All News
EMA Crack Down on JAK Inhibitors
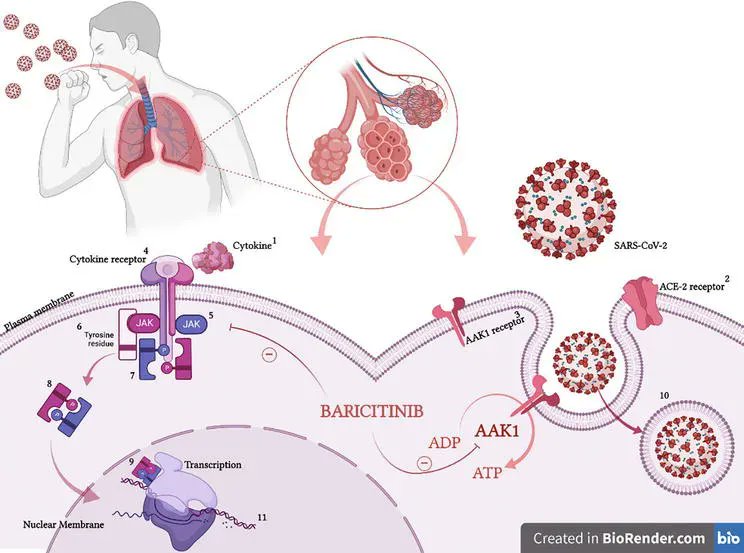
The European Medicines Agency’s (EMA's) safety committee (PRAC) has issued a recommendation that the use of oral JAK inhibitors (tofacitinib, baricitinib, upadacitinib and filgotinib) be limited to certain at-risk patients unless no other suitable treatment alternatives are available.
Read ArticleInfections & Lupus (10.28.2022)
Dr. Jack Cush discusses the news and journal reports from the past week on RheumNow. This week more lupus nephritis, more drug induced lupus and more lupus infections that may kill.
Read ArticleUpdate to Axial Spondyloarthritis Guidance
EULAR and the Assessment of SpondyloArthritis International Society (ASAS) has updated their guidance on axial spondyloarthritis (axSpA) management.
Read ArticleFDA Approves Upadacitinib for Non-Radiographic Axial Spondyloarthritis
On Friday October 21st the U.S. Food and Drug Administration approved upadacitinib (Rinvoq) for the treatment of active non-radiographic axial spondyloarthritis in patients with objective signs of inflammation who have had an inadequate response or intolerance to tumor necrosis factor blocker therapy.
Read ArticleACR Award Winners (10.21.2022)
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com. Highlighted discussion of implantable stimulators, urine proteomics and gut-immune responses to Prevotella in RA.
Read ArticleNo Special Heart Risk With JAK Inhibitors, Study Finds
Patients taking Janus kinase (JAK) inhibitors for rheumatoid arthritis experienced similar cardiovascular outcomes as those using the tumor necrosis factor (TNF) blocker adalimumab (Humira), analysis of French national registry data indicated.
Read Article


Links:

Links:


Links:
Links:

Links:

Links:
Links:
Links:
Links:

Links: