Articles By Jack Cush, MD

The RheumNow Week in Review - 20 October 2017
The RheumNow Week in Review discusses the past week's news, journal articles and highlights from RheumNow.com. In this week's report, Dr. Jack Cush discusses presentations from APLAR 2017 in Dubai, Catastrophic APS, safety of low-dose prednisone, frequency of psoriatic arthritis and a new drug for PsA and a hepatitis A outbreak in San Diego.
Read Article
Hepatitis A Outbreak in San Diego
On Sept. 1, 2017, the San Diego County declared a local public health emergency due to the ongoing hepatitis A outbreak in the county.
Read ArticleTofacitinib Scores in TNF inhibitor Resistant Psoriatic Arthritis
Mease et al has reported in the NEJM the results of a phase 3 trial showing tofacitinib (Tofa) to be significantly more effective than placebo in active psoriatic arthritis (PsA) patients who previously failed to respond to tumor necrosis factor inhibitor (TNFi) therapies.
Read Article
Rheumatologists are Slow to Change DMARDs
The paradigm of rheumatoid arthritis (RA) therapy mandates early diagnosis and aggressive treatment. Yet a recent cohort study has shown that RA patients with moderate to high disease activity (MHDAS) were met with infrequent DMARD adjustments; with median time to DMARD adjustment being 5 months and median time to low disease activity (LDAS) was roughly 10 months.
Read Article
Gout Increases Risk of Stroke Moreso than MI
Gout coassociates with many of the disorders within the metabolic syndrome - diabetes, hypertension, hyperlipidemia and obesity. Not surprisingly, gout is a risk factor for cardiovascular disease, but the risk magnitude for myocardial infarction (MI), and stroke are unclear.
Read Article
Stelara FDA Approved for Use in Adolescent Psoriasis
Johnson & Johnson has announced that they have received an expanded FDA approval for the use of ustekinumab (Stelara) in treating adolescent patients, aged 12 and over, with moderate to severe plaque psoriais.
Read Article
Across the Table: Cush & Erkan on Antiphospholipid Syndrome
The antiphospholipid syndrome (APS) is a common disorder affecting patients with and without autoimmune disease. Despite wider recognition of APS among physicians as well as the expanding research collaborations, many clinical questions are still encountered in clinical practice, which require further evidence-based studies. In this “Across the Table” edition, Drs. Cush and Erkan discuss some of these APS-related questions. Our guest expert, Dr. Doruk Erkan offers up his approach to diagnosis and management of APS.
Read Article
High Drug Discontinuations and Nonadherence in Fibromyalgia
Compliance and adherence was shown to be quite poor in fibromyalgia (FM) patients, with high rates of drug discontinuation and greater than 60% demonstrated low adherence.
Read Article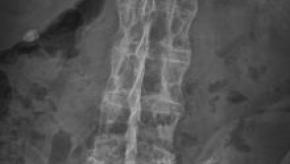
Spinal X-ray Inhibition in Ankylosing Spondylitis Best Achieved by Control of Activity
Although tumour necrosis factor inhibitors (TNFi) are highly effective (on clinical grounds) in ankylosing spondylitis (AS), proof that they may alter the course of spinal radiographic progression has been scant.
Read Article
13 October 2017 The RheumNow Week in Review
The RheumNow Week in Review discusses the past week's news, journal articles and highlights from RheumNow.com. In this week's report, Dr. Jack Cush discusses a novel antiviral drug or the use of stem cell therapy for scleroderma, rituximab in HSP, when to withdraw NSAIDS in RA, and congrats to PAs and NPs!
Read Article



