All News
FDA Puts Boxed Warnings on JAK Inhibitors
Based on the safety review of tofacitinib in Pfizer's Oral Surveillance (1133) study, the FDA has added serious boxed warnings to all three marketed JAK inhibitors (for inflammatory diseases) and formalized the recommendation that patients should be started on a TNF inhibitor (TNFi) before trying a JAK inhibitor. These recommendations apply to tofacitinib, baricitinib and upadacitinib.
Read ArticleDoes Control of Inflammation Lower Cancer Risk?
Dr. Jonathan Kay's provocative video addresses whether the increased cancer risk associated with tofacitinib (Tofa) seen in the Pfizer Oral Surveillance (1133) study represents a real increased risk, an increased risk relative to a decreased risk with TNF inhibitors (TNFi) or an actual decreased cancer risk (unknown as there was no placebo comparator in this trial).
Read ArticleACR21 Best Abstracts - Day 2
The RheumNow faculty reporters have been scouring and reporting on the best abstracts from the ACR. Here is a sampling of their choice abstract presentations reported during ACR 2020 Day 2 (#ACRbest).
Read Article
Links:

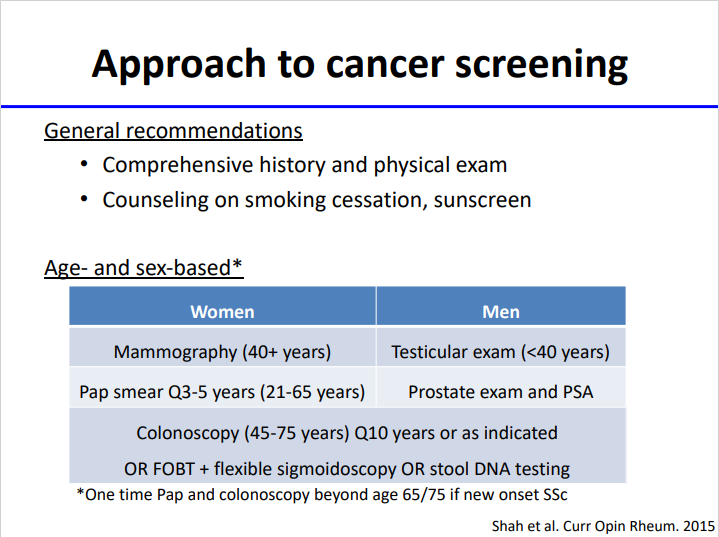
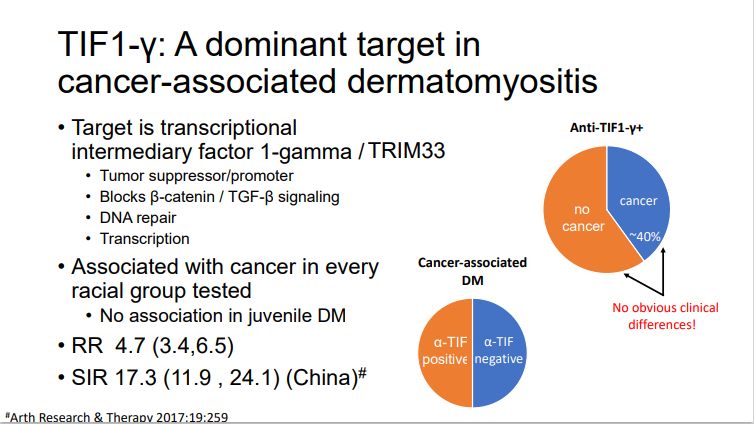
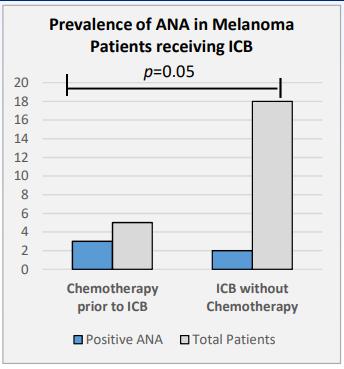
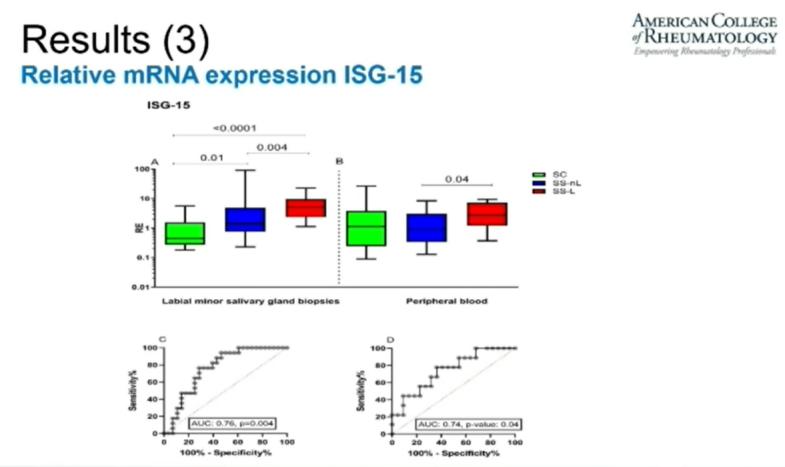



David Liew drdavidliew ( View Tweet)

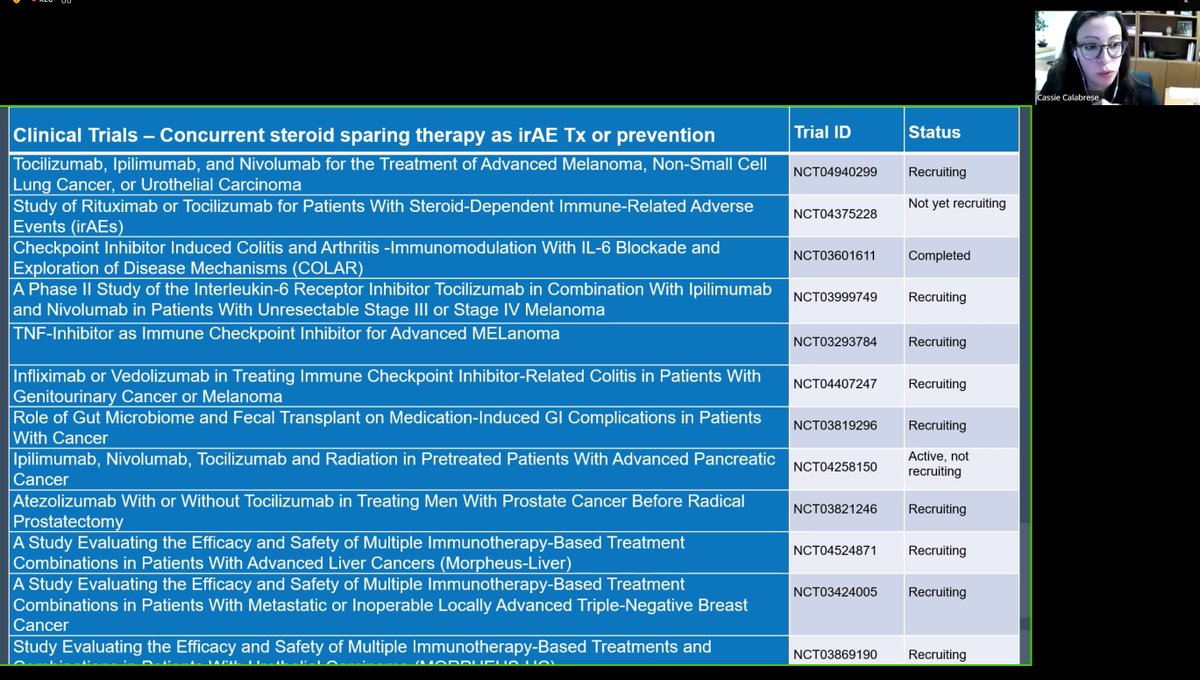
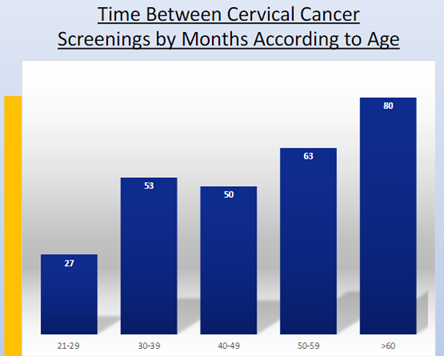

Meral K. El Ramahi, MD MeralElRamahiMD ( View Tweet)
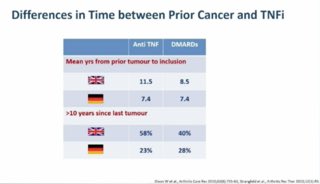

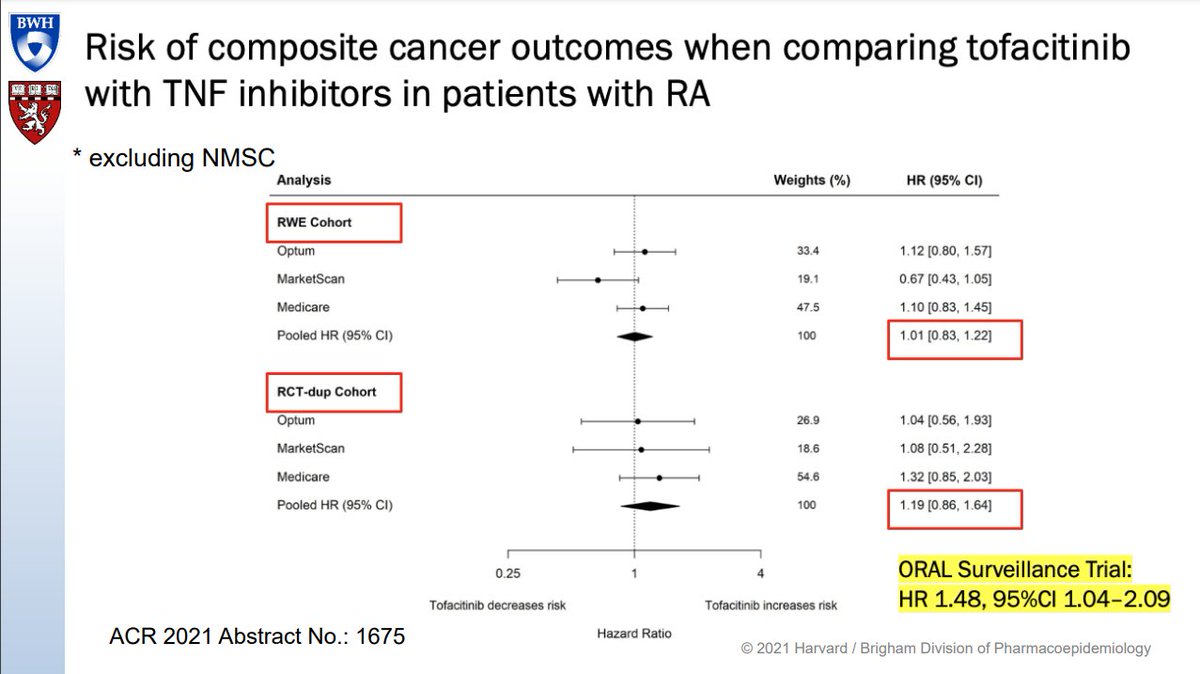

Olga Petryna DrPetryna ( View Tweet)






