All News
Advantages of Intravenous Pulse Cyclophosphamide in ANCA-associated Vasculitis
Researchers from the Nottingham University Hospitals have analyzed the efficacy and safety of oral (PO) and intravenous (IV) cyclophosphamide (CTX) in ANCA-associated vasculitis (AAV) patients and demonstrated a trend for fewer relapses, better 1 year survival and less toxicity w
Read ArticleThe RheumNow Week in Review - 15 December 2017
Dr. Jack Cush reviews the news from the past week on RheumNow.com. Subscribe to the podcast on iTunes and SoundCloud.com.
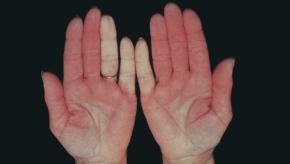
Read ArticleCochrane Review: Calcium Channel Blocker Efficacy in Raynaud's
The Cochrane Database has published its review of calcium channel blockers (CCB) in Raynaud's phenomenon, showing CCBs may be useful in reducing the frequency, duration, severity of attacks, pain and disability associated with Raynaud's phenomenon, especially with primary Raynaud's.
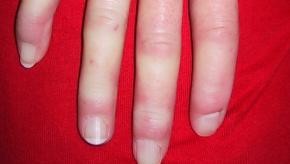
Read ArticleFatigue, HAQ and Hand Disability Correlated in Early Systemic Sclerosis
Early diffuse cutaneous systemic sclerosis (SSc) was associated with high levels of disability and fatigue, according to analysis of data from the European Scleroderma Observational Study cohort.
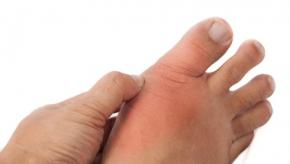
Read ArticleFebuxostat Works in Early Gout But Fails to Protect from Xray Damage
Dalbeth and colleagues have published a novel report in Arthritis & Rheumatology, novel in that it is a trial of early gout patients that looks at clinical and radiographic outcomes. They showed that febuxostat improves magnetic resonance imaging (MRI) measures of synovitis and reduced gout flares, but failed to alter X-ray progression with 2 years follow-up.
Read ArticleFDA Approves Mepolizumab for Churg-Strauss (EGPA)
The Food and Drug Administration (FDA) approved use of Nucala (mepolizumab) for use in treating adults with eosinophilic granulomatosis with polyangiitis (EGPA), previously known as Churg-Strauss vasculitis. This is first FDA-approved therapy specifically to treat EGPA.
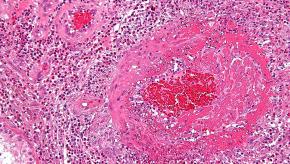
Read ArticleKidney Dysfunction Frequent in RA
Renal dysfunction is common among patients with rheumatoid arthritis (RA), particularly among older patients, women, and those with hypertension, Japanese researchers reported.
Read ArticleOffspring of RA Women Have Higher Risk of RA and Other Diseases
Despite a growing body of evidence suggesting that maternal health is more important than maternal medications to fetal and infant outcomes, little is known about the long term oucomes of infants born to mothers with rheumatoid arthritis (RA). Using Danish population data, researchers have shown in utero exposure to maternal RA is associated with an increased risk of thyroid disease, and an increased risk of future RA.
Read ArticleAASM Guidelines for Sleep Apnea Testing
A task force from the American Academy of Sleep Medicine (AASM) performed a systematic review of the scientific literature on obstructive sleep apnea and screening tests.
Read Article2017 Update of Treat to Target Recommendations for SpA/PsA
A 2017 international committee of experts met to assess and revise its earlier 2012 recommendations regarding treating to target in patients with psoriatic arthritis (PsA) and axial peripheral spondyloarthritis (SpA).
Read ArticleWhy TNF Inhibitors May Work in Some Autoinflammatory Patients
The NLRP3 inflammasome is a critical component of the innate immune system and activation of NLRP3 inflammasome results in caspase-1–dependent secretion of the proinflammatory cytokines IL-1β and IL-18.
Read ArticleThe RheumNow Week in Review - 8 December 2017
Dr. Jack Cush covers the news and journal articles published on RheumNow.comin the past week.
Read ArticleInfliximab Does Not Increase Perioperative Infection
Staying on a TNF inhibitor (TNFi) throughout major surgery has generatlly been associated with higher rates of perioperative infection (https://buff.ly/2iBFVjp). On the other hand, discontinuation of the TNFi prior to surgery is associated with lower ra
Read ArticleRheumatology Year in Review
During 2017, the improvements and refinements seen during previous years in the treatment of rheumatoid arthritis have been extending to other conditions ranging from psoriatic arthritis to lupus and for both monoclonal antibodies and oral small molecule medications.
Read ArticlePRECISION Subanalyses Question Aspirin Use
The PRECISION trial reported last year that celecoxib appears to be safer than the NSAIDs naproxen or ibuprofen in treating osteoarthritis (OA) and rheumatoid arthritis patients who are at increased cardiovascular risk. New data suggests that adding aspirin may nullify this advantage, according to a study presented at the annual American Heart Association meeting and reported by Medscape.
Read ArticleFibromyalgia Diagnosed by Two Simple Tests
Researchers from the Oregon Health Science Center have reported that fibromyalgia can be distinguished from chronic pain by primary care providers (PCPs) who employ two simple screening tests: BP cuff-evoked pain and a single patient question, and pain induced by pinching the Achilles tendon.&nbs
Read ArticleIxekizumab (Taltz) FDA Approved for Psoriatic Arthritis
On Friday, December 1st, the FDA approved Lilly's drug Taltz (ixekizumab) for use in adult patients with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
Taltz, an IL-17 inhibitor, has previously been approved for plaque psoriasis where it has shown high level PASI75 and PASI100 responses.
Read ArticleThe ACR17 RheumNow Week in Review - 1 December 2017
Dr Jack Cush reviews nighlights and news from the past 2 weeks on RheumNow.com. This week's report includes new drug approvals, disappointing ACR guidelines, Lyme & Zika, infertility, dermatomyositis skin outcomes and myositis-associated cancer testing.
Read ArticleSkin Remission in Dermatomyositis is Uncommon
JAMA Dermatology reports that clinical remission was relatively uncommon in dermatomyositis, despite aggressive systemic therapy, but was best in those receiving mycophenolate during a 3-year study.
Read Article