All News
Lupus Nephritis Despite Low Level Proteinuria
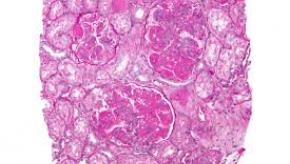
Kidney International reports on a series of systemic lupus erythematosus (SLE) patients, who despite inactive urinary sediment and low level proteinuria, had a high rate of glomerulonephritis (GN) proven by renal biopsy; moreover, the LN was not predicted by laboratory abnormalities.
Read ArticleJAK Inhibitor Misses Endpoint in Safety Study. Now What?
Pfizer announced results Wednesday from its FDA-mandated postmarketing safety study of tofacitinib (Xeljanz), and they don't bode well for the drug and possibly others in its class.
Read ArticleRheumNow Podcast – Don’t Hold the Benlysta, Take the Vaccine (1.22.2021)
Dr. Jack Cush reviews the news and journal articles from the past week on RheumNow.com, with more than a dozen items to discuss on this week's podcast.
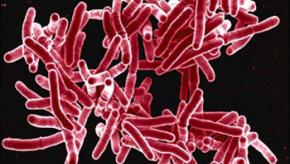
Read ArticleLow Risk of TB with Secukinumab
JAMA Dermatology has reported on a longitudinal cohort study of 12,319 secukinumab (SEC) treated patients (psoriasis, psoriatic arthritis, or ankylosing spondylitis); no new cases of active tuberculosis (TB) and very few cases of latent tuberculosis infection (LTBI) were f
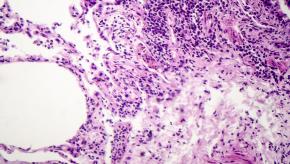
Read ArticleMortality Risk in Rheumatoid Lung Patients
A Medicare claims analysis of rheumatoid arthritis (RA) patients showed that RA-associated interstitial lung disease (RA-ILD) was seen in nearly 5% of patients and was found to increase mortality risks, including respiratory and cancer mortality not explained by other factors.
Read ArticleRheumNow Podcast – Steroids, IVIG & the Vaccine (1.15.2021)
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com. How to handle COVID19 Vaccine issues, IL-6 inhibition in COVID, Comorbidites in RA, IVIG in dermatomyositis, & more...
Read Article2020 Rheumatology Year in Review
2020 was historic, memorable and game-changing. Under the cloud of COVID-19, there were many significant and memorable advances and setbacks for the rheumatology world. In our accounting of most read articles for 2020, (not surprisingly) 17/20 were COVID-related.
Read ArticleProDERM Study: IVIG in Dermatomyositis
The ProDERM study design and outcomes are previewed in Medicine; and as promised the results were revealed at ACR Convergence in November 2020 showing that IVIg is safe, effective and superior to placebo i(PBO) in patients with dermatomyositis (DM).
Read ArticleDelays in Diagnosis of Axial Spondyloarthritis
The diagnosis of axial spondyloarthritis (and anklylosing spondylitis) (axSpA) is often delayed and usually not diagnosed by rheumatologists; a recent metanalysis shows that longer delays were attributable to lower education levels, younger age at onset and absence of extra-articular manifes
Read ArticleRheumNow Podcast – Lights, Camera, Zoom (1.8.2021)
Dr. Jack Cush reviews the news, journal articles and favorite blogs from this past week on RheumNow.com.
Read ArticleEULAR Guidance on Patient Adherence to Meds
Non-adherence may be the single most untold risk to suboptimal outcomes in the care of musculoskeletal disease. It has been estimated that 30-80% of patients with rheumatic and musculoskeletal diseases (RMDs) are nonadherent to prescribed regimens. EULAR has commissioned a task force
Read Article
Links:
Links:

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Dr Philip Robinson philipcrobinson ( View Tweet)

Dr. John Cush RheumNow ( View Tweet)

Links:

Links:

Links:
Dr. John Cush RheumNow ( View Tweet)