Pregnancy Outcomes Improve in Lupus
Changing Patterns of Drug Use in Pregnant Rheumatic Patients: Steroids Down, Biologics Up
Desai and coworkers analyzed public and private insurance claims data (2001-2012) to assess patterns of drug use in pregnant woment with RA, SLE, PsA and AS. Specifically, they looked at immunosuppressive drug use in the 3 months preceding and during pregnancy.
Increased Congenital Anomalies in Children from Women with a Juvenile Arthritis History
A Quebec administrative claims analysis examined the birth outcomes of women with a history of juvenile idiopathic arthritis (JIA) who subsequently had a first-time birth (between 1983-2010). JIA patients were matched 4:1 with normal controls from the same era and adjusted for age and residence.
The JIA women included 1681 women who had a mean age at delivery of 24.7 years compared with the non-JIA group who were 25.0 years a the time of delivery (n = 6724).
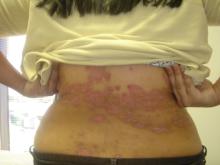
Pregnancies are Adversely Affected Even Before the Diagnosis of Lupus
Arkema and colleagues assessed maternal and fetal outcomes in patients with SLE, pre-clinical (prior to onset) SLE and prevalent maternal SLE during pregnancy compared with the general population. These cohorts were identified from a population-based Swedish registry that included 13,598 women with first pregnancies and compared 551 prevalent SLE, 65 pre-SLE (within 0-2 years), 133 pre-SLE (within 2-5 years), and 12,847 general population controls. (Citation source http://buff.ly/1TmInbe)
ACR 2020 Reproductive Guidelines - Contraception & Counseling
Best of 2020: ACR 2020 Reproductive Guidelines - Contraception & Counseling
Tocilizumab in Pregnancy
Tocilizumab is currently listed as a Category "C" pregnancy risk based on limited pre-clinical (animal) data that was negative. The product label states that "adequate and well-controlled studies with ACTEMRA have not been conducted in pregnant women....and that ACTEMRA should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus."
Pregnancy Outcomes in Psoriatic Arthritis
Adverse Maternal and Fetal Outcomes in Lupus - and Pre-Lupus Too
A population-based claim study from Sweden has shown that lupus and pre-lupus patients are at risk for adverse maternal and fetal outcomes.
Investigators used population-based Swedish registers to identify 13,598 women with first singleton pregnancies. They defined cohorts as: SLE as prevalent at delivery; incident within 0–2 years after delivery; and incident 2–5 years after delivery, assuming that this would serve as a proxy for immunologic burden during pregnancy.
Pregnancy and Contraception in Patients with Rheumatic Diseases
A number of abstracts at this year’s ACR in Chicago highlighted issues around pregnancy and contraception advice in routine clinical practice.
Azzouqah et al (Abstract 2653) examined the records of 131 women and found that 61% had contraceptive use documented in the medical records . Of patients taking teratogenic drugs, 67% had contraceptive use documented. Notably, more than 50% of patients taking potentially drug were not taking reliable contraception (abstinence, condoms) and 6% were using no contraception.
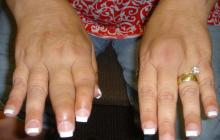
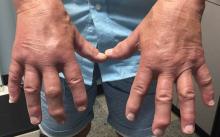
Psoriatic Arthritis Does Not Add to Pregnancy Problems
When psoriatic arthritis (PsA) patients become pregnant, they do not have more infertility or adverse pregnancy outcomes compared to healthy controls.
This study is relevant as the average onset age of PsA ranges between the 4th-6th decades of life and therefore includes a period of child-bearing potential.
Top 16 Drugs in Rheumatology 2016
Using data compiled from annual reports, SEC filings, press releases, company websites, recently released sales figures show that in 2016, 11 of the top 16 rheumatology drugs demonstrated blockbuster sales (>$1 billion per annum). Highlights from this report include:
Elder Rheumatoids Less Likely to Receive Biologics
Multiple studies have shown that elderly rheumatoid arthritis (RA) patients tend to be under-treated and receive DMARD therapies less often than younger RA patients. Now a VAMC study shows that the elderly are less likely to receive biologic agents yet are as likely to be subjected to glucocorticoid use.
A study using national US Veteran’s Affairs databases between 2005–2016 identified RA patients after their first-ever methotrexate (MTX) therapy and analyzed subsequent drug use.
Generic Price Fixing Alleged by State Prosecutors
The NY Times and 60 Minutes have reported that Connecticut, along with 43 other states, filed a mega-lawsuit accusing the generic drug makers of a massive, systematic conspiracy to bilk consumers out of billions of dollars. (Citation source: https://buff.ly/2vSmv03)
TNF Inhibitors Lag Behind Ustekinumab Survival in Psoriasis
A prospective national dermatology pharmacovigilance cohort (BADBIR) of 3523 biologic-naïve patients with chronic plaque psoriasis were analysed for drug survival. The overall survival rate in the first year was 77%, falling to 53% in the third year. Predictors of discontinuation included being female, current smoker or high baseline DLQI scores, while psoriatic arthritis improved drug survival. Data for patients on adalimumab (n=1879), etanercept (n=1098), infliximab (n=96) and ustekinumab (n=450) were available. Patients with ustekinumab had the best drug survival.
Brodalumab vs. Ustekinumab: Head-to-Head Studies of Psoriasis
AMAGINE-2 and AMAGINE-3 are in two phase 3 studies that compare multiple doses of brodalumab (anti-IL-17) and ustekinumab (anti-12/23) in adults with moderate-to-severe psoriasis. For 12 weeks, patients were blindly assigned to either 210 mg or 140 mg brodalumab (every 2 weeks), ustekinumab (45 mg or 90 mg every 12 weeks), or placebo.
Long-Term Benefits of Ixekizumab in Psoriatic Arthritis
The SPIRIT-P1 study has demonstrated the safety and efficacy of ixekizumab (an IL-17A antagonist) when given to adults w/ active psoriatic arthritis (PsA), and also demonstrating radiographic protection and improvement in skin outcomes as well.
This phase 3 study, enrolled biologic-naïve active PsA patients who randomly given either placebo, adalimumab 40 mg every 2 weeks (ADA) or ixekizumab 80 mg every 2 weeks (IXEQ2W) or every 4 weeks (IXEQ4W) after an initial dose of 160 mg. At week 16 or 24 ADA and placebo patients were re-randomized to IXEQ2W or IXEQ4W.
High Level Skin Responses Seen with Inhibitor of IL-23p19 in Psoriasis
Rheumatology News reports the results of a novel trial presented at the European Academy of Dermatology and Venereology meeting in Copenhagen.
In a head-to-head trial, 166 patients with moderate to severe plaque psoriasis were randomized to receive different doses of BI 655066 (selective inhibitor of the p19 subunit of interleukin-23) or ustekinumab. BI 655066, achieved higher rates of PASI 90 (81%). PASI 100 (54%) and PASI 75 (93%), but it did so much faster and maintained such responses longer than ustekinumab.
FDA Approves Taltz for Psoriasis
Tuesday the FDA approved the IL-17 inhibitor, Taltz (ixekizumab) for adults with moderate to severe plaque psoriasis. (Citation source http://buff.ly/1py10wd)
Taltz’s is an antibody (ixekizumab) that binds interleukin (IL)-17A. It's safety and efficacy were established in three randomized, placebo-controlled clinical trials involving 3,866 participants with plaque psoriasis who were candidates for systemic or phototherapy therapy.
Enbrel Approved for Pediatric Chronic Plaque Psoriasis
Etanercept (Enbrel) received FDA approval Friday for use in children with moderate to severe, chronic plaque psoriasis. Previously approved for adults with plaque psoriasis the new indication will be for chronic plaque psoriasis for those over age 4 years. Etanercept is now the first systemic therapy approved to treat pediatric psoriasis patients (ages 4-17).
Etanercept is already approved for adult rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis and polyarticular juvenile idiopathic arthritis (JIA) aged 2 years or above.
Future 2 Trial Shows Secukinumab Efficacy in Psoriatic Arthritis
Secukinumab (Cosentyx) is an anti-IL-17 monoclonal antibody currently approved for use in moderate to severe psoriasis. It has also been studied in psoriatic arthritis (PsA) and shown to be safe and effective.
Secukinumab Improves Patient-Reported Outcomes in Psoriatic Arthritis (FUTURE 1)
Secukinumab (Cosentyx) has recently been approved for use in ankylosing spondylitis and psoriatic arthritis; the latter largely based on the FUTURE 2 and the just reported FUTURE 1 clinical trial. FUTURE 1 demonstrates the efficacy and safety of secukinumab, with inital weekly intravenous loading, given monthly as a subcutaneous injection.
Minimal Disease Activity Falls Short in Psoriatic Arthritis
A small cohort study has evaluated the utility of the minimal disease activity (MDA) criteria that has been advocated for use in psoriatic arthritis (PsA) studies and clinical trials.
Among 41 patients with MDA, 7.4% did not fulfill the tender/swollen joint count. Moreover, 49% did not fulfill the skin criteria. Of the 42 patients not fulfilling MDA, 100% did not fulfill the patient pain score, 76.5% did not fulfill the patient’s global assessment, and the Psoriasis Area and Severity Index (PASI) was abnormal 65%.
Guselkumab, an IL-23 Inhibitor, Effective in Psoriatic Arthritis
The DISCOVER‐2 trial has shown that guselkumab, an interleukin‐23p19 was shown to be safe and effective in biologic‐naïve psoriatic arthritis (PsA) patients in a one year study.
This was a phase III trial that enrolled biologic‐naïve PsA patients with active disease (≥5 swollen+≥5 tender joints; CRP ≥0.6mg/dL) despite prior ineffective DMARD treatment. Patients were randomized to receive either subcutaneous injections of guselkumab (GUS) 100mg every‐4‐weeks (Q4W); guselkumab 100mg at Week0, Week4, Q8W; or placebo with crossover to guselkumab 100mg Q4W at Week24.
SPIRIT P1 Study - Ixekinumab Sustains Efficacy at 52 Weeks in Psoriatic Arthritis
Ixekinumab (IXE) is an IGG4 monoclonal antibody bindings with high affinity to IL -17A. Currently marketed at Taltz and approved for use in chronic plaque psoriasis, the agent is also being developed for use in psoriatic arthritis.
Today the results of the week 52, phase 3 SPIRIT P1 study were presented by Dr. Philip Mease and showed IXE to be superior to placebo (PBO) in achieving ACR 20 response in bDMARD-naive active PsA patients.
Obesity Associated with Late-Onset Psoriatic Arthritis
A recent study reports that obesity is linked to late-onset psoriasis and psoriatic arthritis (PsA). By contrast being of normal weight may be associated with an earlier onset of the disease adn HLA-B27.
Patients with early PsA were compared to those without arthritis (PsC), with regard to their body mass index (BMI) and HLA risk alleles for psoriatic disease (HLA-B*27, B*3901, B*3801, B*0801, B*4402, B*4403, and C*0602). A case-controlled designed was utilized to detect associations.
Several Agents Score on Radiographic Progression in Psoriatic Arthritis
Reduction on radiographic progression has now become the standard of care in the therapies used to treat psoriatic arthritis and, during the 2017 ACR Meeting, several agents reported the results of their studies with radiographic endpoints.