All News
ACR 2023 – Day 4 Report
The last day at ACR23 was a ghost town as most left, largely because there was limited programming on the fourth and final day. The main attraction on Day 4 was the late-breaking abstract – oral presentations. Here’s a list of the late breakers; below, I provide commentary on the ones that caught my attention.
Read ArticleCanadian Consensus on MSK Ultrasound
Dr. Jack Cush discusses abstract 0726 presented at the 2023 ACR Convergence meeting in San Diego, CA.
Read ArticlePMR & Popeye's Spinach (10.6.2023)
Dr. Jack Cush reviews the big PMR reports from this week along with other journal reports from RheumNow.com.
Read ArticleGout Flare Prophylaxis - Colchicine or NSAIDs?
Flare prophylaxis is the standard when initiating urate-lowering therapy (ULT) in patients with gout. A new study suggests that colchicine, more so than non-steroidal anti-inflammatory drug (NSAID) prophylaxis, may increase the odds of adverse events, when initiating allopurinol for gout.
Read ArticleRheumNow Podcast – with Special Guest Dr. Kathryn Dao (9.29.2023)
This week on the podcast, Dr. Jack Cush is joined by special guest, Dr. Kathryn Dao to review the news and journal reports from the past week on RheumNow.com. They discuss unmet needs in gout, obesity and CV disease in PsA and should you check ANCA antibodies in SLE?
Read ArticleWhat Changed FDA's Mind About Ilaris for Gout?
The FDA quietly approved the interleukin (IL)-1 inhibitor canakinumab (Ilaris) for gout 10 days ago, with no announcement from either the agency or the drug's manufacturer, Novartis -- leaving outsiders to guess at what changed in the dozen years since the company's first attempt to gain app
Read ArticleRheumNow Podcast – with Special Guest Dr. Janet Pope (9.8.2023)
This week, Dr. Jack Cush reviews the news with special guest, Dr. Janet Pope. They discuss "Not-non-Inferior", large scale genetic screening, Zoster risk w/ newer lupus drugs, tapering, RA referral problems and the TNR webinar session on "ORAL Surveillance Revisited".
Read ArticleHard Decisions in RA (9.1.2023)
Dr. Jack Cush reviews the news and journal reports from this past week on RheumNow, and introduces our September campaign - "Hard Decisions in RA".
Read ArticleFDA Approves Canakinumab for Gout Flares
The US Food and Drug Administration (FDA) has approved canakinumab (Ilaris) for the treatment of gout flares in adults who cannot be treated with NSAIDs, colchicine, or repeated courses of corticosteroids. The drug is also indicated for people who could not tolerate or had an inadequate response to NSAIDs or colchicine.
Read Article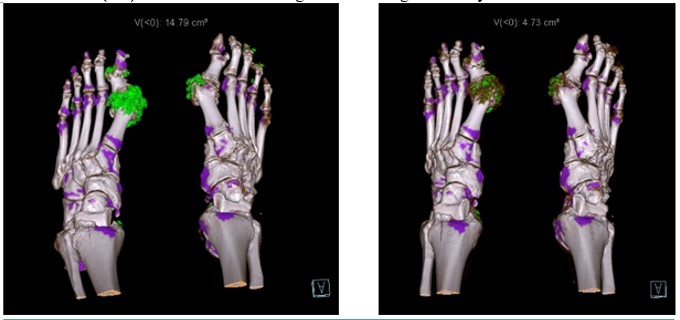
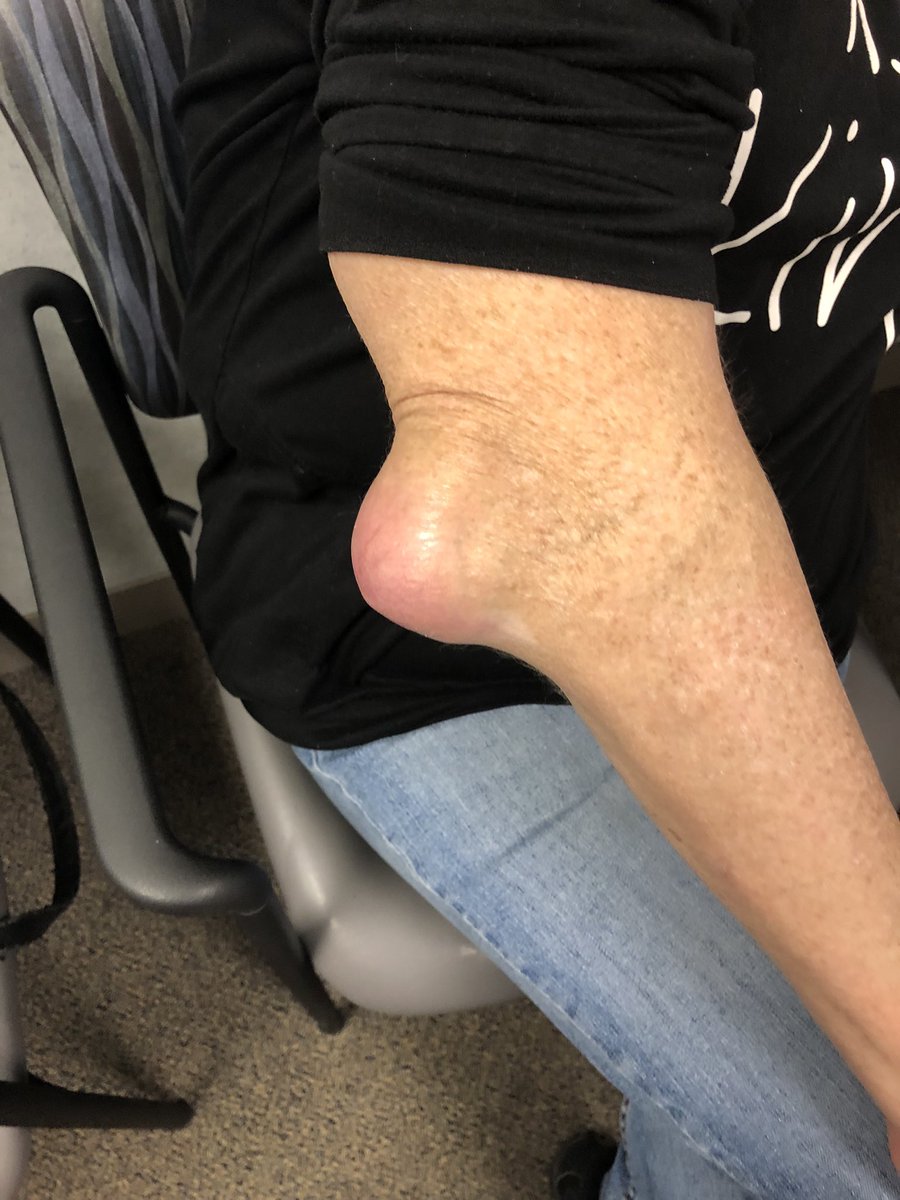
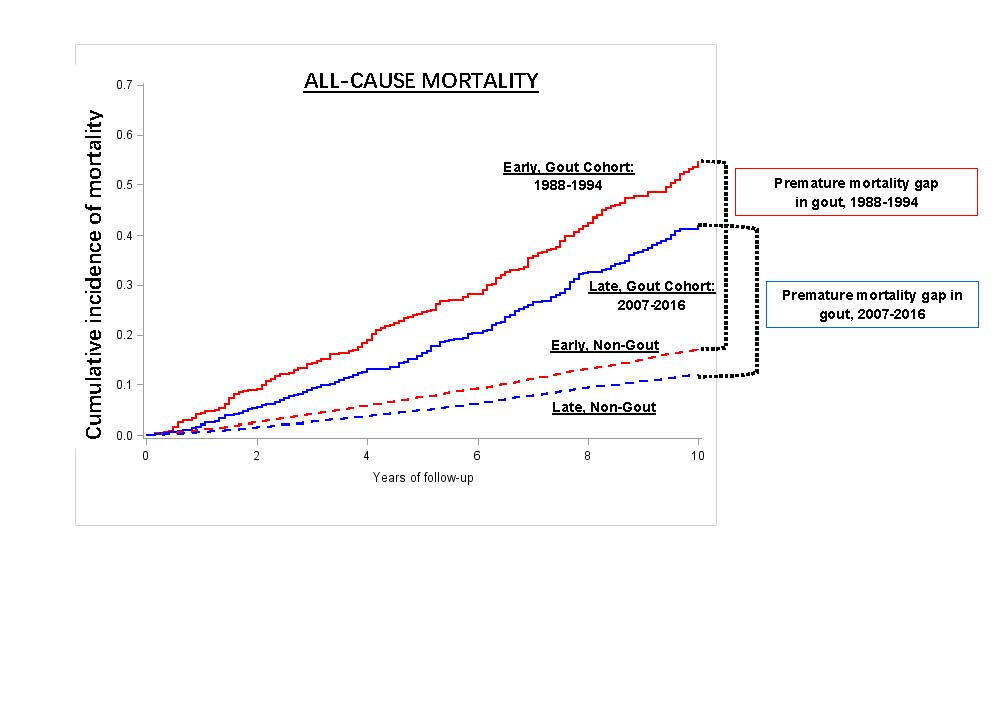
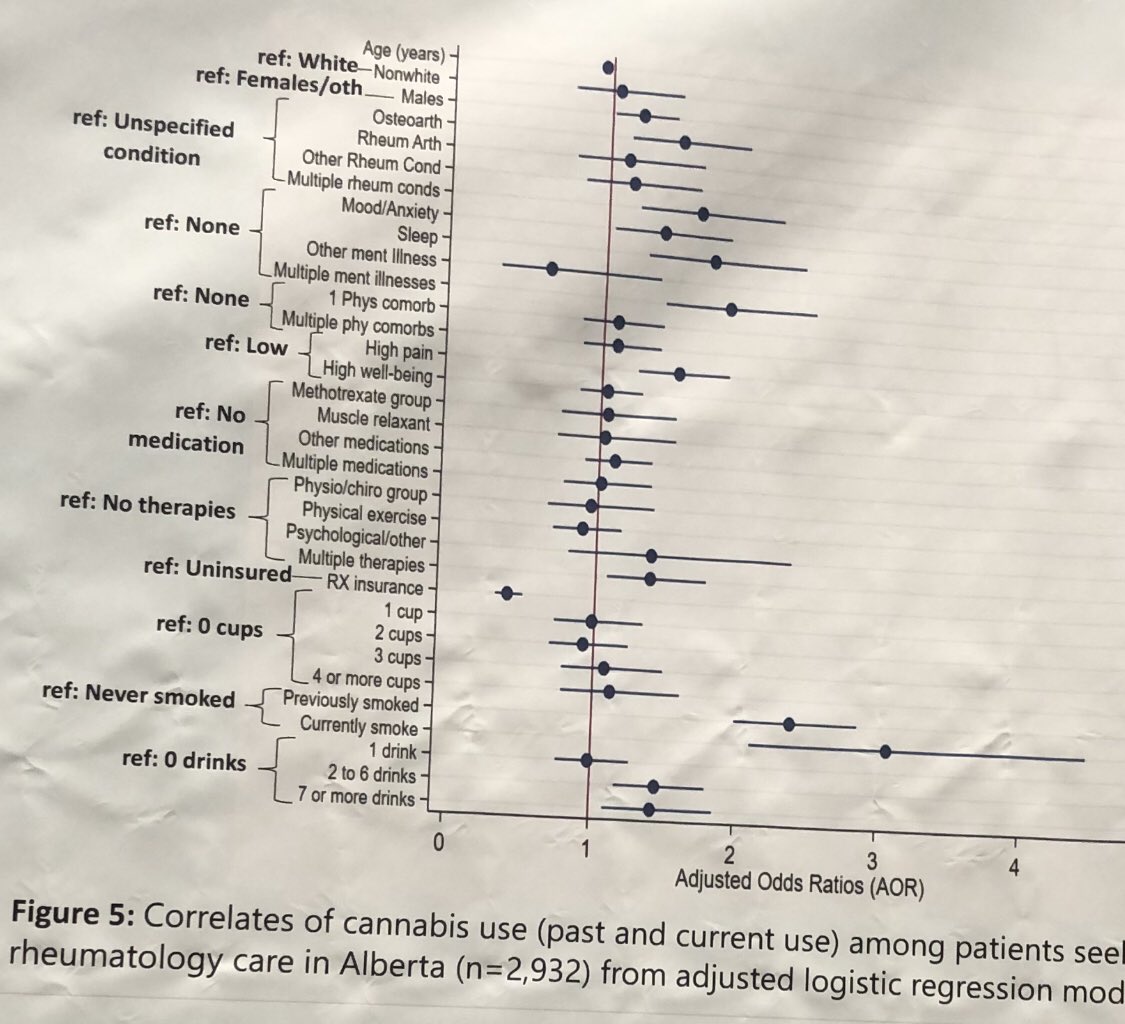
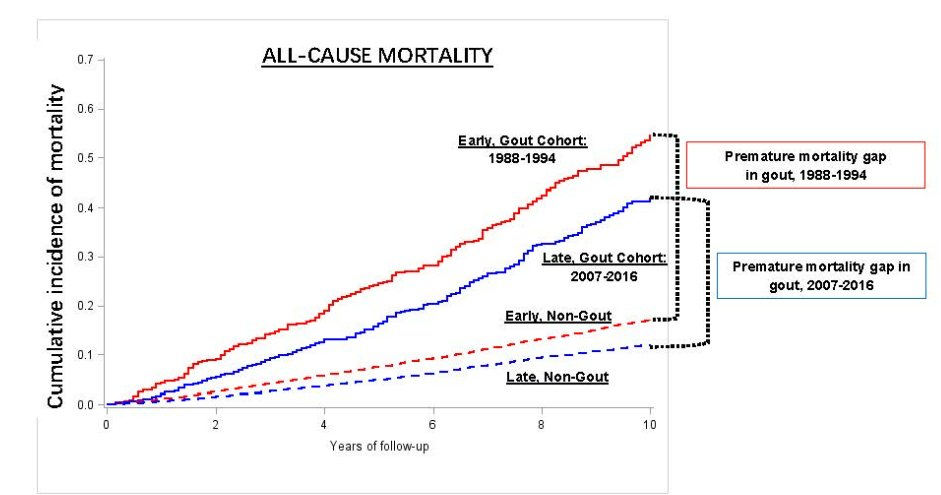
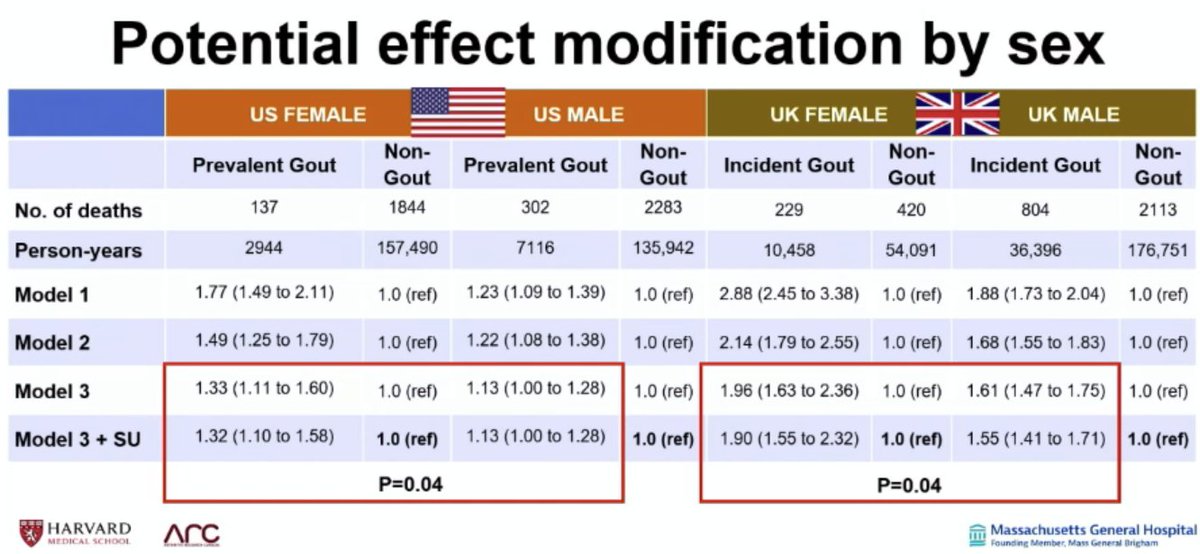

Dr Gurdeep S Dulay ( View Tweet)


Links: