All News
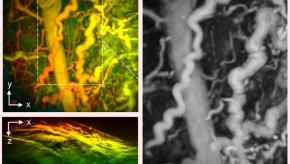
Hand-Held Imaging Could Transform Arthritis Care
A new hand-held scanner developed by UCL researchers can generate highly detailed 3D photoacoustic images in just seconds, paving the way for their use in a clinical setting for the first time and offering the potential for earlier disease diagnosis.
Read ArticleASAS Imaging Recommendations for Suspected Spondyloarthritis
ASAS (Assessment of Spondyloarthritis International Society) has convened specialized task force to guide imaging request on patients with suspected/known axial spondyloarthritis (axSpA).
Read ArticleDeclining Menopausal Hormone Therapy in Postmenopausal Women
A two decade cross-sectional population study has shown a marked downward trend in menopausal hormone therapy (MHT), with an over 80% reduction, especially in women aged 52 - 65 years.
Read ArticleStratified Cancer Screening in Dermatomyositis
The International Myositis Assessment and Clinical Studies Group (IMACS) have published evidence and consensus based cancer screening guidelines for patients with idiopathic inflammatory myopathies, including dermatomyositis.
Read ArticleBimekizumab FDA Approved for Psoriatic Arthritis, Non-radiographic Axial Spondyloarthritis and Ankylosing Spondylitis
UCB announced today that the U.S. Food and Drug Administration (FDA) has approved bimekizumab-bkzx (Bimzelx) for the treatment of 3 chronic inflammatory disorders.
Read ArticleI Can't Treat Ugly (9.20.2024)
Dr. Jack Cush reviews the news, journal articles and a new FDA approval for EGPA, this week on the Podcast.
Read ArticleTREAT EARLIER - Methotrexate in Clinically Suspect Arthralgia Patients
Lancet Rheumatology has published the results of the TREAT EARLIER trial, showing the prevention of rheumatoid arthritis may be enhanced by risk stratification, particularly in anti-citrullinated protein antibody (ACPA)-negative people with clinically suspect arthralgia.
Read ArticleIncreased Cardiovascular Events in Lupus
A New York surveillance registry (Manhattan Lupus Surveillance Program) estimates higher rates of cardiovascular events (CVE) among systemic lupus erythematosus (SLE) patients, especially amongst younger males, Hispanic/Latinos and non-Hispanic Black patients.
Read ArticleBSR Guidelines for Systemic Sclerosis Management
The British Society for Rheumatology (BSR) has updated its 2015 guidelines for the management of patients with sytemic sclerosis (SSc) based on published evidence, systematic literature review and expert opinion.
Read ArticleIncreasing Incidence of Morphea
A large EMR derived cohort study of over 10,000 patients with morphea (localized scleroderma) sheds light on its prevalence and associated features.
Read ArticleACR Speaks Against Repeated Cuts to Medicare Reimbursements
The American College of Rheumatology has submitted its comments to the CMS regarding the proposed CY 2025 Medicare Physician Fee Schedule and Quality Payment Program rule and its impact on rheumatologists and rheumatology interprofessional team members’ ability to provide care to the 53 million Americans living with rheumatic disease.
Read ArticleACR Recommends QOL Self-Management Strategies
American College of Rheumatology (ACR) experts identified research suggesting that patient self-management is not a
Read ArticleConsensus Guidelines on Pediatric Methotrexate Use
Methotrexate (MTX) is commonly used in the treatment of pediatric inflammatory skin conditions, often for off-label indications. Consensus based recommendations were published to address 5 major subjects.
Read ArticleMoving Forward on RA Prevention Trials
Now that multiple prevention clinical trials for rheumatoid arthritis (RA) have been completed, a group of investigators have reviewed the results and published their recommendations for future trial designs and drug development to assess interventions that may alter disease development.
Read ArticleRunning with Data (8.30.2024)
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com - including tips on steroids, MAS, and myositis testing.
Read ArticleACR Backs FDA Rule Change on Interchangeable Biosimilars
The American College of Rheumatology submitted comments to the U.S. Food & Drug Administration supporting their recent proposal to update the approval process for biosimilars’ interchangeability status by repealing the requirement for switching studies while also encouraging the FDA to continue monitoring the downstream effects of this policy to ensure transparency for patients and prescribing providers.
Read Article

Links: