All News
FDA Approves Icotrokinra for Plaque Psoriasis
Today the FDA approved an oral IL-23 inhibitor, icotrokinra (Icotyde), for use in moderate-to-severe plaque psoriasis in adults and children 12 years of age and older (who weigh at least 40 kg) who are candidates for systemic therapy or phototherapy.
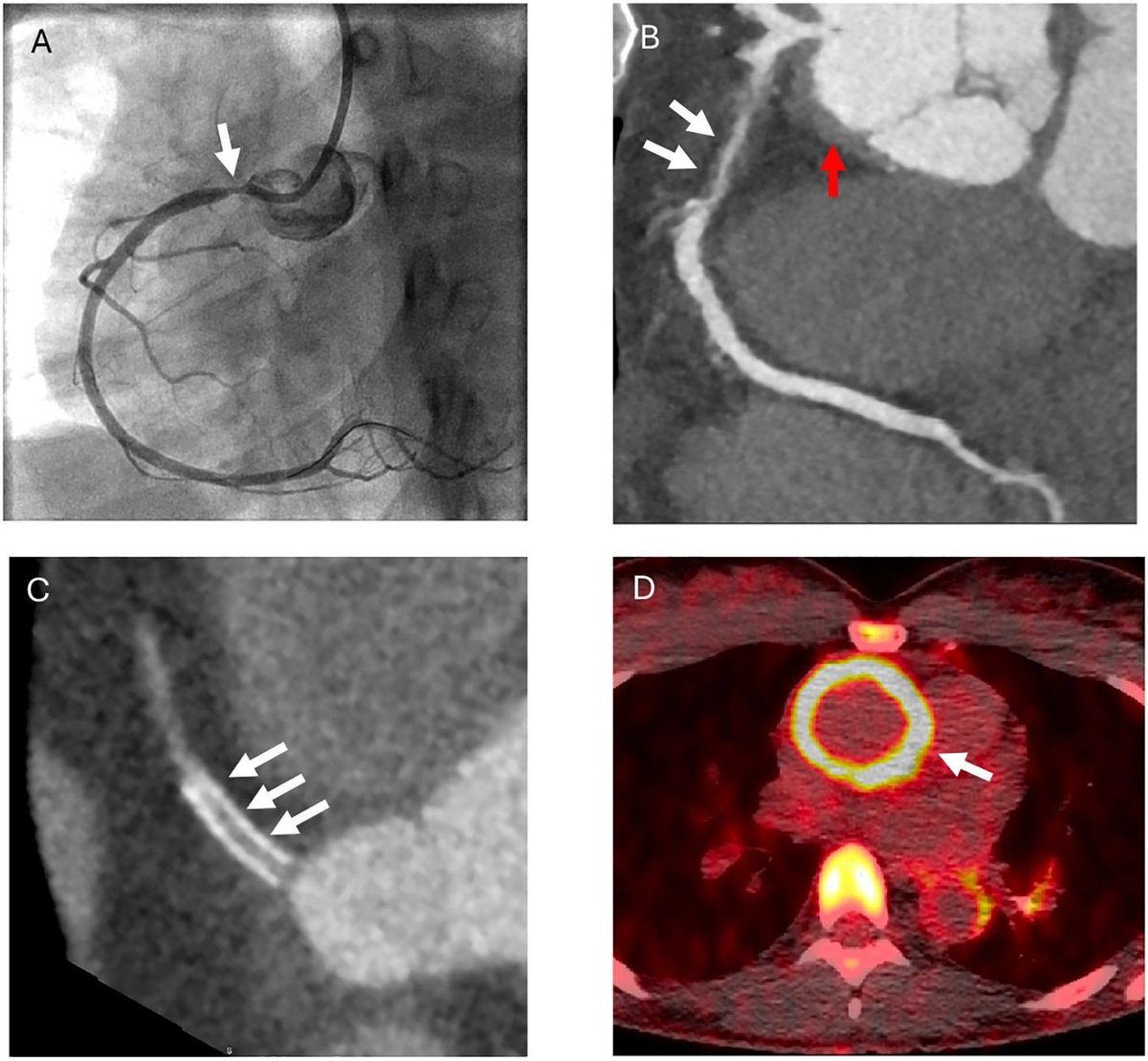
Read ArticleHydralazine and the Rare Risk of Vasculitis
Since the 1980s, hydralazine has been a notable cause of drug-induced lupus, and inferred to cause vasculitis as well.
2025 Update: EULAR Recommendations on Rheumatoid Arthritis Management
The European Alliance of Associations for Rheumatology (EULAR) has updated their rheumatoid arthritis (RA) management recommendations, notably with fewer recommendations (total of 9, down from 11 in 2022 version), by merging and removing previous recommendations.
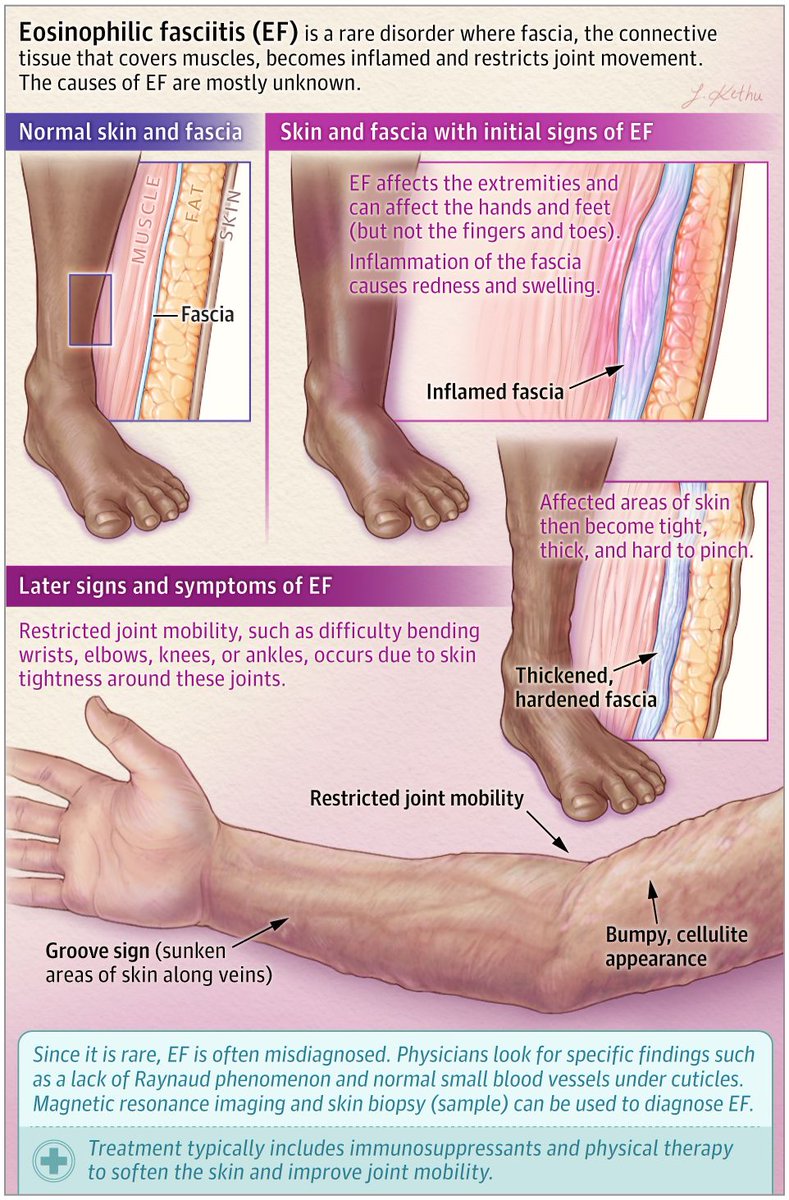
Read ArticlePitfalls of autoimmune serologic testing
Misuse of laboratory testing may lead to misdiagnoses and mismanagement; this is particulary prevalent when clinicians consider the use of the diagnostic use of serological autoimmune markers.
Read ArticleWorldwide Trends in Hyperuricemia
A Lancet Rheumatology systematic review shows the prevalence of hyperuricaemia has risen markedly over the past two decades in both men and women, owing to population growth, aging, and increasing age-specific rates
Read Article


Dr. John Cush RheumNow ( View Tweet)


Dr. John Cush RheumNow ( View Tweet)
Links:



Links:

Links: