All News
ACR Best Abstracts - Day 1
The RheumNow faculty reporters have been scouring the meeting and online presentations to find the best abstracts from ACR22. Here are some of their choice abstracts reported today on day 1 of ACR 2022 (#ACRbest).
Read Article
Plenary:
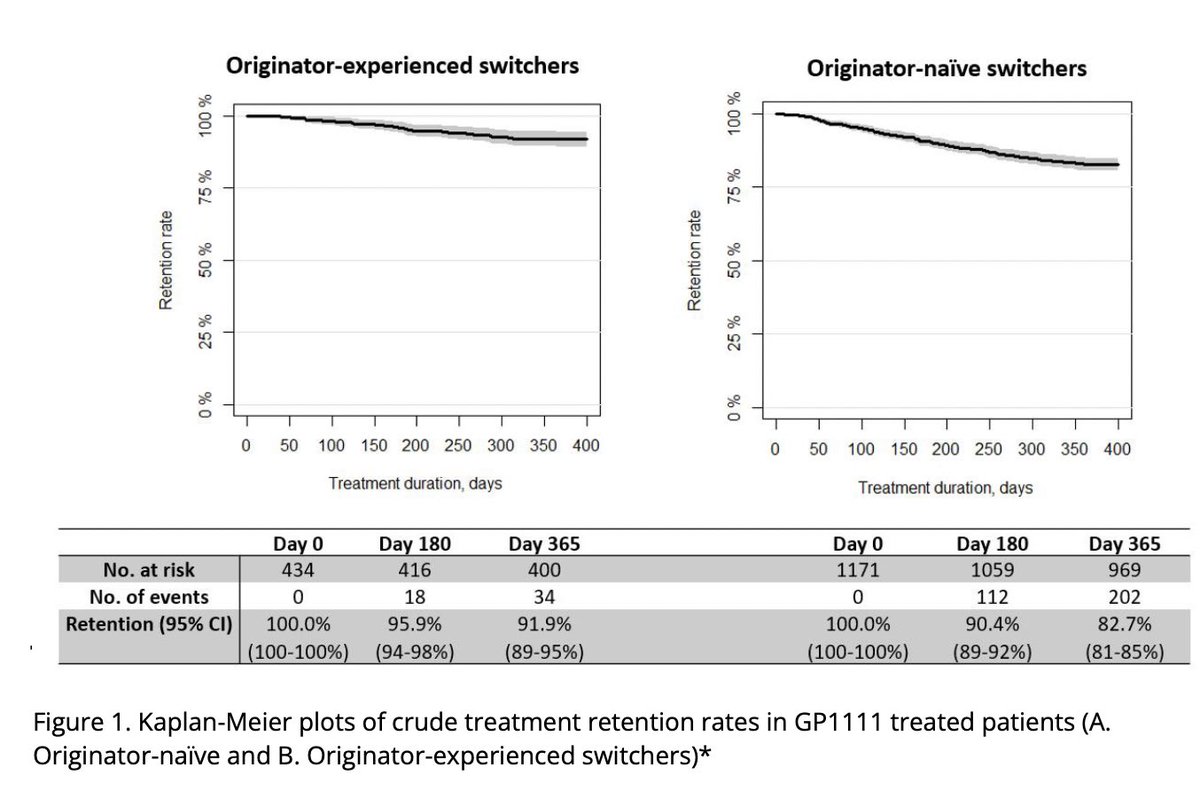
Real-world observational study >1500 pts with IA, 1 year retention infliximab biosimilar CT-P13 to GP1111 switch was high. Retention was higher in originator-experienced (92vs83%) & low disease activity
Nabi H, DANBIO Abs1112 https://t.co/z84AMUS3GK #ACR22 @RheumNow https://t.co/SgGhd33Esl
Dr. Antoni Chan synovialjoints ( View Tweet)
Plenary:
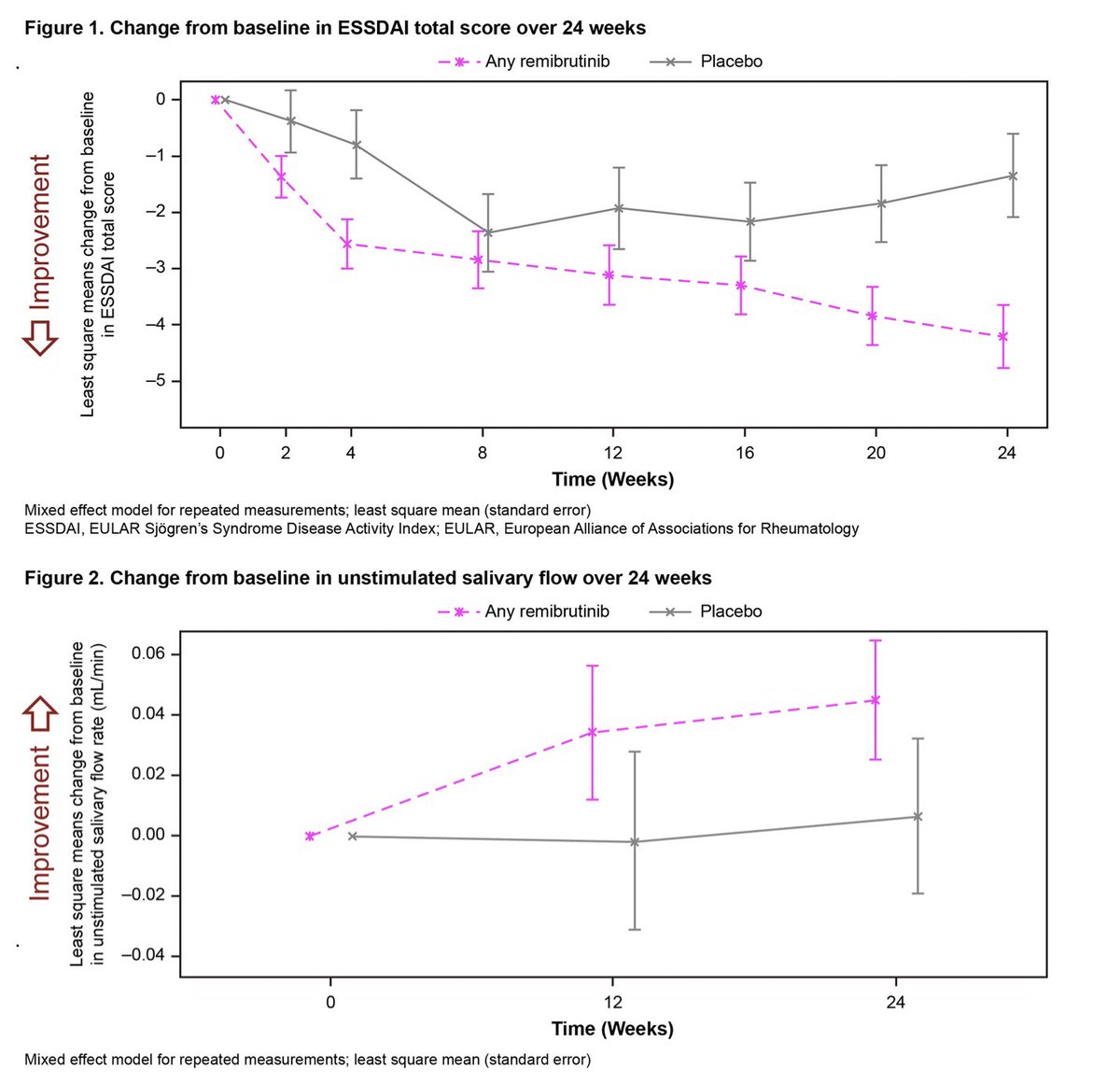
Remibrutinib (LOU064) a BTK inhibitor in Sjogren’s
◦ Favourable safety profile
◦ Well-tolerated over 24 Wks
◦ Improved ESSDAI, salivary flow, Igs
◦ May be first effective oral disease-modifying therapy for SS
Dorner T Abs1113 https://t.co/FkgJpwKAJL #ACR22 @RheumNow https://t.co/znxMc27EBI
Dr. Antoni Chan synovialjoints ( View Tweet)
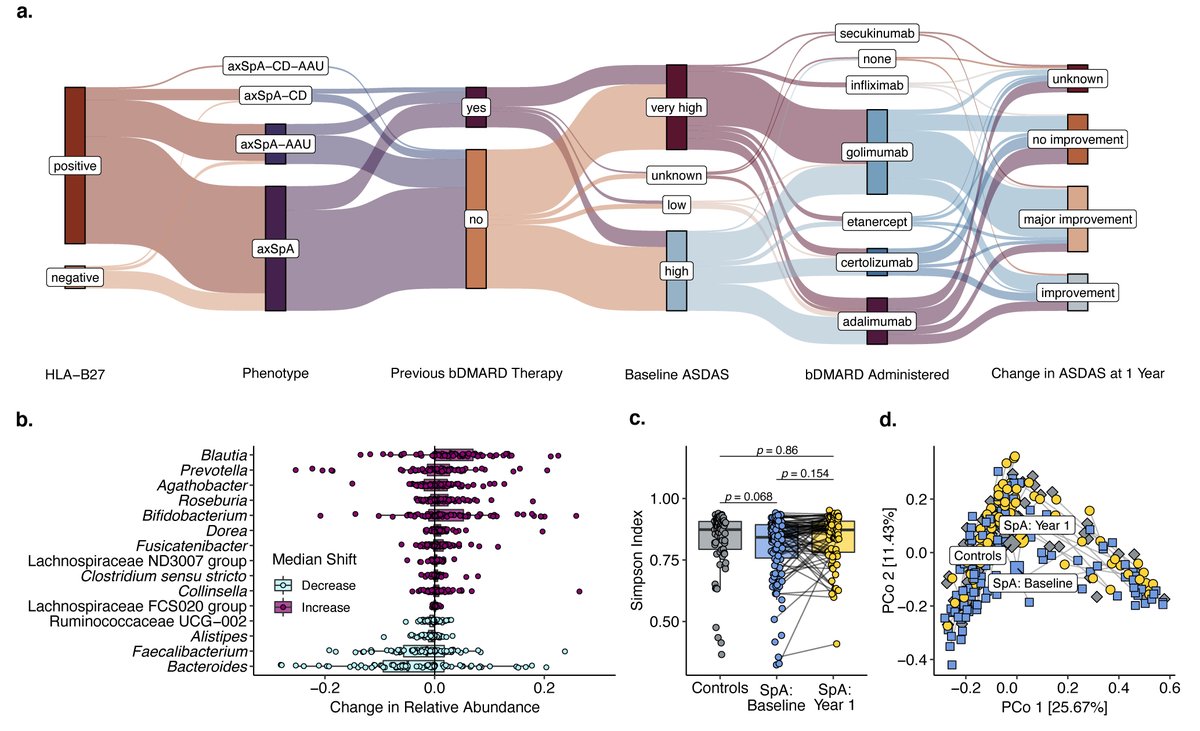
Biologics to treat gut microbiome dysbiosis??
In axSpA pts treated with bDMARDs for one year, gut microbiome resembled those of controls.
Collinsella remained stable - possible biomarker?
Abs#1162 @RheumNow #ACR22
#ACRBest https://t.co/vmQFJMpNPP
Robert B Chao, MD doctorRBC ( View Tweet)
Singh @NamrataRheum et al. 27,421 RA patients, 8225 on b/tsDMARD. b/tsDMARDs did not increase risk of lymphoma. IR 1.68/1000PY (95% CI 1.5-1.9) on csDMARD vs IR 1.73/1000PY (95%CI 1.4-2.1) on b/tsDMARD. @RheumNow #ACR22 Abstr#1210 https://t.co/3Uvz3mvsGM https://t.co/6k3Vqb21KF
Richard Conway RichardPAConway ( View Tweet)
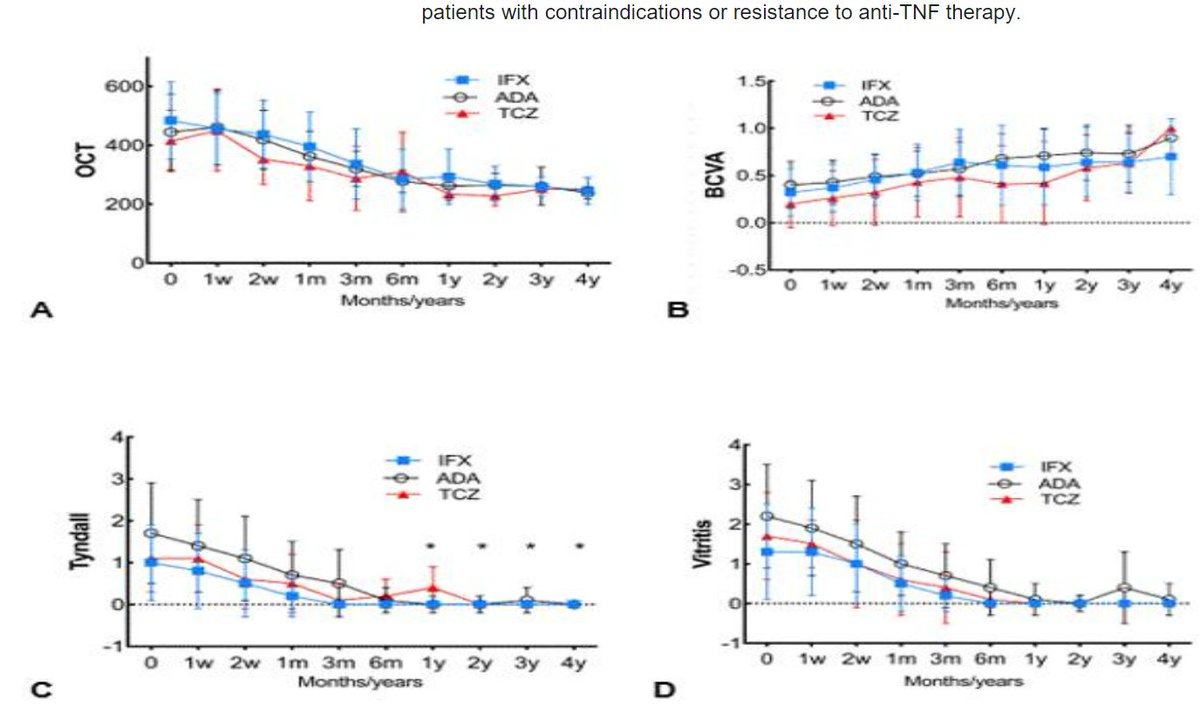
Barroso-García et al. ADA vs IFX vs TCZ in severe ocular Behcets with refractory cystoid macular edema. Remission 76% (ADA), 60% (IFX), 67% (TCZ). @RheumNow #ACR22 Abstr#1564 https://t.co/pRVgIdGAGS https://t.co/gOb5wQ9pHv
Richard Conway RichardPAConway ( View Tweet)
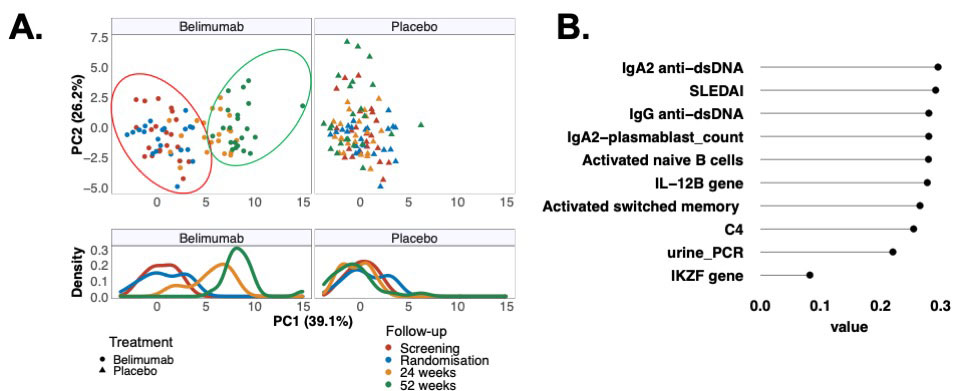
BEL after RTX targets IgA2 anti-dsDNA abs and favors formation of anergic/resting B cells while reducing potentially pathogenic B-cell subsets. Abs 0978 #ACR22 @RheumNow https://t.co/Y798OAwtbt https://t.co/IfXOv67MLV
Dr. Rachel Tate uptoTate ( View Tweet)
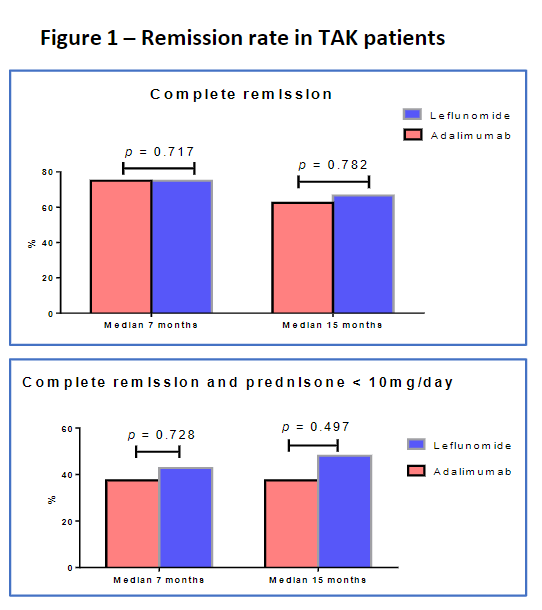
Filho et al. Retrospective Brazilian study. ADA vs LEF in Takayasu arteritis. Comparable renission, relapses, steroid dose, ESR, CRP, imaging over median 15 weeks. More AEs in LEF but non-severe. @RheumNow #ACR22 Abstr#1553 https://t.co/499dUNmpUD https://t.co/AEk5cG7dc4
Richard Conway RichardPAConway ( View Tweet)
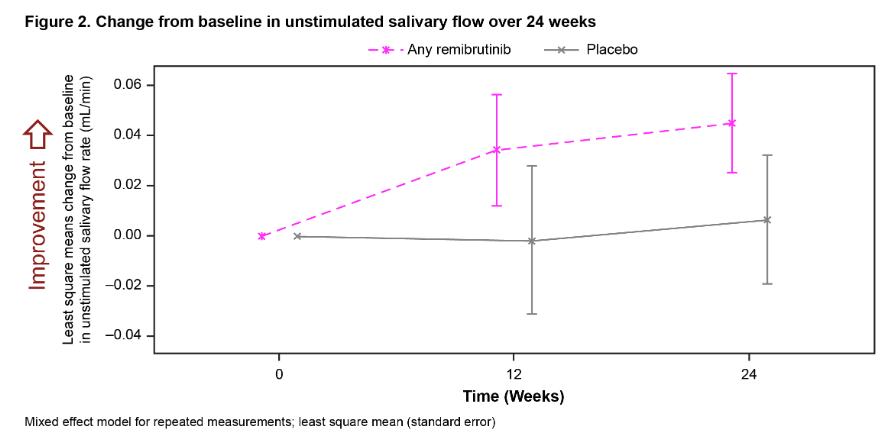
Ab1113 #ACR22 Remibrutinib (LOU064, BTK inhib) in Sjogren's
Will there be new Rx for Sjo?
T Dorner: 73 pts Ph2 BTK 100mg BID/100 QD/PBO
Better ESSDAI at wk 24 (p=0.003)
Unstim saliv flow: improved vs PBO, not stat signif
Decreased CXCL13 chemokine
PROs not improved
@RheumNow https://t.co/hJp33JPpHt
Eric Dein ericdeinmd ( View Tweet)
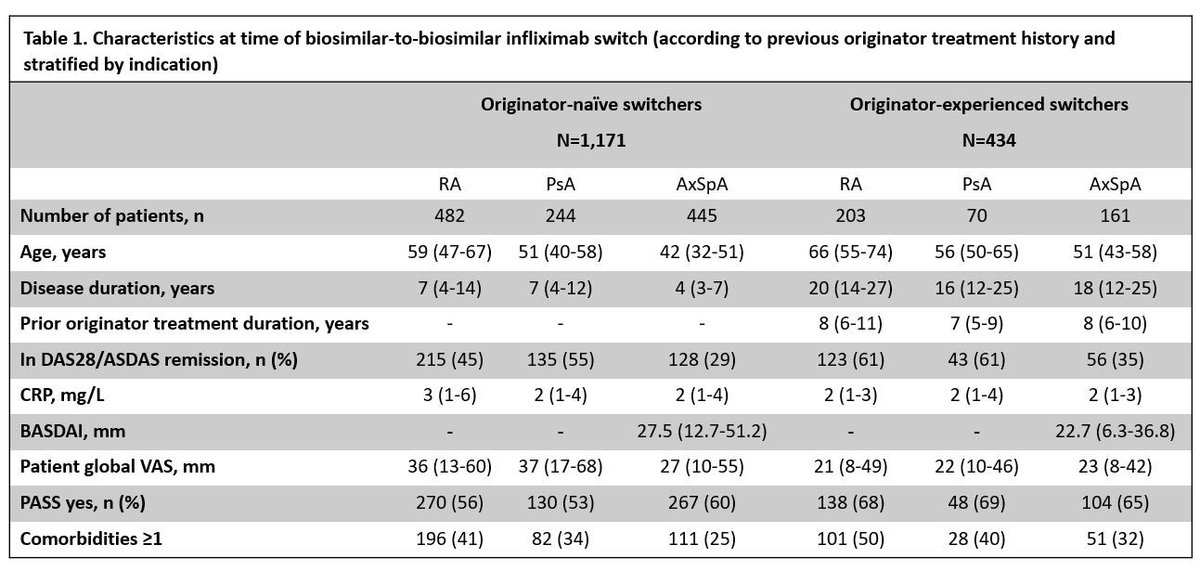
DANBIO registry plenary Abs 1112 finds 1 yr retention following a mandatory nationwide infliximab biosimilar-to-biosimilar switch was high. Retention higher in originator-experienced and in pts w/ low disease activity. #ACR22 @RheumNow https://t.co/ZAV2dcVzXl https://t.co/Kti2bZ1pkZ
Dr. Rachel Tate uptoTate ( View Tweet)
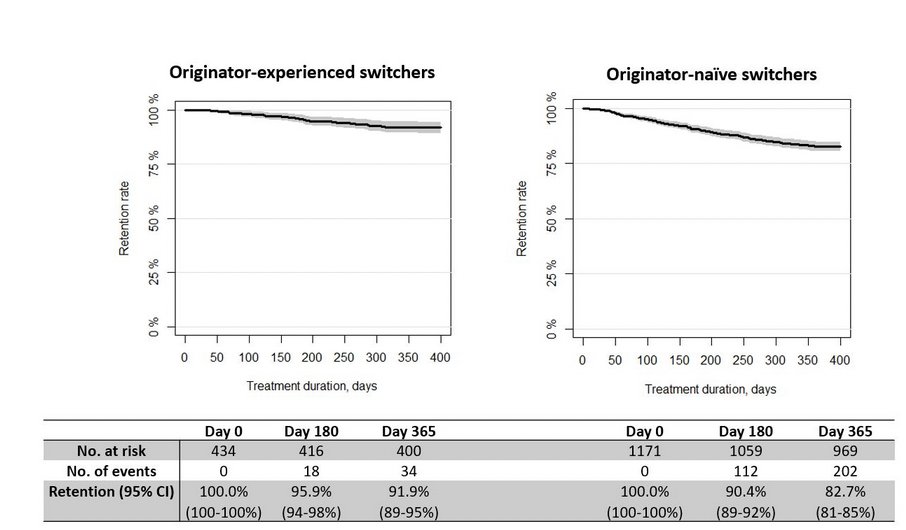
DANBIO registry: 1600+ pts RA, AxSpA, PsA
Infiximab biosimilar to biosimilar switch (CT-P13 -> GP1111)
Maintenance at 1yr:
>80%
> withdrawal in original IFX naive, but higher BL DA
Do second time switchers do better because of past switch experience?
Abs#1112 #ACR22 @RheumNow https://t.co/IgjhGLVESz
Aurelie Najm AurelieRheumo ( View Tweet)
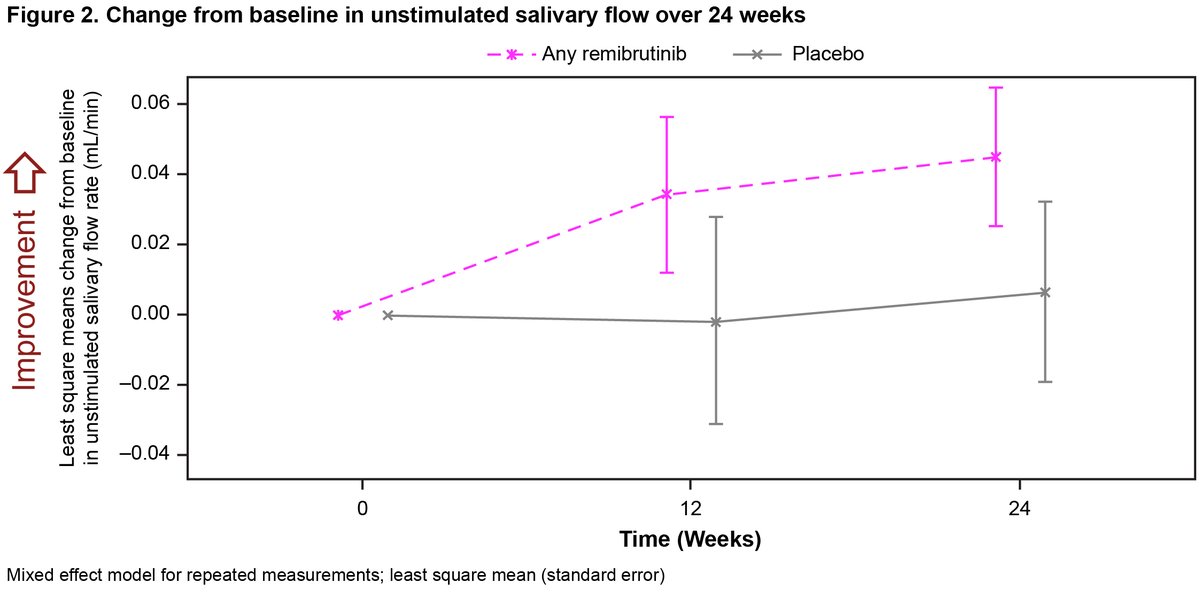
2nd Plenary Session!
Remibrutinib (BTK inhibitor) for treatment of Sjögren's syndrome (phase 2 study)
Improvement of ESSDAI, salivary flow and pathologically elevated Ig's
Abs#1113 @RheumNow #ACR22 https://t.co/vdgxBPe6qd
Robert B Chao, MD doctorRBC ( View Tweet)
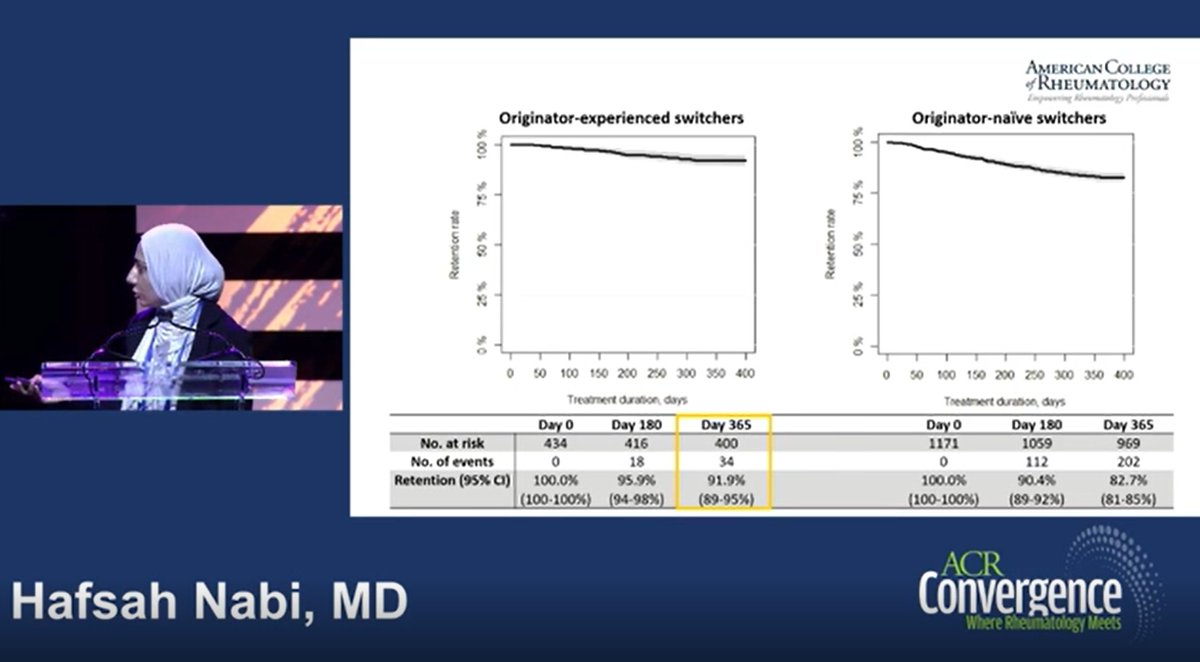
We're used to originator-to-biosimilar switches.
Widespread biosim use means we now see biosim-biosim switches (incl as second switch)
DANBIO data
retention good
I see the hallmarks of nocebo still an issue
I think we can all agree: ongoing education important
#ACR22 @RheumNow https://t.co/6PcMfEEozk
David Liew drdavidliew ( View Tweet)
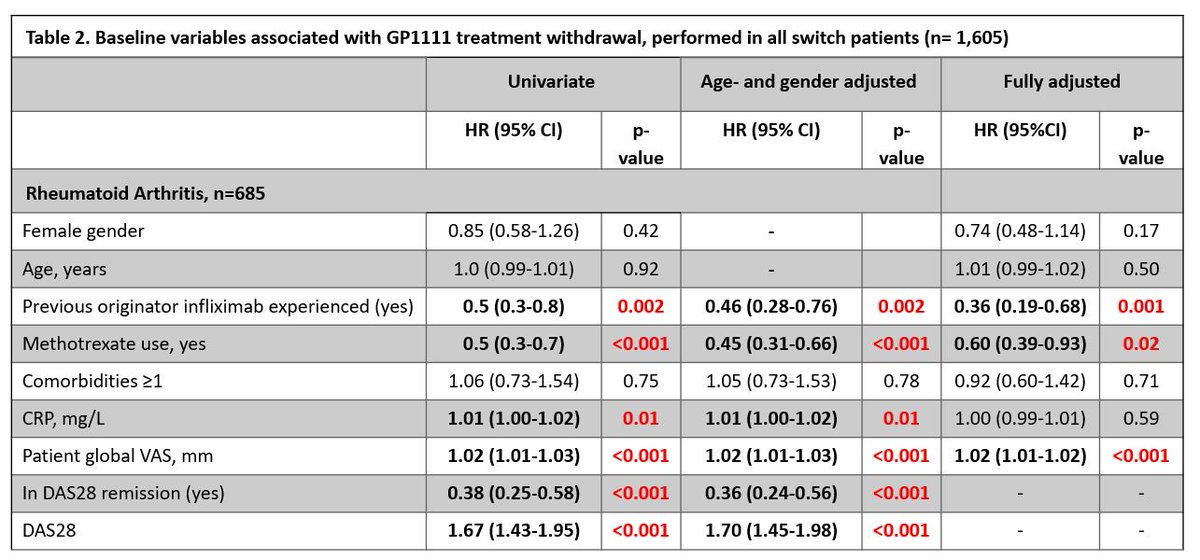
Biosimilar to biosimilar effective in treatment of RA/PsA/AxSpA.
Large study looking at infliximab biosimilar switching - resulted in high retention rates and virtually no change in disease activity.
@RheumNow #ACR22 Abs#1112 https://t.co/uNRXLewo4M
Robert B Chao, MD doctorRBC ( View Tweet)

Valero et al. Combined biologics in PsA. @RheumNow #ACR22 Abstr1044 https://t.co/7nNf0Ktf0M https://t.co/CIE9BnBFvx
Richard Conway RichardPAConway ( View Tweet)
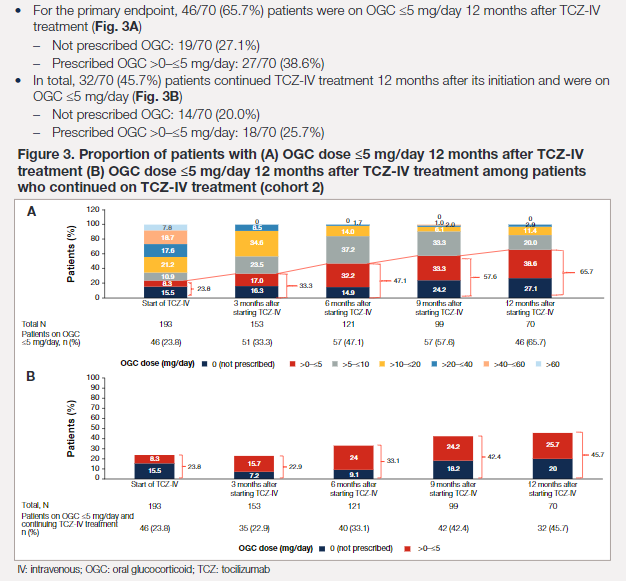
#ACR22 Abstr#0826 2 years on since FDA approved #Tocilizumab in Adults Onset Still's Disease, real-world data from Japan affirmed its effectiveness and as a steroid-sparring agent @RheumNow https://t.co/jbmhEnGwXO
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)
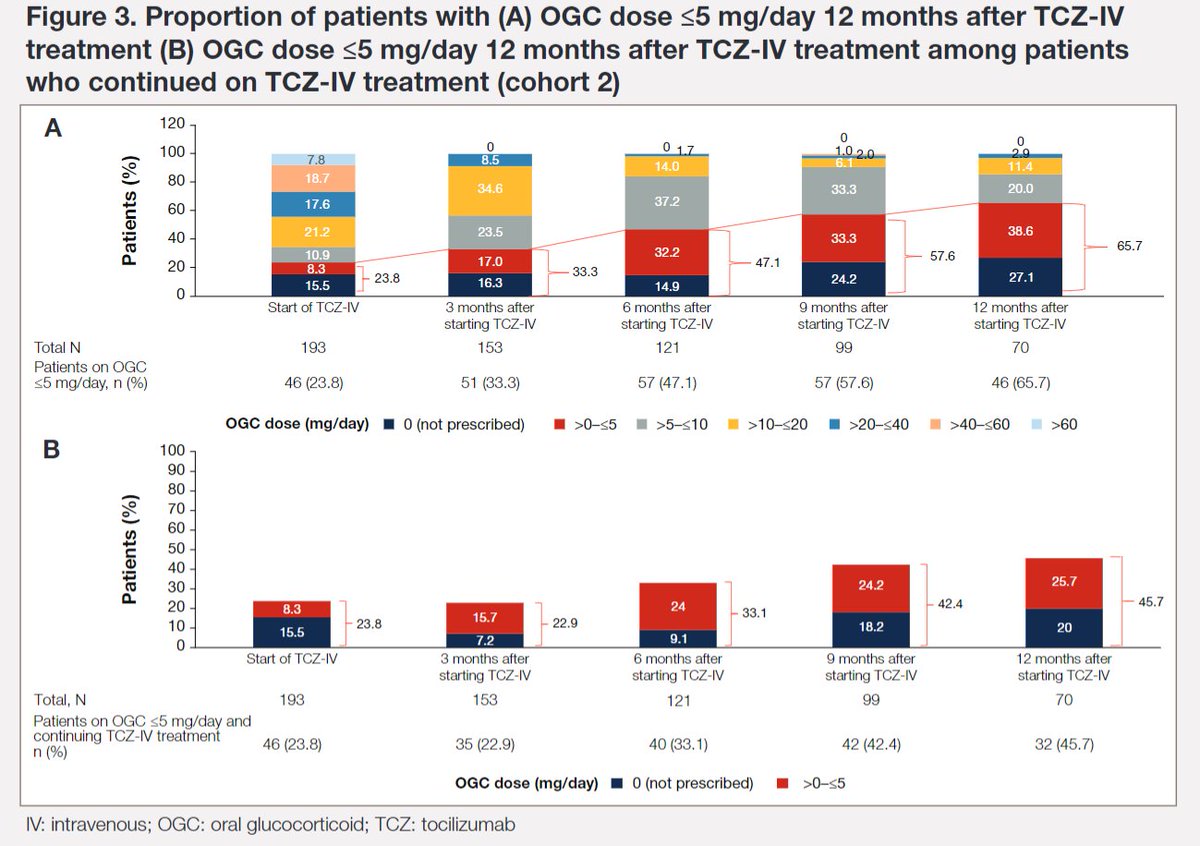
Kaneko et al. Tocilizumab leads to significantly lower steroid doses in Adult onset Stills Disease @RheumNow #ACR22 Abstr#0826 https://t.co/jc1S6sdfkm https://t.co/z0cHB4wYu6
Richard Conway RichardPAConway ( View Tweet)
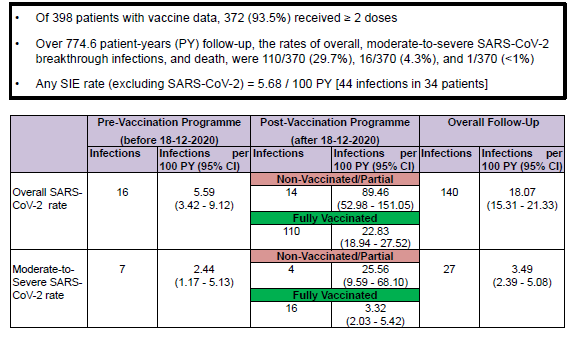
#ACR22 Abstr#0783. Rituximab impacts on humoral immunity. Our study on 400 RMD pts showed:
Breakthrough infection 30% mostly mild; Severe 4%; 1 death
Poor outcomes predicted by comorbidities & low IgG
Risk reduced by 50% with each vac dose
OK for RTX if vaccinated @RheumNow https://t.co/xyJsukm1Rh
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)
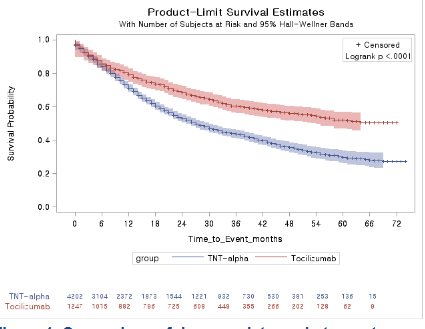
Song et al. Korean study shows better persistence of tocilizumab than TNFi as first-line biologic in RA. No difference in incidence of TB. @RheumNow #ACR22 Abstr#0887 https://t.co/EuQkfDZd77 https://t.co/VxJmKEzAVY
Richard Conway RichardPAConway ( View Tweet)

Trend of reduced opioid use when initiating biologics and JAKi in RA though effect size small and very high baseline use (>50%) in this insurance claims database.
Residual pain and harm minimisation still areas of need.
@RheumNow #ACR22 ABST0925
https://t.co/XGHu0fIMF0 https://t.co/PutNNQiKnY
Julian Segan JulianSegan ( View Tweet)




