All News
Calculating Serious Infection Risk in IMID Patients
Predictive modeling of data derived from the DANBIO registry (of treated IMID patients) revealed a 4 fold increased risk of serious infection (SIE) in those starting biologic DMARD (bDMARD) treatment. From this large dataset, researchers developed a simple prediction model to estimate future infection risk that may inform shared decision-making in individual patients.
Read ArticleHigh Placebo Responses in SLE Trials
A systematic review by Urowitz and colleagues examined placebo responses in randomized controlled trials of non-renal, non-neuropsychiatric SLE patients showing unexpectedly high placebo responses; with more than one-third of the placebo-treated SLE patients responding by validated outcome measures.
Read ArticleJAK Inhibitor Misses Endpoint in Safety Study. Now What?
Pfizer announced results Wednesday from its FDA-mandated postmarketing safety study of tofacitinib (Xeljanz), and they don't bode well for the drug and possibly others in its class.
Read ArticleUrate Lowering Therapy During Acute Gout
Acute gout has its well defined protocols, and most state that urate lowering therapy (ULT) should be continued; but does ULT affect outcomes in an acute gout attack?
Read ArticleProtective Effects of Colchicine in Non-Hospitalized COVID-19
A press release from Montreal Heart Institute announced that the COLCORONA clinical trial provided evidence of the protective effect of colchicine; demonstrating a 21% reduced risk of death or hospitalizations in patients with COVID-19 compared to placebo. COLCORONA trial was a contact-less, randomized, double-blind, placebo-controlled study of 4,488 non-hospitalized patients from Canada, the U.S., Europe, South America and South Africa.
Read ArticleVoclosporin FDA Approved for Lupus Nephritis
On January 22nd, the FDA approved voclosporin (Lupkynis) for use in adults with active lupus nephritis; voclosporin is a calcineurin-inhibitor immunosuppressant indicated for use in combination with a background immunosuppressive therapy. FDA approval was based on data from the pivotal AURORA Phase 3 study and the AURA-LV Phase 2 study, which demonstrated the ability of voclosporin to significantly improve LN outcomes after 52 weeks of followup in SLE nephritis patients receiving background standard of care therapies like MMF, and low dose steroids.
Read ArticleRheumNow Podcast – Don’t Hold the Benlysta, Take the Vaccine (1.22.2021)
Dr. Jack Cush reviews the news and journal articles from the past week on RheumNow.com, with more than a dozen items to discuss on this week's podcast.
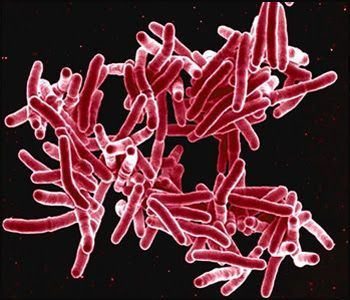
Read ArticleLow Risk of TB with Secukinumab
JAMA Dermatology has reported on a longitudinal cohort study of 12,319 secukinumab (SEC) treated patients (psoriasis, psoriatic arthritis, or ankylosing spondylitis); no new cases of active tuberculosis (TB) and very few cases of latent tuberculosis infection (LTBI) were f
Read ArticleMortality Risk in Rheumatoid Lung Patients
A Medicare claims analysis of rheumatoid arthritis (RA) patients showed that RA-associated interstitial lung disease (RA-ILD) was seen in nearly 5% of patients and was found to increase mortality risks, including respiratory and cancer mortality not explained by other factors.
Read ArticleFactors Predictive of SLE Flare After HCQ Taper/Discontinuation
The benefits of hydroxychloroquine in lupus are many, but if you were to withdraw HCQ? A recent study of five Canadian cohorts, suggests that certain baseline variables, including age, race, and steroid use, were predictive of flare in patients with SLE who tapered or discontinued treatment with hydroxychloroquine.
Read Article
Medicine Matters rheumatology Rheum_Matters ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:

Links:
Dr. John Cush RheumNow ( View Tweet)
Links:

Links:
Dr. John Cush RheumNow ( View Tweet)

k dao KDAO2011 ( View Tweet)

Links:

Links: