All News
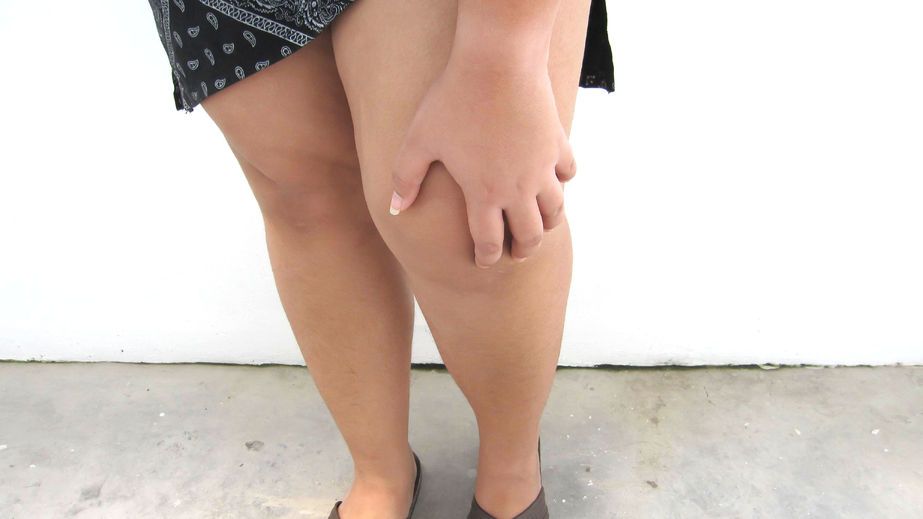
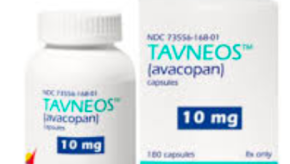
FDA Targeting the Safety of Avacopan
The future of avacopan is up in the air. In April, the FDA issued a drug safety warning, previously requesting that Amgen to pull its rare disease drug Tavneos from the market. Now, there appears more pressure to remove it from the market.
Read ArticleEULAR Recommendations for Physical Activity in Arthritis
EULAR has published a 2025 update for physical activity recommendations for patients with osteoarthritis (OA) and rheumatoid arthritis (RA).
Read ArticleNeoplasia and Autoimmune Disease
Malignancy rivals cardiovascular disease as a leading cause of death in patients with systemic autoimmune diseases. Chronic inflammation and immune dysregulation can drive oncogenesis, while antitumor immune responses can trigger autoimmune phenomena (paraneoplastic syndromes, checkpoint inhibitor-induced rheumatic disease).
Read ArticleSwitch or Cycle - Upadacitinib vs Adalimumab in Refractory RA
After the first tumour necrosis factor inhibitor (TNFi) failure, patients with active rheumatoid arthritis (RA) responded by switching to upadacitinib, compared to cycling to a second TNFi, adalimumab.









Dr. John Cush RheumNow ( View Tweet)


Links:
Dr. John Cush RheumNow ( View Tweet)