All News
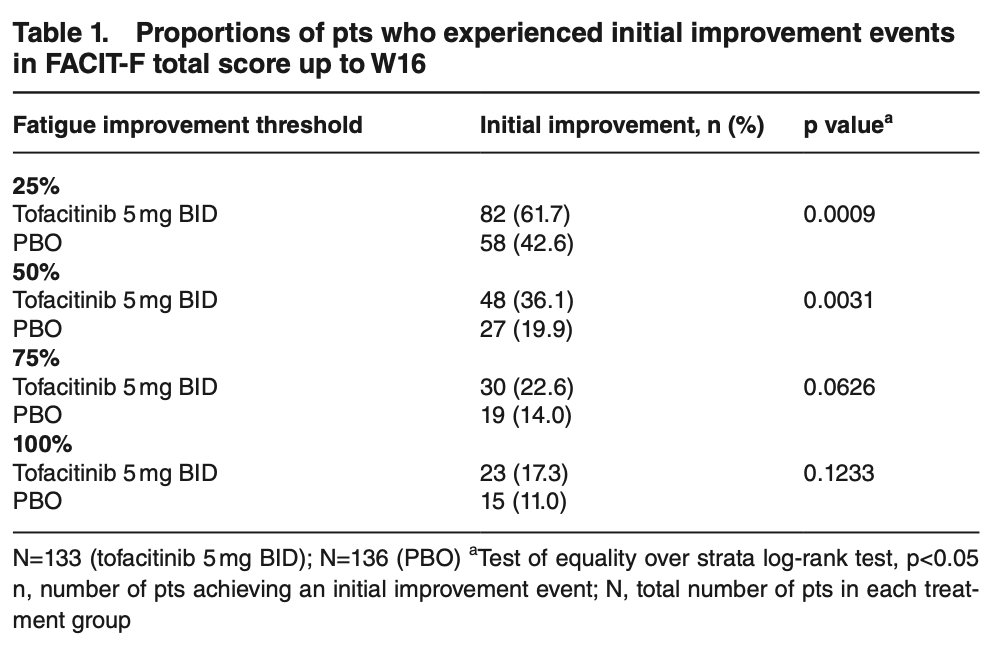
Tofacitinib had faster and larger impact on improvement of fatigue compared to placebo in treatment of ankylosing spondylitis pts.
Note 100% fatigue resolution roughly same vs. placebo
@RheumNow #EULAR2022 ABST#POS0305 https://t.co/Doep91QIRa
Robert B Chao, MD doctorRBC ( View Tweet)
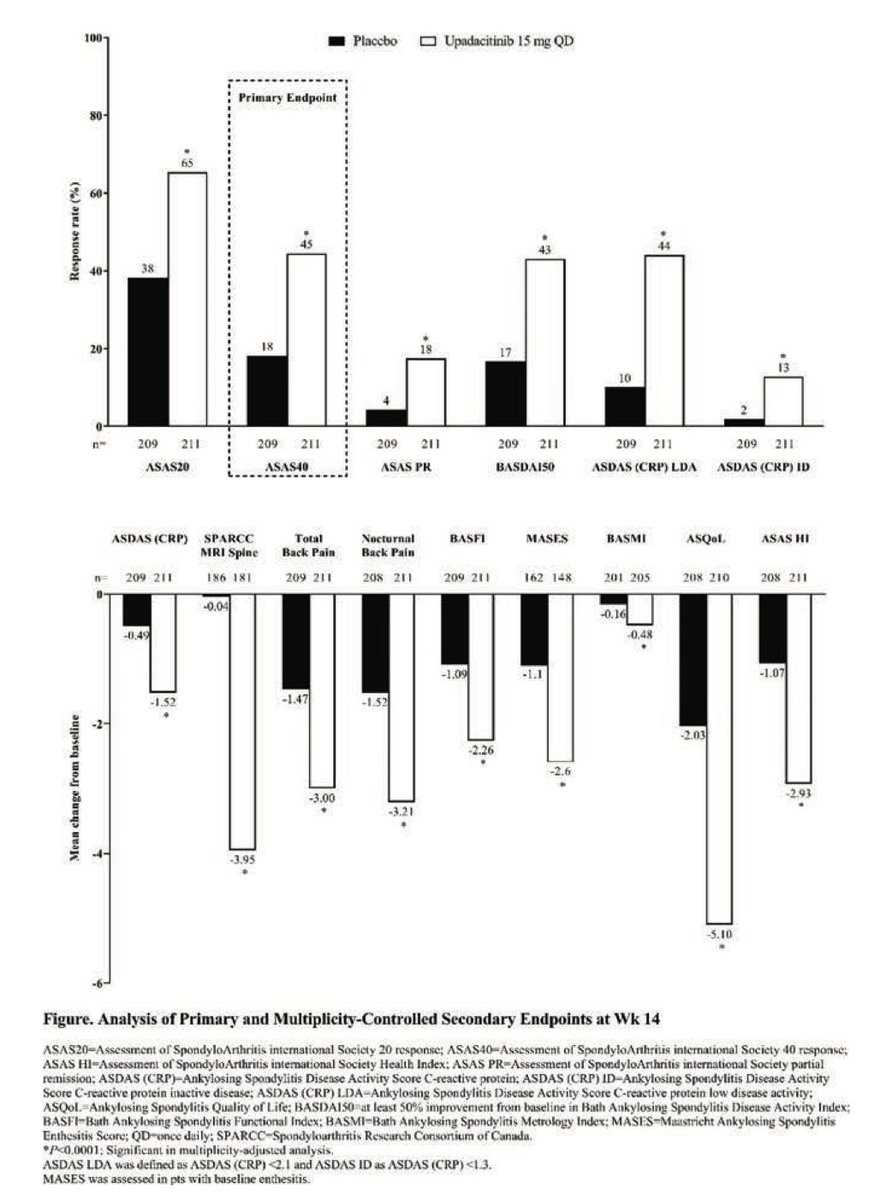
Upadacitinib effective in treatment of ankylosing spondylitis pts refractory to biologic tx - phase 3 trial
Primary endpoint (ASAS40) and secondary endpoints met
No malignancy, MACE, VTE, death
@RheumNow #EULAR2022 ABST#POS0306 https://t.co/UL4hUJ0rQV
Robert B Chao, MD doctorRBC ( View Tweet)

#LB0002 #EULAR2022 An interesting study design looking at flare rate following withdrawal of drug after WK12. Phase 3 RCT of Baricitinib in paediatric JIA showed 76% met ACR30 at WK12. The Double-blind withdrawal showed less patients on Bari flared vs PBO (17% vs 51%) @RheumNow https://t.co/T0mSDkoHJo
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)
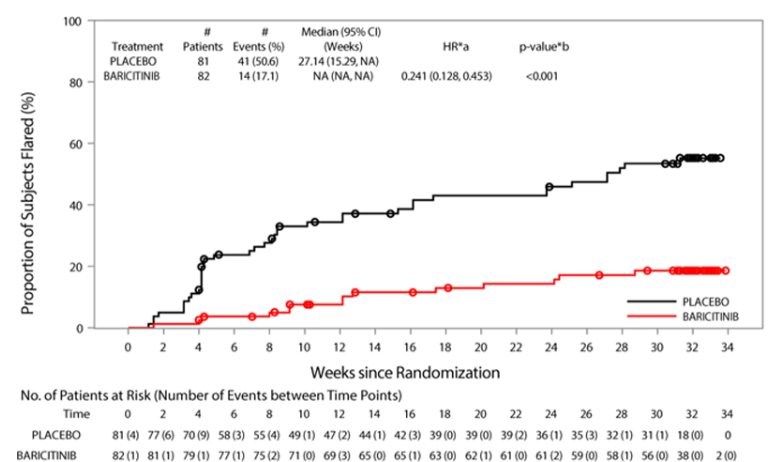
Ramanan et al Baricitinib in JIA. Double blind withdrawal study of prior responders. Earlier flare with PBO vs bari (HR 0.24 [95%CI 0.13,0.45], p<0.001. Less flares with bari vs PBO (17.1% vs 50.6%) p<0.001 @RheumNow #EULAR2022 LB0002 https://t.co/22rp70njRu https://t.co/LtJE9dz0GT
Links:
Richard Conway RichardPAConway ( View Tweet)
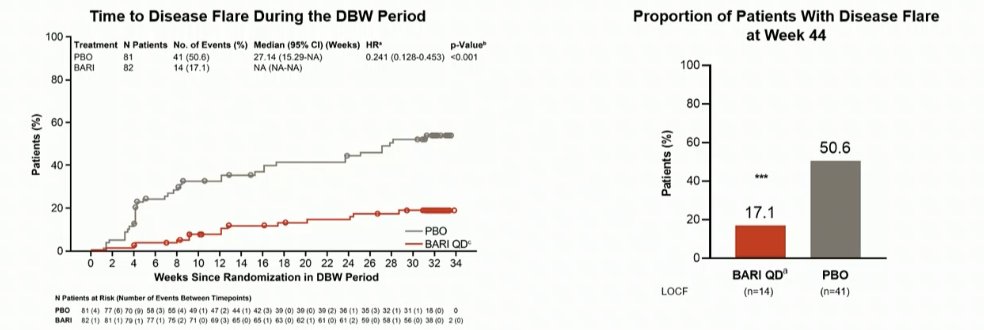
#EULAR2022 LB002
P3 Double-Blind W/d study: Baricitinib for JIA
⭐️All pts start in open label w good benefit (1/2 with ACR70)
⭐️PBO W/d led to 50% flare vs 17% in BARI group, flares earlier
@RheumNow https://t.co/apSSLqu1mp
Eric Dein ericdeinmd ( View Tweet)
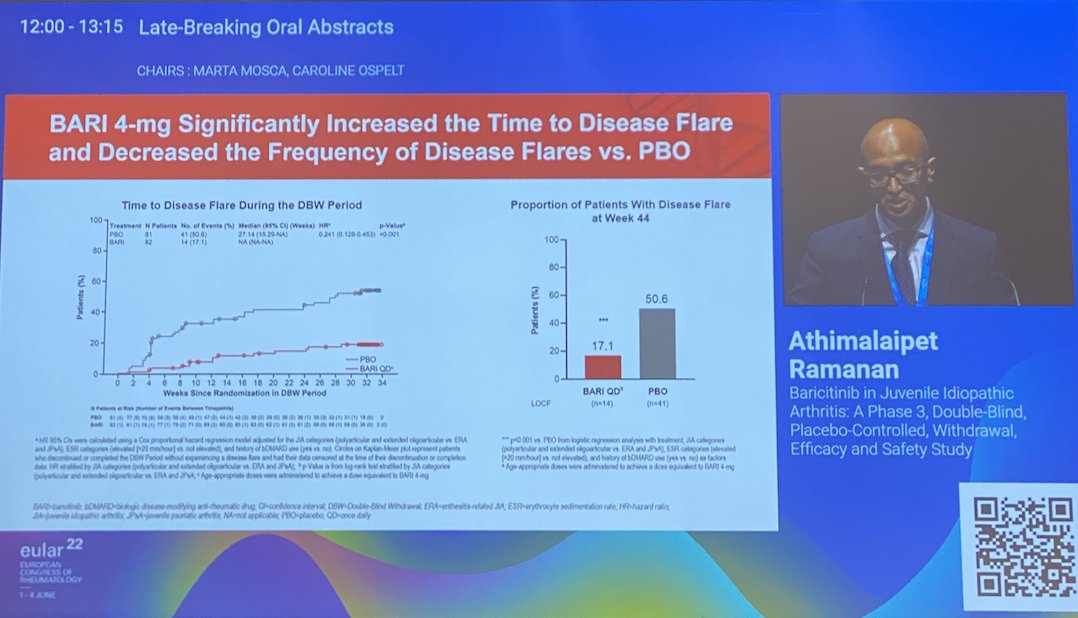
JUVE-BASIS Baricitinib in JIA
Phase 3 RCT wk12
Cs or BioDMARDs IR
⚡️Time to disease flare HR 0.24
⚡️JIA-ACR50% 63%
⚡️% flares 17% vs. 50% in PBO
Safety profile similar than adults
@RheumNow
#EULAR2022 LB0002 https://t.co/dX3DpB4QHT
Aurelie Najm AurelieRheumo ( View Tweet)
@EricFMorand up first in late-breakings
PAISLEY: deucravacitinib (TYK2i) ph2 in SLE - add-on, steroid wean
Good outcomes:
primary: SRI(4) at 32w
skin/joint/LLDAS & dsDNA/C4
Safety good (like PsO/PsA) but higher dose less so
Watch for: other clinical, ph3
#EULAR2022 @RheumNow https://t.co/XOQWuiHkTx
David Liew drdavidliew ( View Tweet)
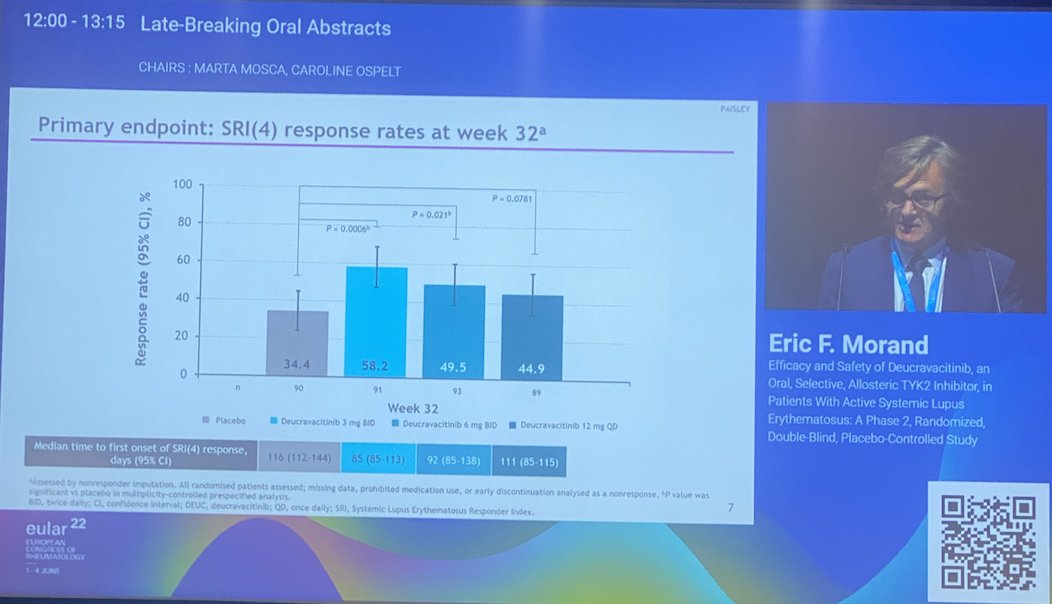
Wow! Phase 2 #deucravacitinib data for #SLE met primary & secondary endpoints: SRI(4) response, BICLA, LLDAS, CLASI-50. AEs include: skin related events and UTIs but no increase in SIE, HZ, MACE/VTE @bmsnews LB0004 #EULAR022 @rheumnow https://t.co/bsKI0mixVT
TheDaoIndex KDAO2011 ( View Tweet)
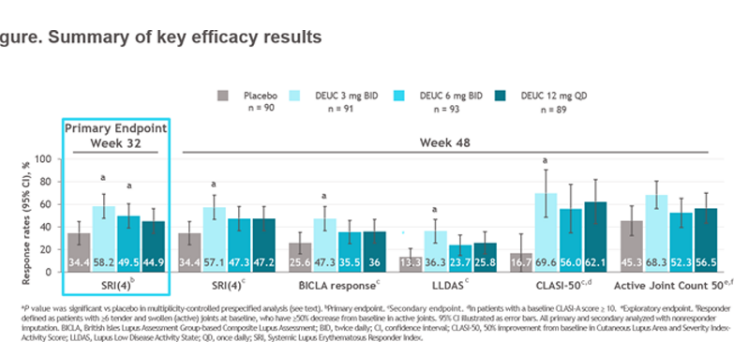
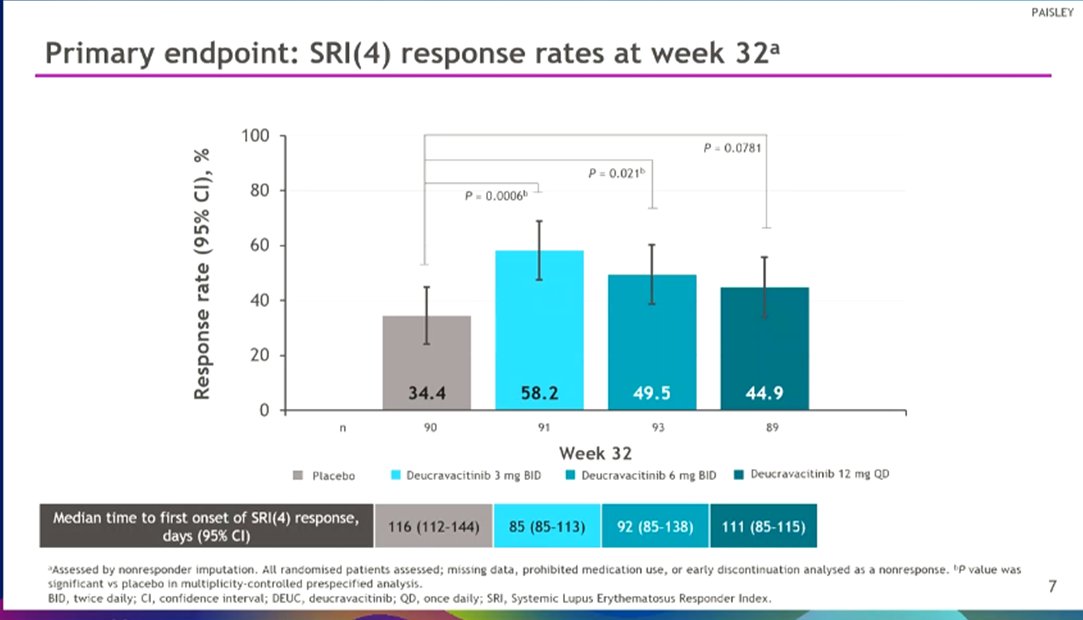
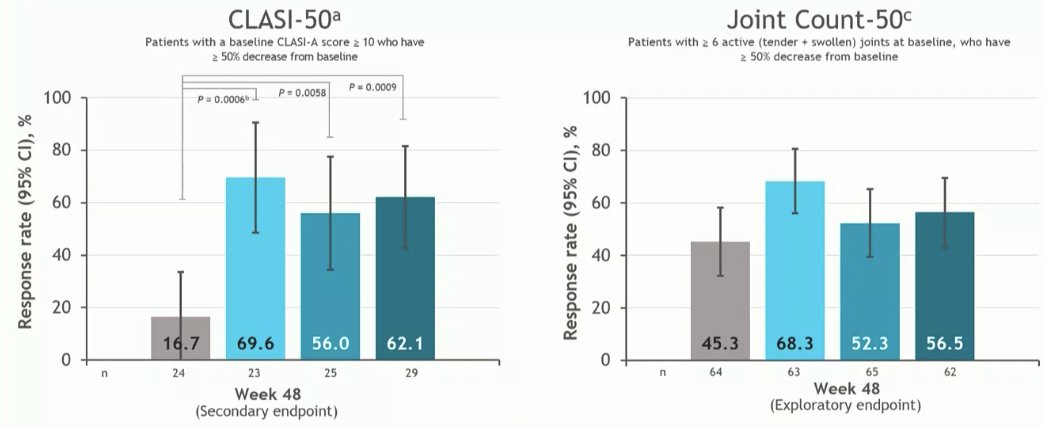
Morand et al Tyk2i deucravacitinib in SLE. 48-week phase 2 RCT. SRI(4) PBO: 34.4%; DEUC 3 mg BID: 58.2%, P =0.0006; DEUC 6 mg BID: 49.5%, P =0.021; DEUC 12 mg QD: 44.9%, P =0.078. BICLA, LLDAS, CLASI-50, active joint count also+ @RheumNow #EULAR2022 LB0004 https://t.co/MBPc9xQFvG https://t.co/n3GUwl0RtD
Links:
Richard Conway RichardPAConway ( View Tweet)
PAISLEY LB004
@EricFMorand on deucravacitinib phase 2 for SLE
⭐️Meets primary endpt: SRI(4)
⭐️Secondary endpts: BICLA, LLD, CLASI, jt count, biomarkers improved
⭐️Safety data wo VTE, CVD events
Earlier at #EULAR2022, BRAVE trial phase 3 baricitinib: No benefit in SLE
@RheumNow https://t.co/3YpPSJcenF
Eric Dein ericdeinmd ( View Tweet)
PAISLEY study Phase 2 RCT
Deucravacitinib in SLE meets primary endpoint wk 32
Results for dose 3mg BID:
*SRI(4) response 58.2% vs.
PBO 34%
*LLDAS 36%
*CLASI 70%
*⬇️ SJC
No new safety signal
Now waiting for Phase 3 👀
@RheumNow LB0004 #EULAR2022 #Lupus https://t.co/2KYSvfq4h7
Aurelie Najm AurelieRheumo ( View Tweet)
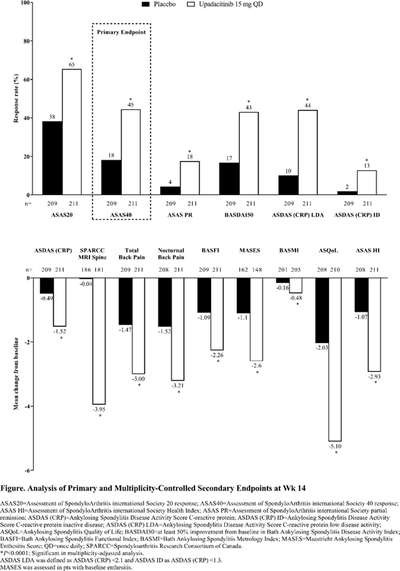
New in AS. Upadacitinib 15mg od was significantly more effective than placebo over 14 weeks in AS bDMARD-IR (ASAS40 45% vs 18% and the onset of effect seen by week 4), no new safety risks identified in the SELECT-AXIS 2 study by Van de Heijde et al #EULAR2022 @RheumNow POS0306 https://t.co/LCQBf38GWe
Dr. Antoni Chan synovialjoints ( View Tweet)
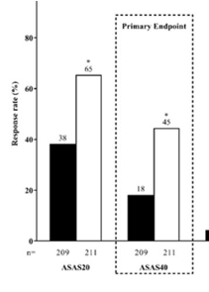
SELECT-AXIS 1 trial of upadacitinib in active #AS with an inadequate resp to prior biologic - pivotal phase 2/3 RCT of 420 AS pts (Dz dur 7.7 yrs; 83% B27+) showed better ASAS40 at wk 14 w/ UPA vs PBO (45% vs 18%; P<0.0001) #EULAR2022 POS0306 https://t.co/6ZBEvjRjqc https://t.co/GL0vlB34mE
Links:
Dr. John Cush RheumNow ( View Tweet)

Please see my short video on Promising Safety Outlook for Rituximab and Baricitinib in Vaccinated Patients #EULAR2022 @RheumNow https://t.co/bqzdWIzoOg
Md Yuzaiful Md Yusof Yuz6Yusof ( View Tweet)
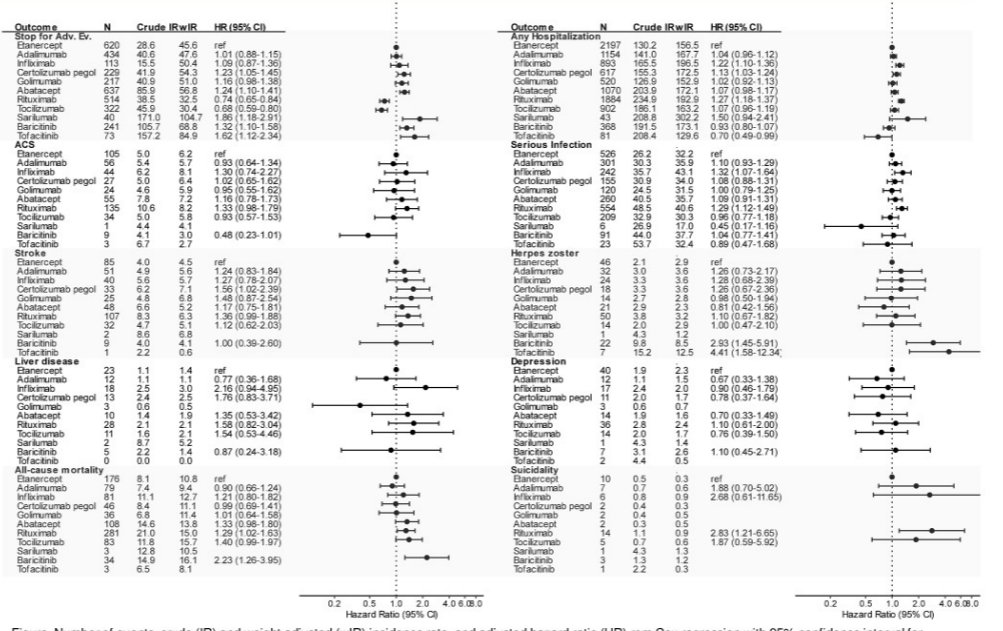
Frisell et al. Safety b/tsDMARDs from 10 years ARTIS. There is a mass of interesting data here, look at that figure! Higher rates of discontinuation due to AEs for tofa, bari, sari, rituximab. May be explained by chanelling and residual confounding @RheumNow #EULAR2022 POS0637 https://t.co/KrspU6c9Cs
Richard Conway RichardPAConway ( View Tweet)

What drives residual pain improvement in #JAKi treated pts? Dunno but #Baricitinib and #Sarilimab showed better pain decrease vs placebo and #Adalimumab. #OP0052 showed both #Tofacitinib & Adalimumab reduced pain more than placebo in PsA & RA if in remission @RheumNow @eular_org
Janet Pope Janetbirdope ( View Tweet)
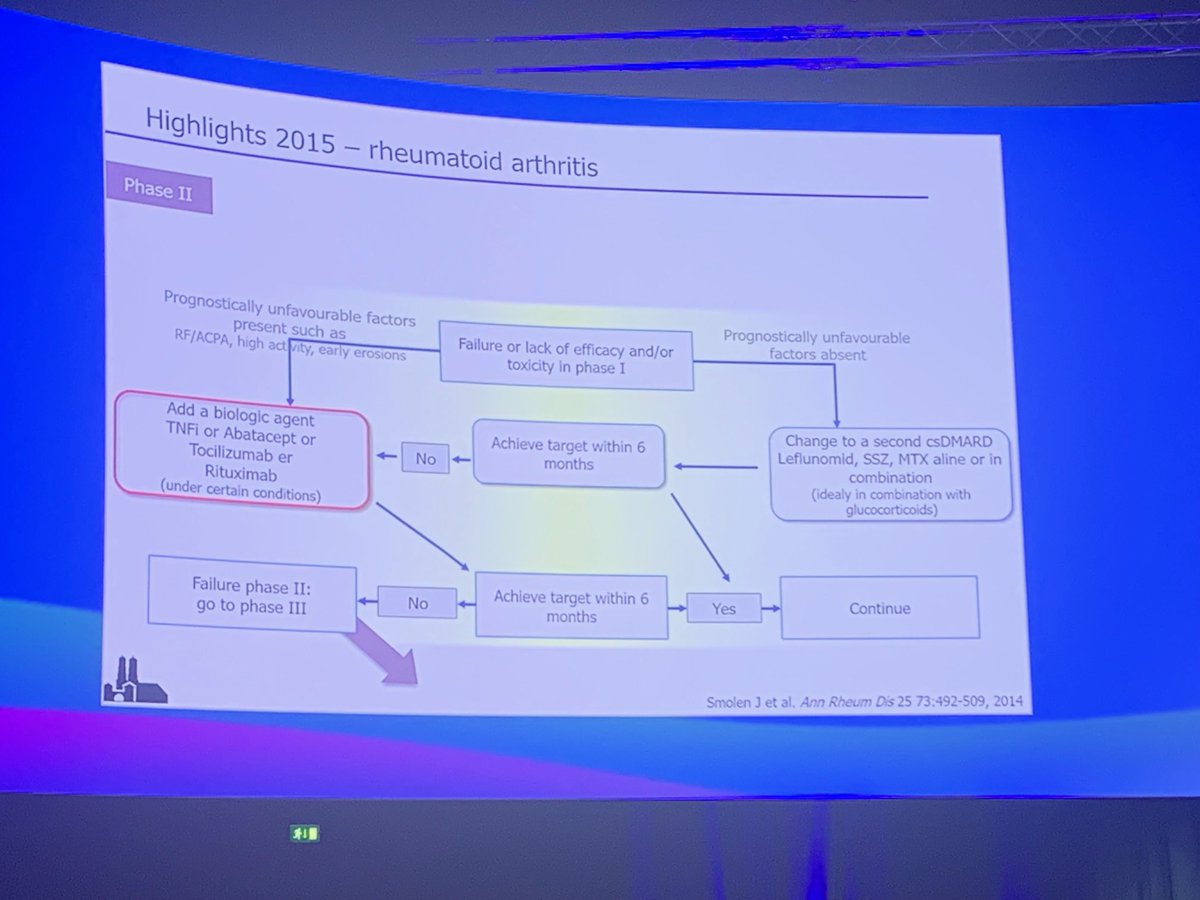
Are JAKi fulfilling their promises in RA? H Schulz-Koops said a survey showed rheumatologists wanted effective fast acting Rx that could be used as monotherapy. @eular_org #EULAR2022 @RheumNow https://t.co/Gdel9YnAzA
Janet Pope Janetbirdope ( View Tweet)
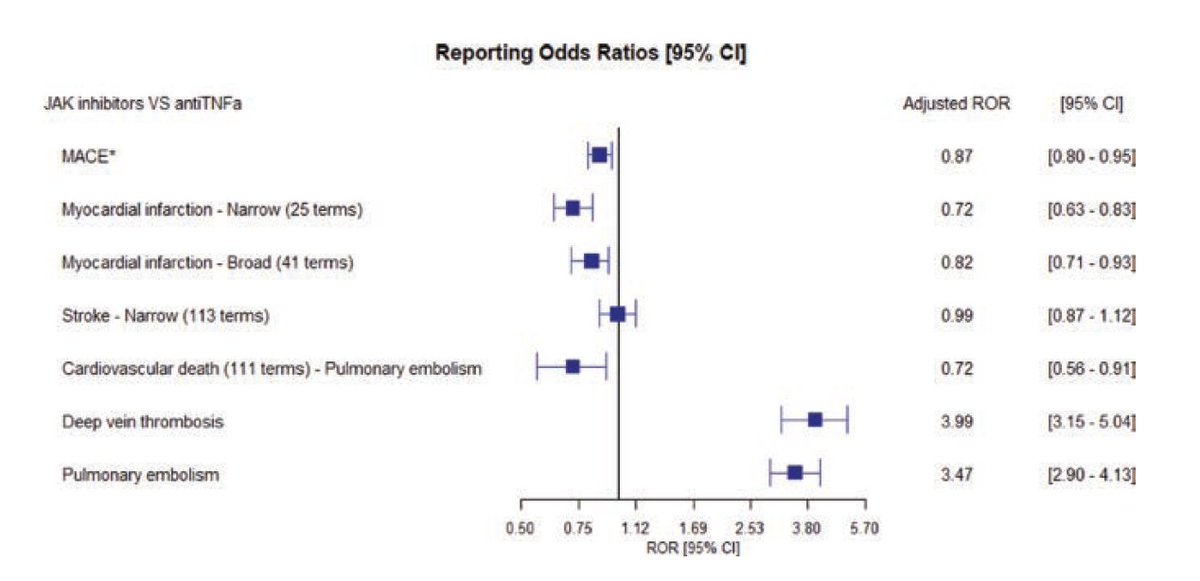
VIGIBASE registry RA
39000+ pts JAKi and 231000+ pts TNFi
*No increase in MACEs with JAKi 1.4% vs. 0.9%
*JAKi Increase in DVT RR 3.99
and PE RR 3.5
adjusted on age and sex
@RheumNow #OP0268 #EULAR2022 #Lupus https://t.co/hiAvKctgky
Aurelie Najm AurelieRheumo ( View Tweet)

Some / most of promises of #JAKi are fulfilled. Efficacy, adherence = on mono vs combination Rx with MTX. But some emerging safety issues. NK cells reduce inflammation of NK cells on fibroblasts re other MOA. @eular #EULAR2022 @RheumNow The promise of #JAKi I RA https://t.co/8Ry3vVMyzO
Janet Pope Janetbirdope ( View Tweet)
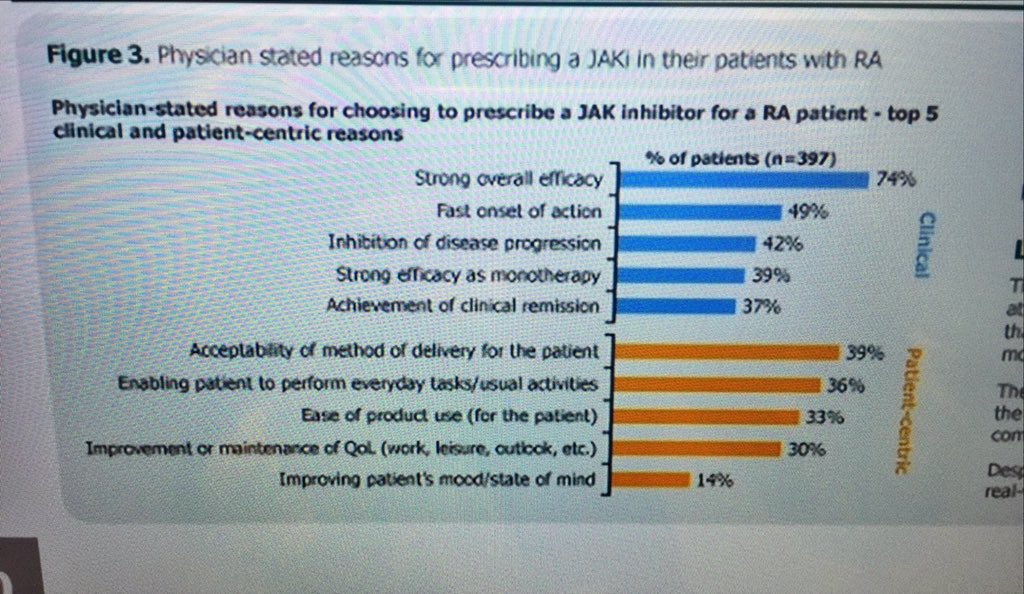
#ClinicalPearl. Why do doctors prescribe #JAKi in RA? Efficacy, fast onset, X-ray inhibition, can work as monotherapy, remission - in order of diminishing frequency BUT I will add access and oral (convenience). POS0680 @RheumNow @eular_org #EULAR2022 https://t.co/XHtNqguqvh
Janet Pope Janetbirdope ( View Tweet)
