All News
ACR Best Abstracts - Day 1
The RheumNow faculty reporters have been scouring the meeting and online presentations to find the best abstracts from ACR22. Here are some of their choice abstracts reported today on day 1 of ACR 2022 (#ACRbest).
Read Article
Don’t be ‘ticked’: TYK2 is not a JAK!
Recently the FDA approved deucravacitinib, a highly selective TYK2 inhibitor for psoriasis. Trials are positive in psoriatic arthritis and a phase II study in SLE. What about the effects? Presentations from the ACR22 meeting may provide answers.
Read Article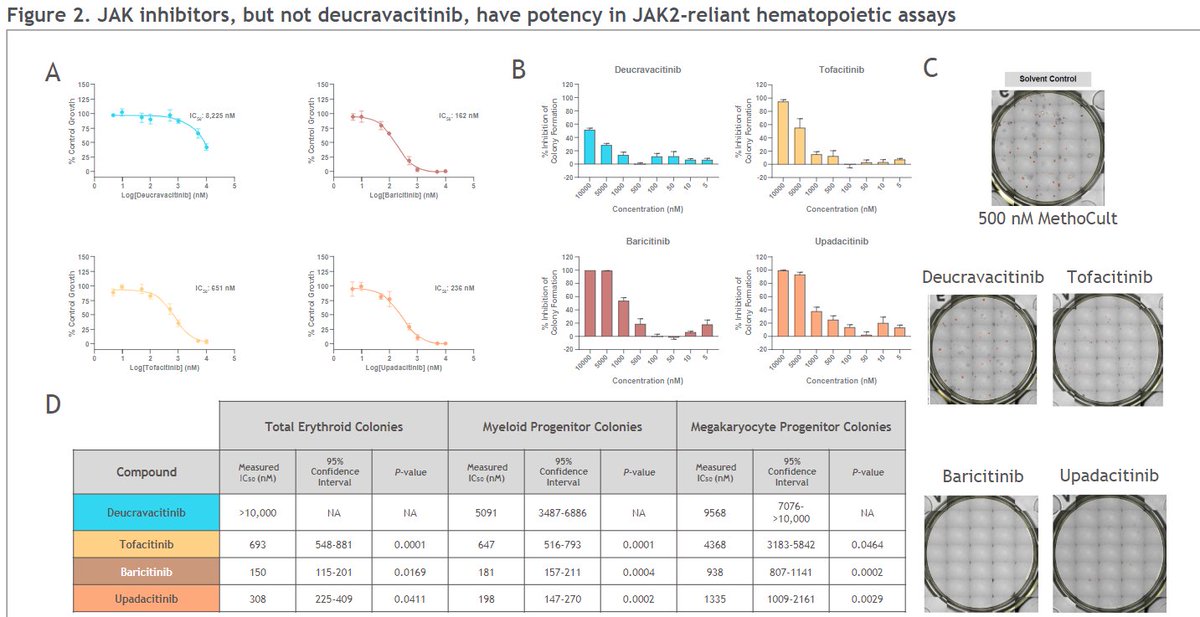
Johnson et al TYK2i deucravacitinib does not inhibit haematologic pathways, Treg function, or IL-15 dependent NK cell function, that JAKi do. @rheumnow #ACR22 Abstr#0584 https://t.co/UTHzUjGmJN https://t.co/NUKKAv3tAr
Richard Conway RichardPAConway ( View Tweet)

Trend of reduced opioid use when initiating biologics and JAKi in RA though effect size small and very high baseline use (>50%) in this insurance claims database.
Residual pain and harm minimisation still areas of need.
@RheumNow #ACR22 ABST0925
https://t.co/XGHu0fIMF0 https://t.co/PutNNQiKnY
Julian Segan JulianSegan ( View Tweet)
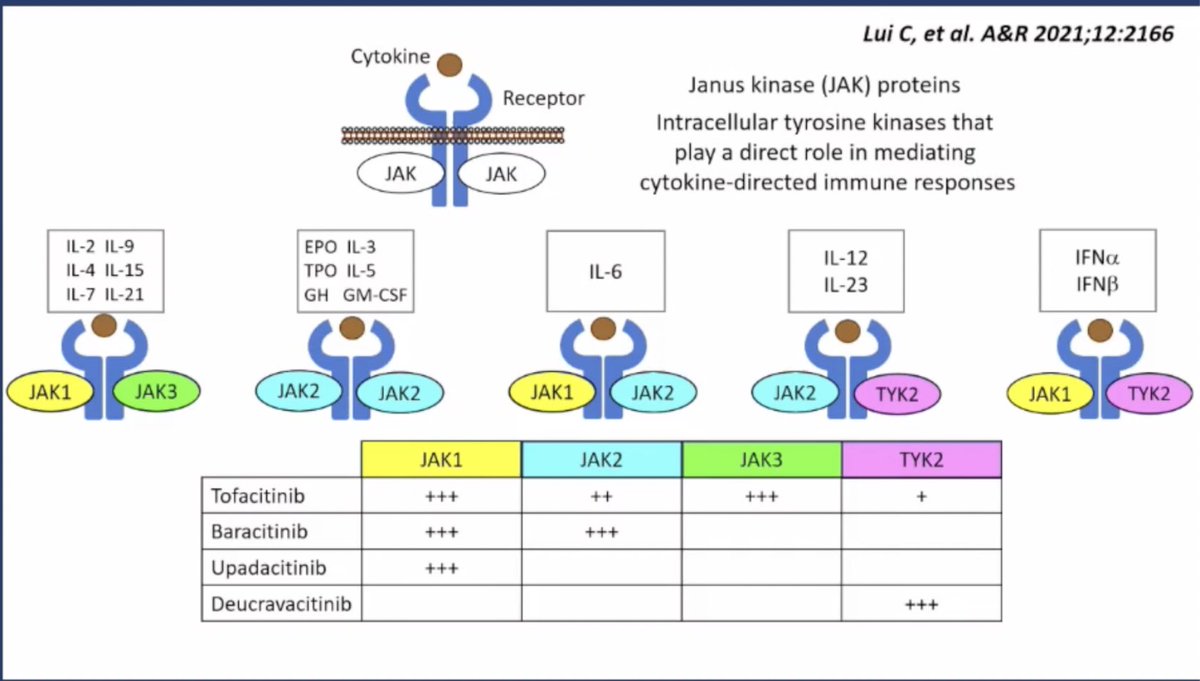
JAK and TYK-2 inhibitors and indications in SpA:
● Tofacitinib ➣ Pan JAKi (AS, PsA)
● Baricitinib ➣ JAK1,2
● Upadacitinib ➣ JAK1 (AxSpA, PsA)
● Deucravacitinib ➣ TYK2 (PsA)
Carol Langford, Year in Review #ACR22 @RheumNow https://t.co/EMLEvptySS
Dr. Antoni Chan synovialjoints ( View Tweet)
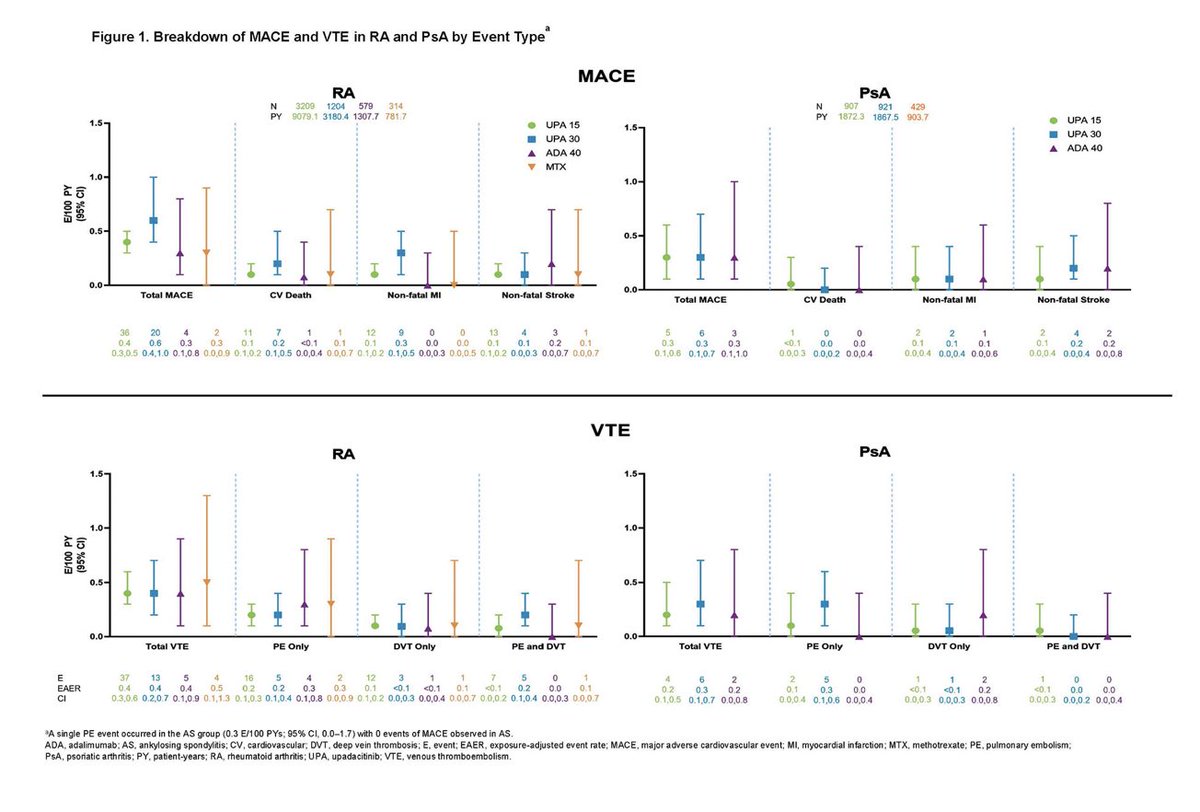
Across 9 trials of Upadacitinib in RA, PsA and AS, 40%–50% of patients had ≥ 2 CV risk factors, ≥ 65 years 6%–23%. Rates of adjudicated MACE and VTE with UPA were infrequent, consistent with background rates in RA, PsA, and AS Abst 0510 https://t.co/HDkQ6ELnAy #ACR22 @RheumNow https://t.co/SRJYdFFNIQ
Dr. Antoni Chan synovialjoints ( View Tweet)
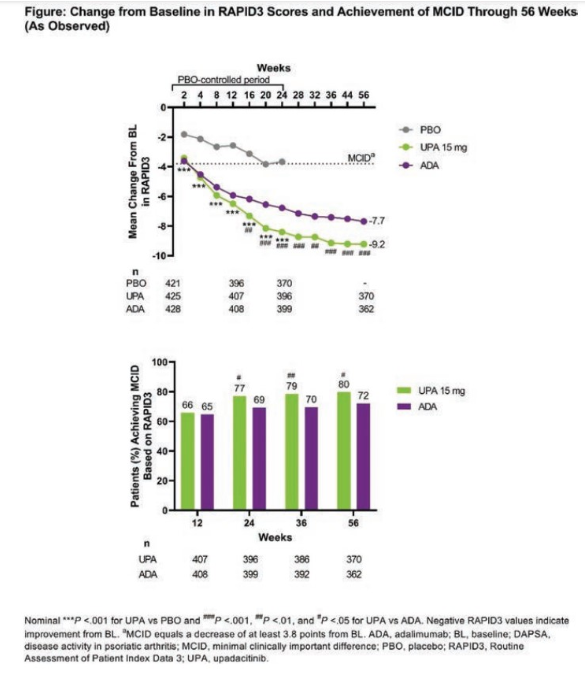
Post hoc analysis from the SELECT-PsA 1 trial by Dr. @DrLauraCoates and team:
💊Pts on UPA showed greater improvement from BL in RAPID3 vs. ADA in all visits
💊Pts on UPA had better responses in all assessments vs. PBO
Reassuring data. #ACR22 @RheumNow ABST0192 https://t.co/NgeUU5bPPf
sheila RHEUMarampa ( View Tweet)
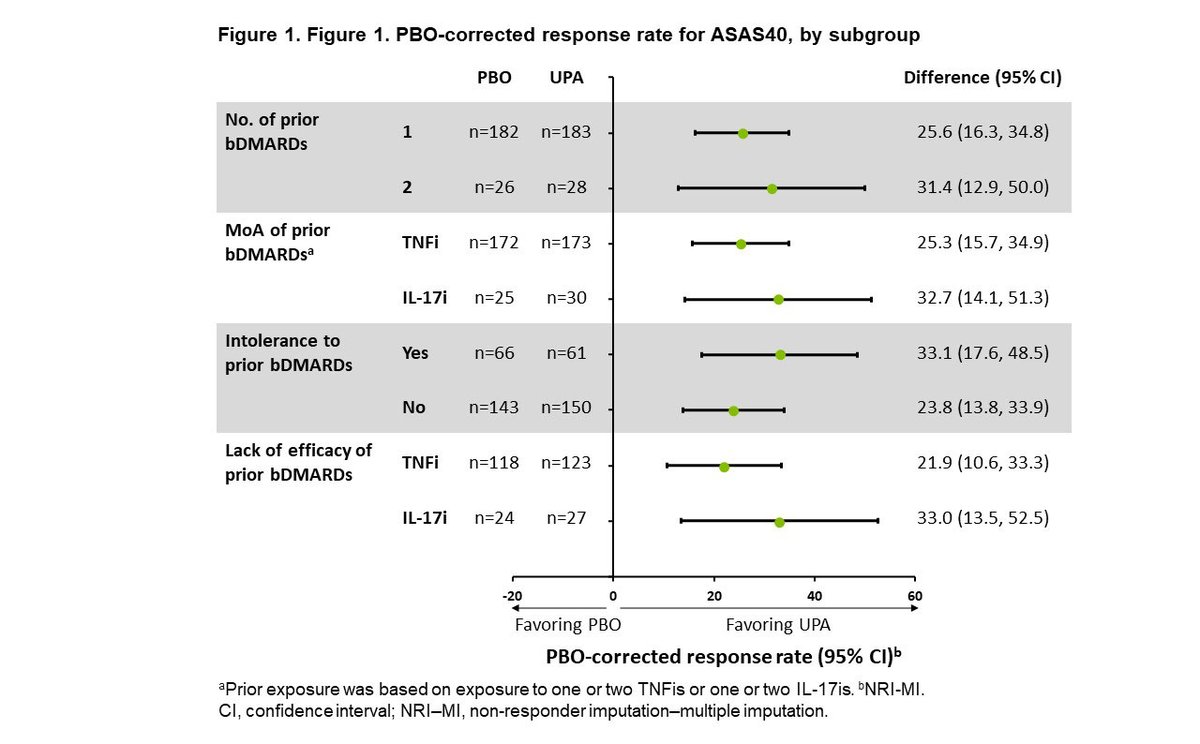
SELECT-AXIS 2 subgroup analysis. UPA demonstrated improved efficacy vs PBO at Wk 14 across all evaluated subgroups of pts with bDMARD-IR AS. No new safety signals. @XBaraliakos et al, Abs 0414 #ACR22 https://t.co/lTfm68XlEY https://t.co/PaWdrOljMO
Dr. Rachel Tate uptoTate ( View Tweet)
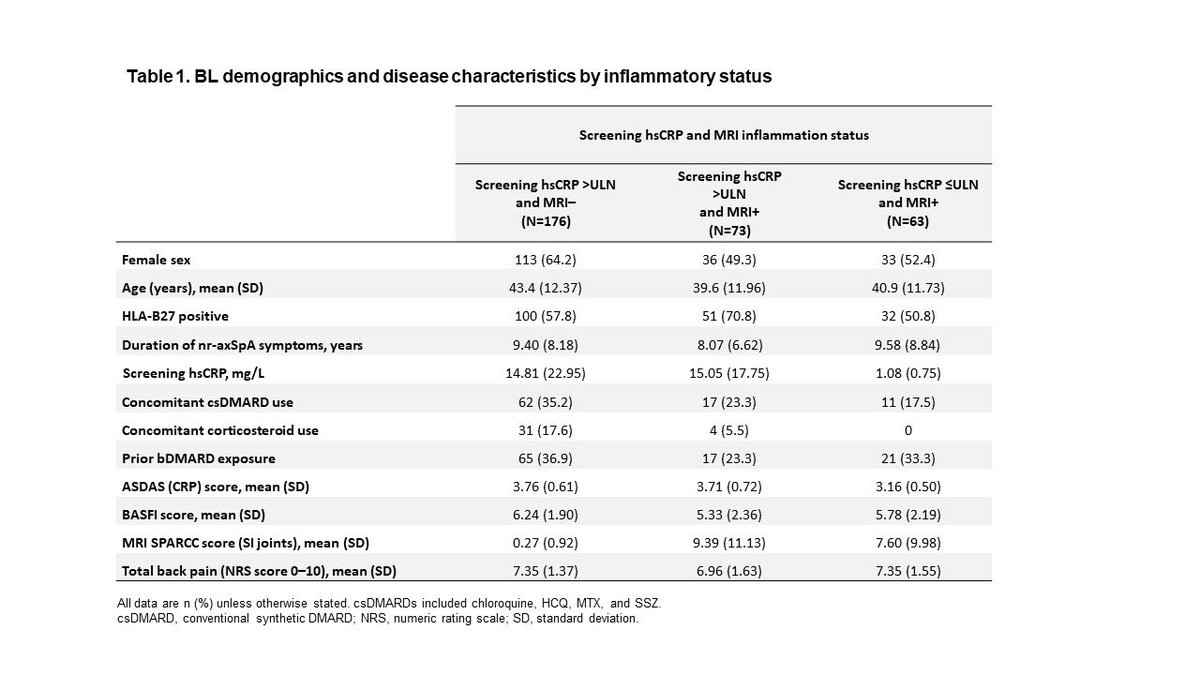
Abs 0419 SELECT-AXIS 2, UPA improved outcomes vs PBO in nr-axSpA pts across all BL inflammation subgroups; greatest benefit obs in pts with both elevated CRP and inflammation on baseline MRI. #ACR22 @RheumNow https://t.co/opaFwXgcvU https://t.co/o27ULcL8Ip
Dr. Rachel Tate uptoTate ( View Tweet)
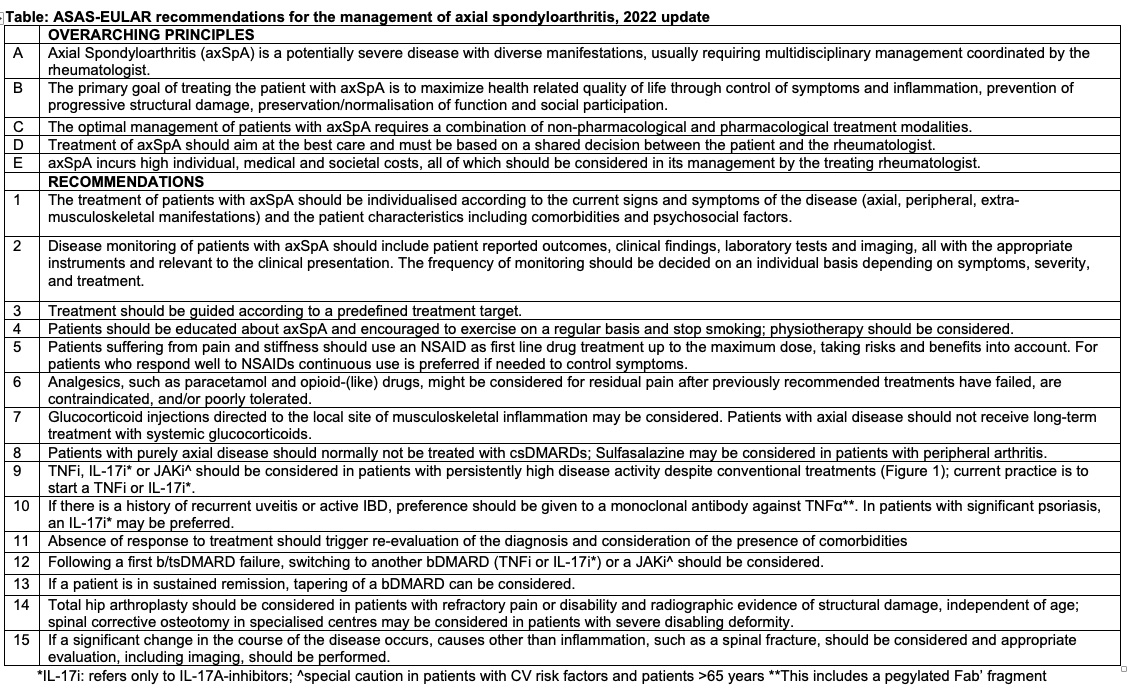
2022 ASAS-EULAR Recommendations of AxSpA management
1) NSAIDs still first line
2) Analgesics/opioids contraindicated
3) TNFi, IL-17i first line bDMARDs, followed by JAKinibs
4) Tapering but not discontinuation of bDMARDs in sustained remission
Abs#0542 @RheumNow #ACR22 https://t.co/ffaN2fMc3v
Robert B Chao, MD doctorRBC ( View Tweet)
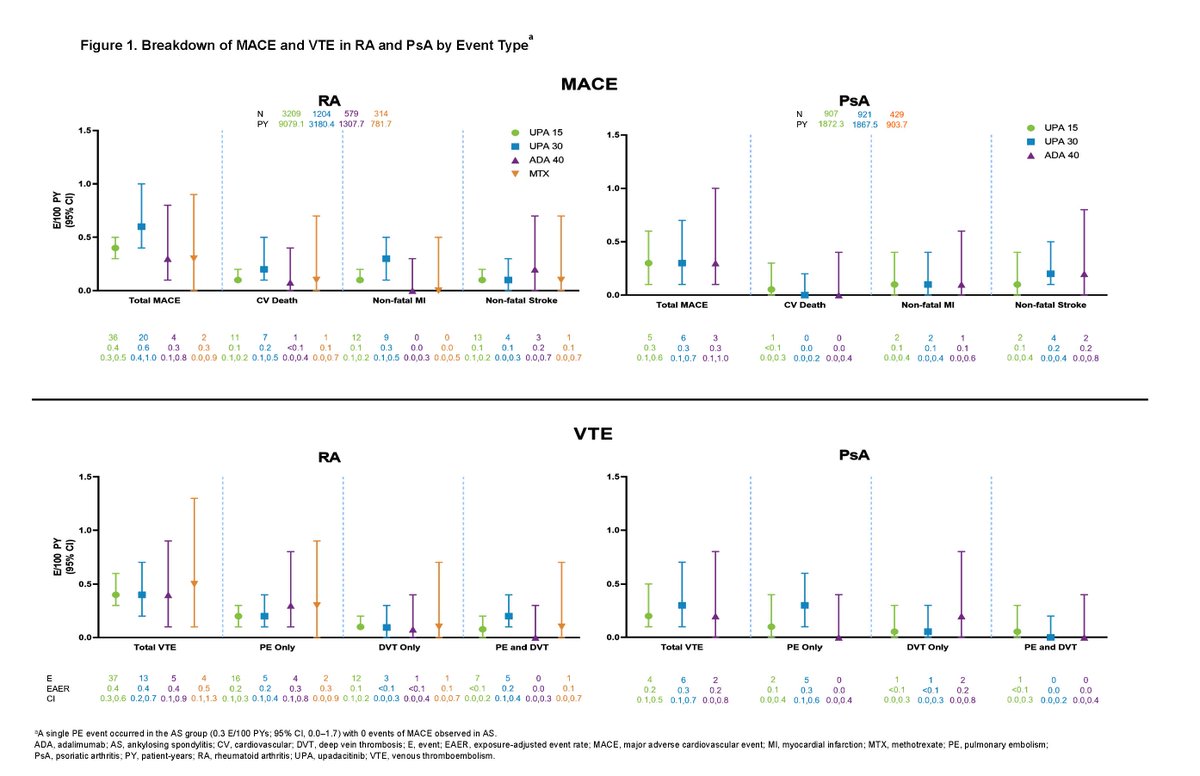
Rates of MACE and VTE with upadacitinib were infrequent and consistent with background rates in RA, PsA and AS patients. Factors associated with MACE/VTE: age>65, HTN, DM, smoking, history CV event/VTE
Abs#0510 @RheumNow #ACR22 https://t.co/AjtJF0p3QK
Robert B Chao, MD doctorRBC ( View Tweet)
Key points in managing MDA5+ DM
1. Consider Tofacitinib 5-10mg bd
2. Low to mid dose Prednisolone 20-30mg tapering
3. Other Rx: Tacrolimus, IVIG
4. Poor prognostic factors: lymphopenia, raised ferritin, old age, rapid ILD progression
Wang GC, IIM session @RheumNow #ACR22 https://t.co/UF2PjAg0Mw
Dr. Antoni Chan synovialjoints ( View Tweet)
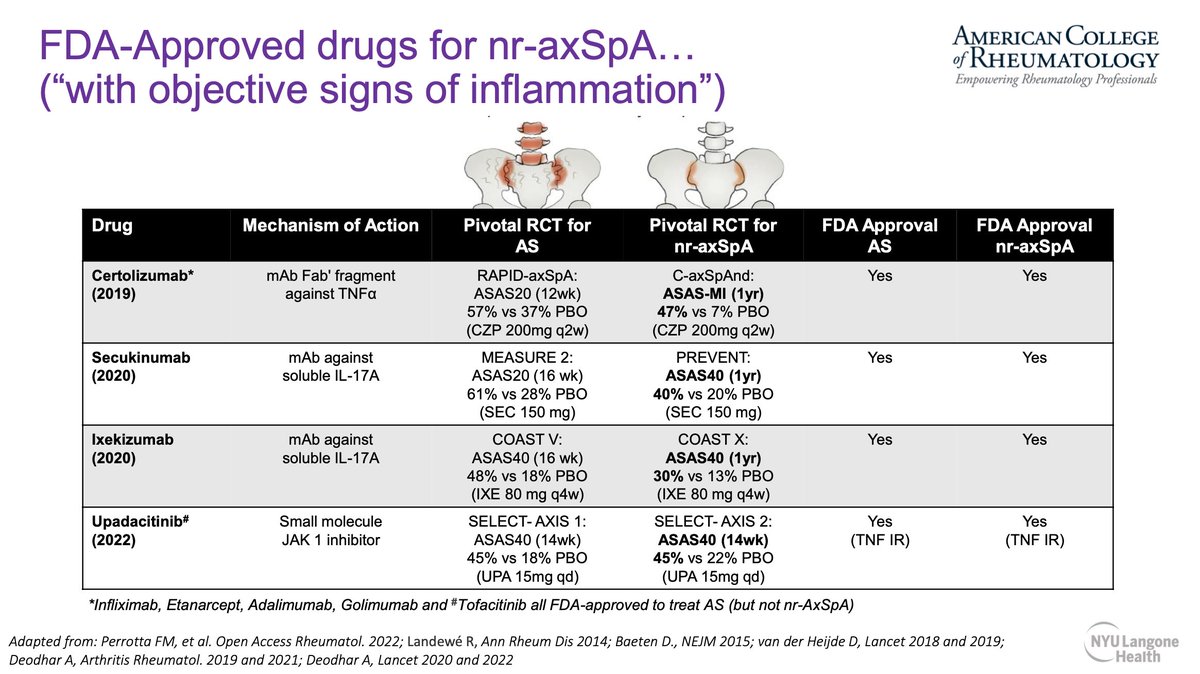
Therapies for AS and nr-AxSpA are coming together. Both AS and nr-AxSpA present with similar burden of disease. TNFi, IL-17i and JAKi now approved for use in nr-AxSpA.
Jose U Scher, SpA Review @RheumNow #ACR22 https://t.co/vcHcd7OyAU
Dr. Antoni Chan synovialjoints ( View Tweet)

What do you do if a person with #rheumatoidarthritis was great on 1st JAKi. It was 1st line advanced Rx @RheumNow #ACR22
Janet Pope Janetbirdope ( View Tweet)
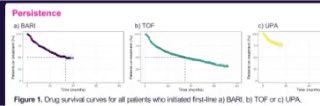
OPAL data is a gem! Large Australian data studying JAKi use in #rheumatoidarthritis N=5900 Switching JAKi~JAKi-common. As in TNFi 1st line Rx had longer retention & better efficacy than 2nd & subsequent. Median 1st advanced Rx - same in all JAKs 34 MOA @RheumNow #ACR22 abst0274 https://t.co/FQU4AKXwe9
Janet Pope Janetbirdope ( View Tweet)

Xeljanz in PsA (@pfizer funded) #ACR22 @RheumNow
⚠️safety data
-Pancytopenia
-Transaminitis
-Dyslipidemia
-Elevated CPK without #myositis
-Avoid in pregnancy and GI strictures
🤰Stop JAKi at least one month prior to conception🤰
Catherine Sims, MD DrCassySims ( View Tweet)
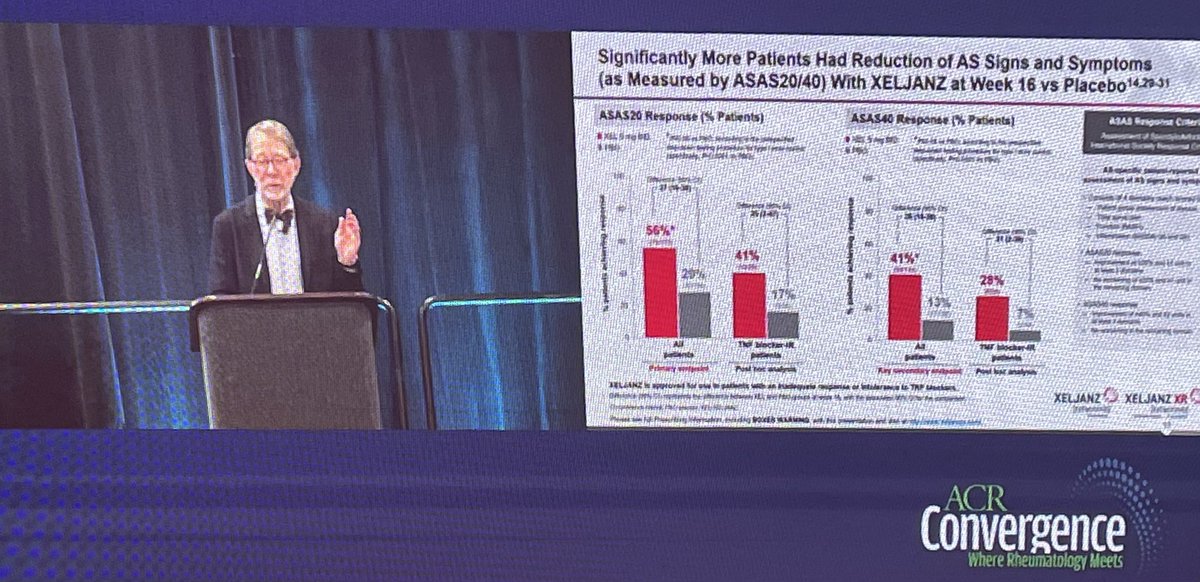
Xeljanz use in AS #ACR22 (@pfizer sponsored) @RheumNow
-Included patients with at least bilateral grade 2 SI or grade 3/4 unilateral SI
-5mg BID Xeljanz
-Significantly more patients had ASAS20/40 response, CRP reduction, & QOL, vs. placebo
-Onset of action noticed at 2 weeks https://t.co/DisKpgR0pt
Catherine Sims, MD DrCassySims ( View Tweet)
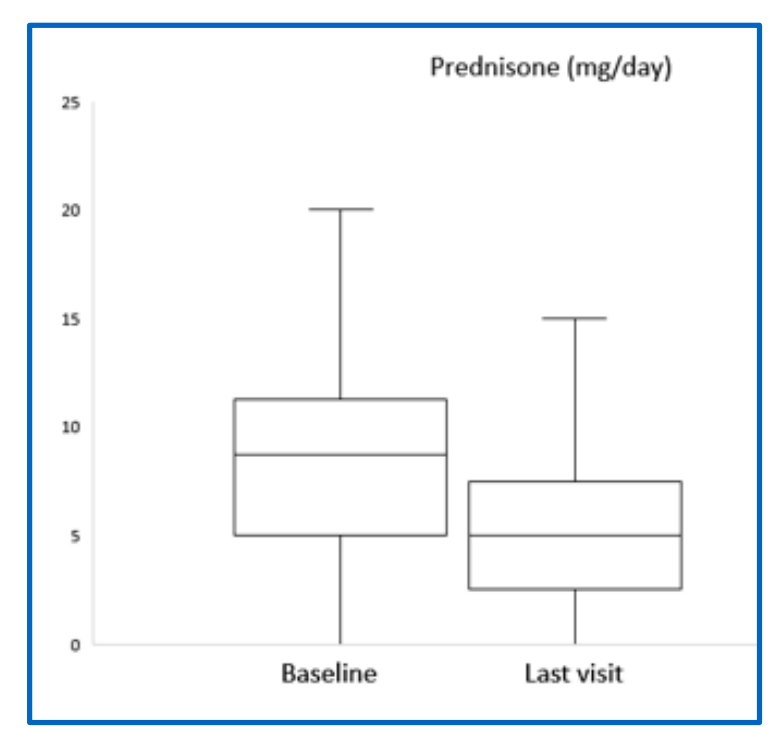
@MayoClinic data for bari in relapsing GCA caused waves at #ACR21
Spanish data looking at real-world practice: slightly longer, slower GCA, but equally encouraging outcomes
waiting for SELECT-GCA with interest! @EBRheum
ABST0464 #ACR22 @RheumNow https://t.co/pVRPXAohhc https://t.co/FgNNfPSWk3
David Liew drdavidliew ( View Tweet)
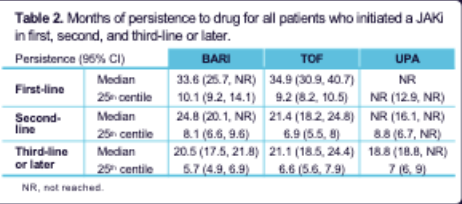
OPAL dataset 🇦🇺: 55000+ RA pts, 1/10 first line JAKi. Persistence > if 1st line,< x2 if 3rd line.
1/2 switch JAKi->TNFi
1/3 switch JAKi ->JAKi
Why high proportion of JAKi cycling, when AE reason for switch low?
https://t.co/APdWW7fzJE
Abst #0274 #ACR22 @Rheumnow @ClaireDeakin10 https://t.co/3xM3lQO1wI
Aurelie Najm AurelieRheumo ( View Tweet)
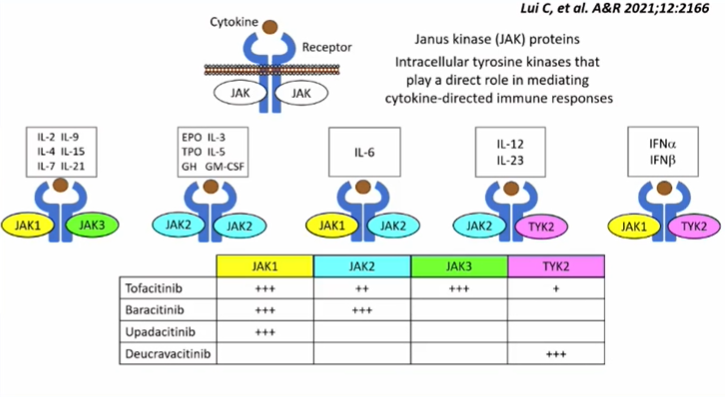
Very helpful slide on the different MOAs of JAK inhibitors (by Lui C et al.) shared by Dr. Langford
#ACR22 @RheumNow #RheumTwitter #yearinreview https://t.co/9VIOmzNoMq
sheila RHEUMarampa ( View Tweet)





