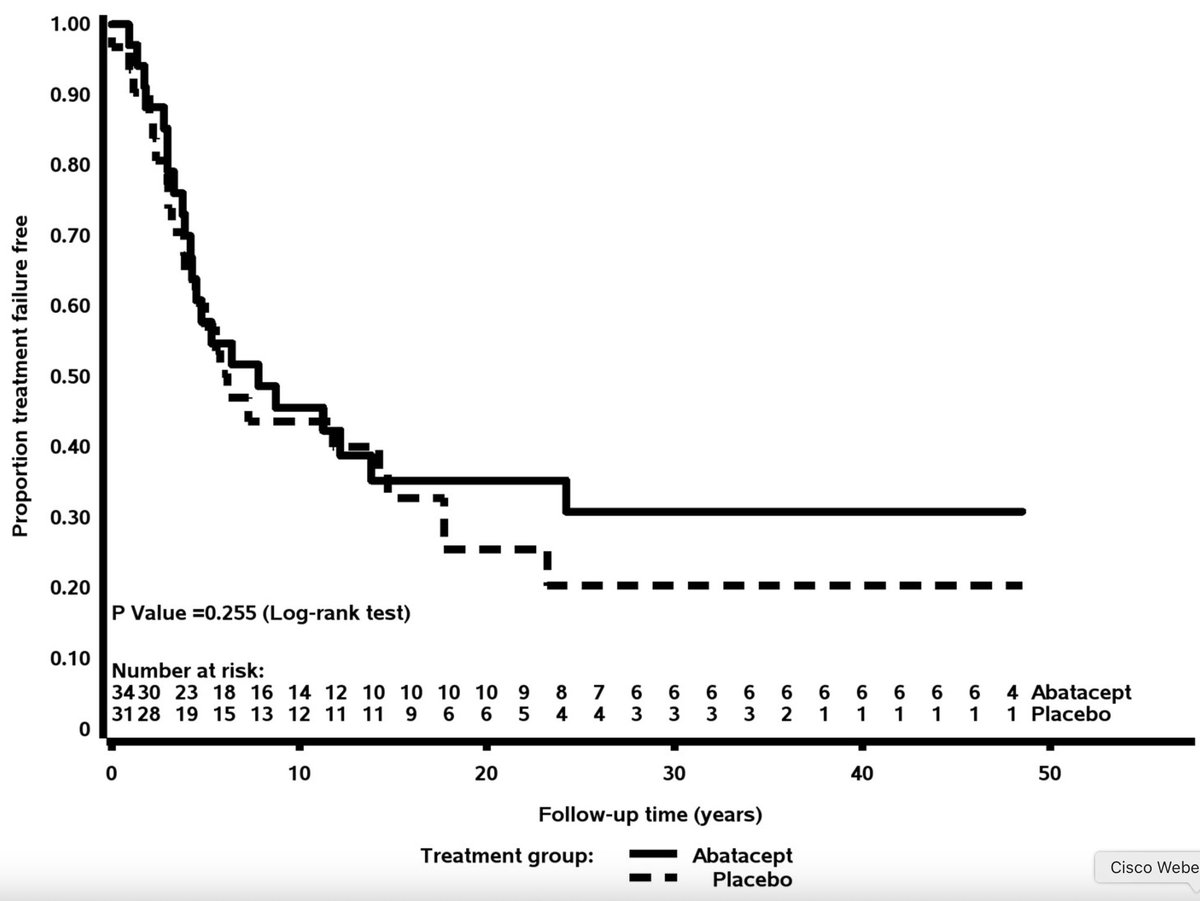
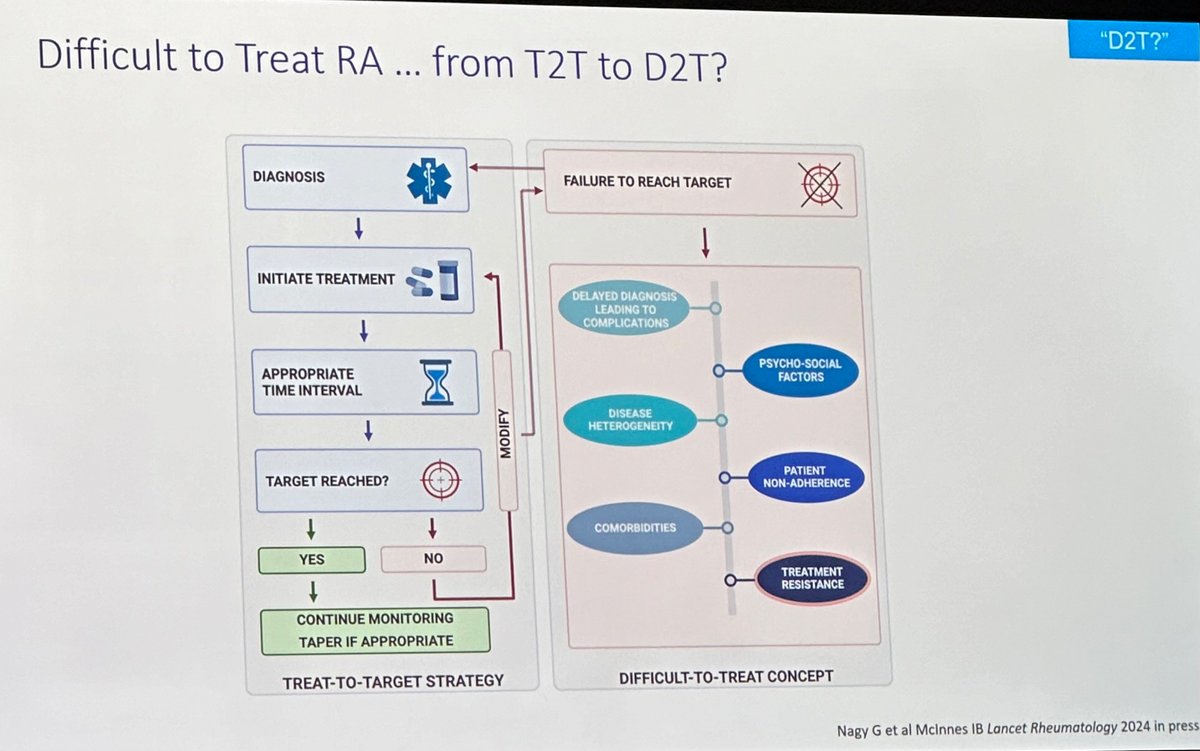
#ACR24 Year in Review: 💡GLP1i significantly decreased knee OA pain over time (or was it the wt loss itself?🤨) 💡CD19 CAR T-Cell therapy has potential to treat/cure many autoimmune diseases (are risks and $ worth the benefit?) 💡 High risk of CV events within 30 days of gout… https://t.co/XF63zr6lk4 https://t.co/oRNri66Il9
Links:

Links:
Links:
Links:
Links: