Real-world effectiveness of interleukin-6 receptor inhibitors compared to methotrexate in steroid- refractory frail patients with polymyalgia rheumatica
Sebastian E Sattui 1, Christian Dejaco 2,3, Kerri Ford 4*, Stefano Fiore 5, Sebastian H Unizony 6, Fenglong Xie 7,8, Jeffrey R Curtis 7,8
1 Division of Rheumatology and Clinical Immunology, University of Pittsburgh, Pittsburgh, PA, USA;
2 Division of Rheumatology and Clinical Immunology, Department of Internal Medicine, Medical
University, Graz, Austria;
3 Rheumatology, Hospital of Bruneck (ASAA-SABES), Teaching Hospital of the Paracelsius Medical University, Brunico, Italy;
4 Sanofi, Cambridge, MA, USA;
5 Sanofi, Morristown, NJ, USA;
6Massachusetts General Hospital, Harvard Medical School, Boston, MA, USA;
7 The University of Alabama at Birmingham, Birmingham, AL, USA;
8 Foundation for Advancing Science, Technology, Education and Research, Birmingham, AL, USA
*At the time of original presentation
Presenter: Matt Ackermann (matt.ackermann2@sanofi.com) from Sanofi presenting on behalf of all the
authors
Disclosures of authors:
Sattui SE: Research funding from Rheumatology Research Foundation Investigator Award, National
Institute of Aging (grant number R03AG082983), Consulting and advisory boards for Sanofi and Amgen
(all funds toward research support), Speaker Fees from Fresenius Kabi (all funds toward research
support), Research support Amgen and Glaxo Smith Kline (clinical trials);
Dejaco C: Consulting/speaker’s fees from AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, Sparrow, Roche, Boehringer, Galapagos, and Sanofi,
Editorial board member of the Annals of the Rheumatic Diseases;
Ford K: Former employee of Sanofi;
Fiore S and Ackermann M (presenter): Employees of Sanofi and may hold stock and/or stock options in
the company; Unizony SH: Consultant - Sanofi, IQVIA, and Harvard Pilgrim Health Care Inc.; Xie F: No
conflicts of interest;
Curtis JR: Consulting, research grants from AstraZeneca, Amgen, AbbVie, Bendcare,
Genentech, GSK, Horizon, Janssen, Lilly, Novartis, Pfizer, Sanofi, Scipher Medicine, Setpoint, and UCB.
Background:
A previous post-hoc analysis of frail patients with PMR on second-line (2L)/3L-treatment
found IL-6 receptor inhibitors (IL-6Ri) initiators were more than twice (HR 2.32) as likely to discontinue
GC vs. conventional synthetic immunomodulators (csIM initiators (HR 1.28 overall cohort). Here, we
compare IL-6Ri to methotrexate (MTX) as 2L-treatment, with frailty as inclusion criteria and propensity
score (PS)-match covariate, minimizing pre-frailty patient inclusion.
Methods: This observational retrospective comparative cohort study compared effectiveness of IL-6Ri to
MTX using US Medicare claims data (10/2016–12/2022). Inclusion criteria: ≥50 years with ≥1
inpatient/≥2 outpatient PMR diagnosis codes ≥30 days apart, ≤25 mg prednisone equivalent dose (PED),
claims-based frailty index (CFI) ≥0.25, IL-6Ri/MTX initiation, no prior IL-6Ri/csIM, continuous enrollment
≥180 days prior (baseline). Exclusion criteria: same day IL-6Ri/csIM initiation, seropositive RA/other
rheumatic disease during baseline. Patients were direct matched on select characteristics, then PS-
matched on demographics and clinical characteristics. Primary outcome: time-to-GC discontinuation
(>60-day gap); key secondary: time-to-minimal (≤2 mg/day PED) or no GC.
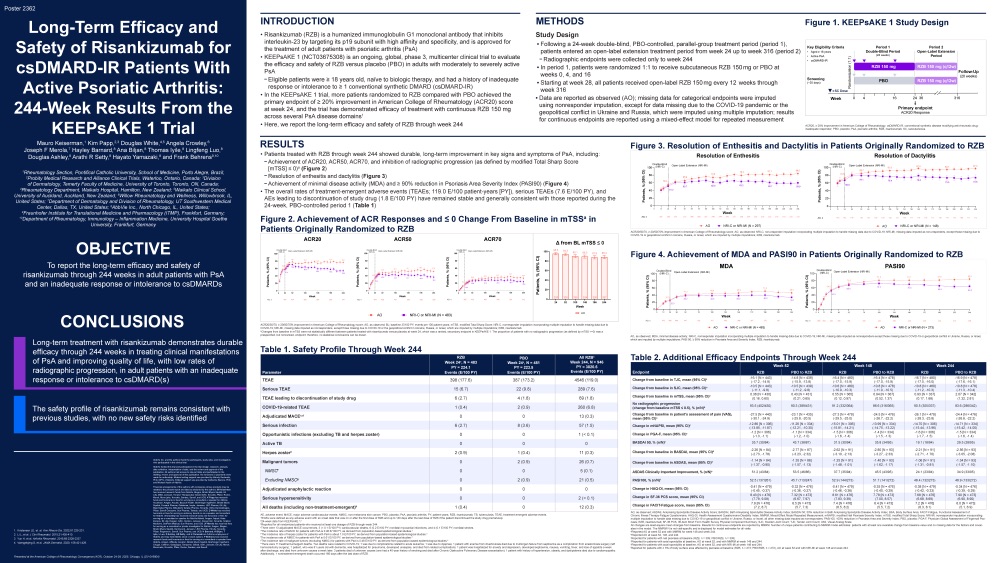
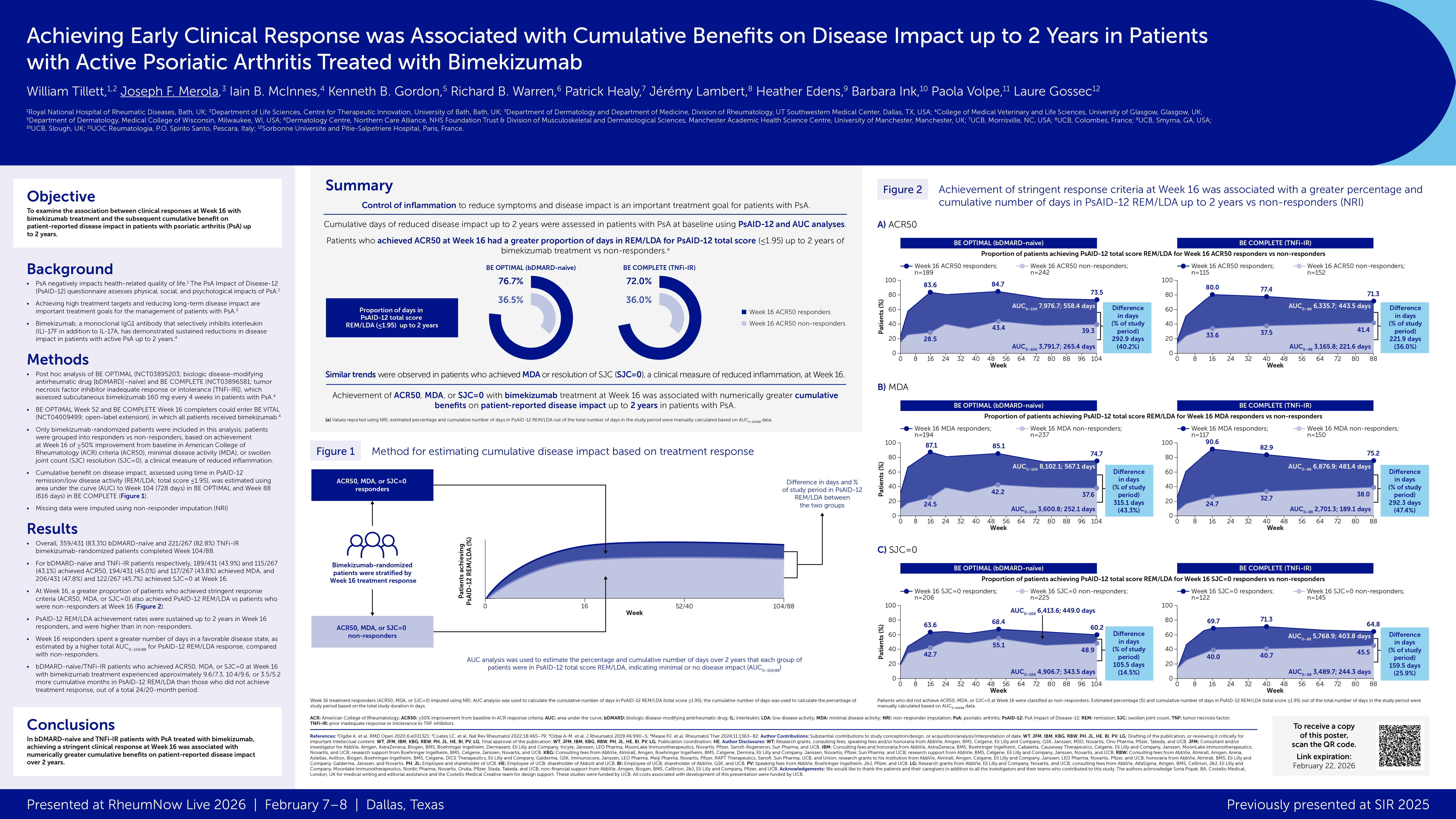
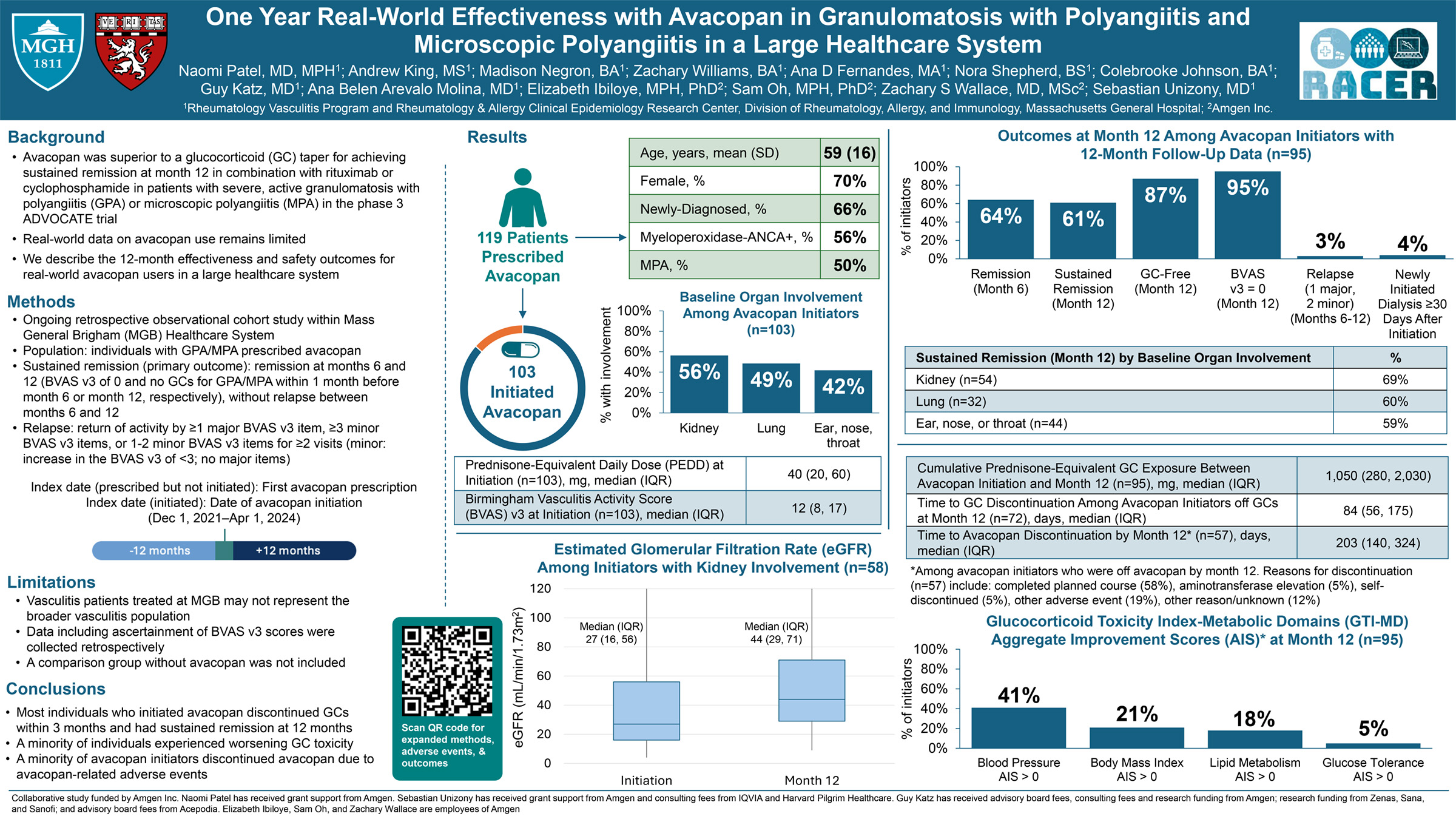
Results: Overall, 290/1,017 (29%) and 1,402/7,645 (18.3%) patients in the IL-6Ri and MTX groups,
respectively, had a CFI ≥0.25; eight-six matched pairs were identified. About 1/4 th were moderately-
severely frail (CFI ≥0.35, 20.9% vs. 26.7%). Time-to-GC discontinuation and time-to-minimal or no GC up
to 1 year were significantly shorter for IL-6Ri vs. MTX groups (HR [95% CI] 1.75 [1.05, 2.93]; p=0.033) and
(HR 1.86 [1.15, 3.00]; p=0.011), respectively (Figure 1).
Conclusions: IL-6Ri enabled more rapid GC discontinuation/minimal GC achievement compared to MTX
in frail patients with PMR, suggesting greater benefit vs. MTX in this vulnerable population.
Funding:
This analysis was sponsored by the Foundation for Advancing Science, Technology, Education and
Research (FASTER) and was funded by Sanofi and Regeneron Pharmaceuticals, Inc.
Acknowledgements:
These data were previously submitted to the European Alliance of Associations for Rheumatology
(EULAR) 2025, Barcelona, Spain (June 11–14, 2025). Medical writing support for the original abstract was
provided by Kritika Dhamija, M. S. (Pharm.), of Sanofi, and editorial support for this encore abstract was
provided by Himani Powle, PharmD, of Sanofi.