All News
Equal Safety of JAK Inhibitors and TNF Inhibitors
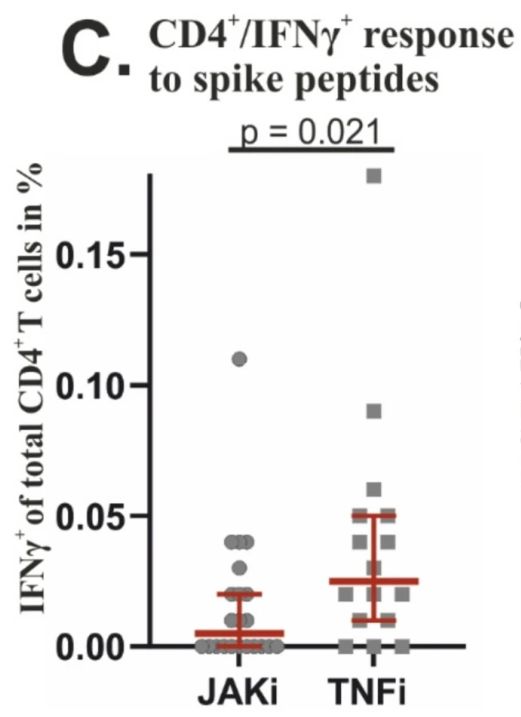
JAMA has published a systematic review and meta-analysis of head-to-head studies showing there was no meaningful difference in safety events observed when taking either JAK inhibitor (JAKi) vs TNF antagonist (TNFi) therapies for the treatment of immune-mediated
ILD Begins (8.29.2025)
Dr. Jack Cush reviews the news and journal reports from RheumNow.com. This week news on vaccines, safety of acetaminophen and more.
Read ArticleUncertainty with immunosuppressive for idiopathic inflammatory myopathies
A current Cochrane review suggests a continued unmet need regarding the status and utility of targeted immunosuppressant and immunomodulatory treatments for the idiopathic inflammatory myopathies (IIM). Overall there has been little progress since the previous Cochrane review (2012) that found little or no evidence to guide treatment.
Read ArticleTNF Inhibitors in Takayasu's Arteritis
A large, retrospective, multicenter study demonstrated the efficacy of intravenous infliximab and subcutaneous adalimumab in patients with Takayasu arteritis.
Read ArticleRheums Speak: Changing Rheumatology Practices & Choices
RheumNow surveyed clinicians worldwide on how their rheumatology practice, treatment decisions, and career outlooks have changed over the past 20 years. 242 respondents (68% from the USA) shared their perspectives on the changing landscape of rheumatoid arthritis care, practice patterns, and professional development.
Read ArticleRepurposing & Pipeline (8.8.2025)
Dr. Jack Cush reviews the news, reports, pipeline and drug repurposing on this week’s podcast.
Read Article
Links: