All News
Opening Day Report
The opening of ACR2 Convergence was a hit for all who signed up and viewed in. The day included the presidential address by outgoing president Dr. David Karp (UT Southwestern) and a keynote talk and interview with Dr. Seema Yasmin (Stanford).
Read ArticleEmerging Therapies in Spondyloarthritis: A Promising Pipeline
Growing awareness of spondyloarthropathies over the last two decades has led to a better understanding of the pathophysiology of spondyloarthritis, and subsequently increased interest in more distinct, disease state specific treatment options.
Read ArticleOutcomes of COVID-19 in Patients with Rheumatoid Arthritis
Patients with rheumatic diseases – requiring lifelong immunosuppressants— are at high risk for respiratory and viral infections. Over the past decade, an armamentarium of biologic and targeted therapies has led to better control of disease activity in patients with rheumatoid arthritis. Whether these patients, especially those receiving newer biological and targeted therapies such as JAK inhibitors, are at an increased risk of severe COVID-19 outcomes remains uncertain.
Read ArticleRheumNow Podcast – ACR Convergence Begins (11.5.2021)
Dr. Cush reviews the news, and kicks off ACR21 learning and how to take in the annual meeting.
Read ArticleProlonging the Controversy of Hydroxychloroquine
Since the start of the pandemic, hydroxychloroquine (HCQ) was forced into the limelight for the prevention/treatment of COVID-19.
Read ArticleTreat-to-Target Works in Gout With No Cardiac Toxicity
New research presented this week at ACR Convergence, the American College of Rheumatology’s annual meeting, shows that allopurinol and febuxostat may effectively lower urate levels when used in a treat-to-target approach. Importantly, both urate-lowering therapies were very effective with 90% of patients reaching target urate levels. Additionally, both appeared safe, with no evidence of increased cardiovascular toxicity (Abstract #1900).
Read ArticleNOR-DRUM Study: TNF Inhibitor Therapeutic Drug Monitoring is Effective
New research presented this week at ACR Convergence, the American College of Rheumatology’s annual meeting, shows that proactive therapeutic drug monitoring, a newer treatment strategy where a patient’s drug serum levels are regularly assessed to adjust the dose and intervals, controlled disease more effectively than standard therapy with infliximab, a tumor necrosis factor (TNF) inhibitor.
Read ArticleInfection Rates in Psoriatic Arthritis Patients on Biologics Have Decreased
New research presented this week at ACR Convergence, the American College of Rheumatology’s annual meeting, shows significant decreases in infections among people with psoriatic arthritis over the years 2012-2017.
Read Article
Links:
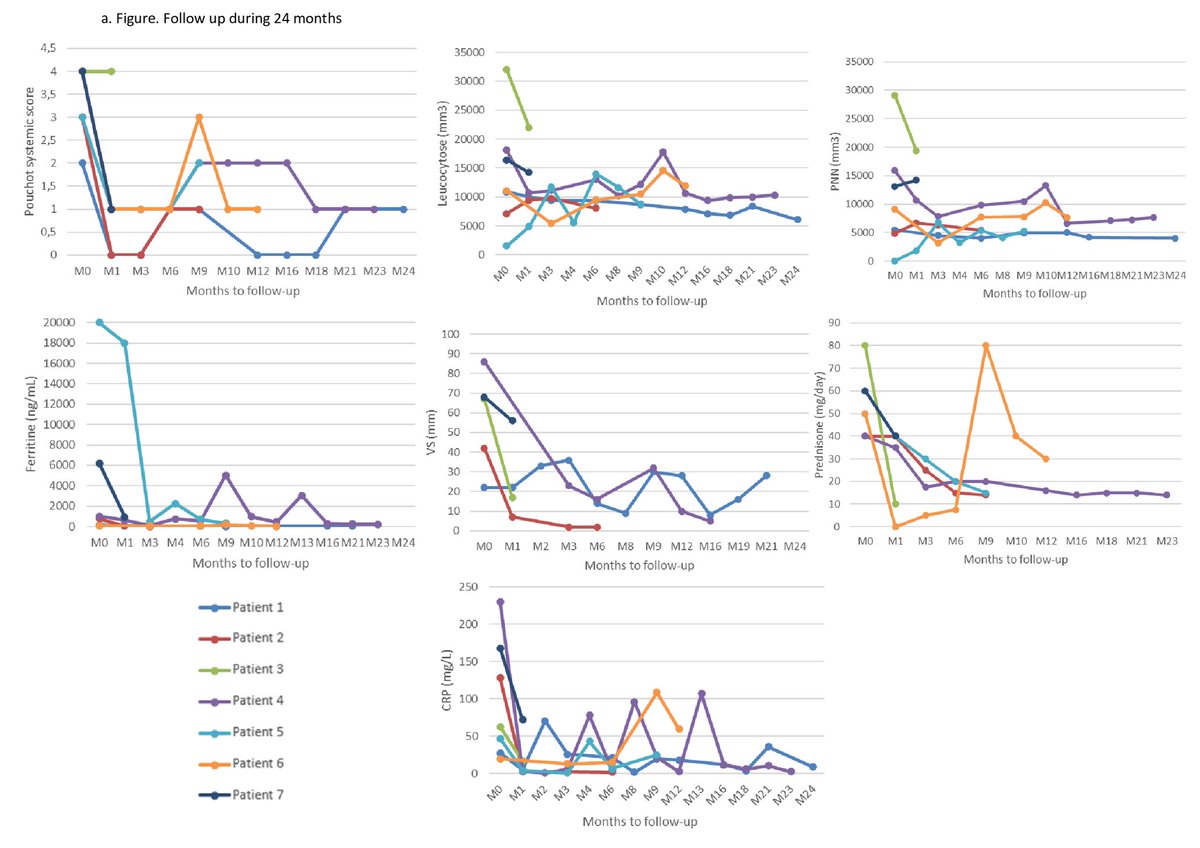


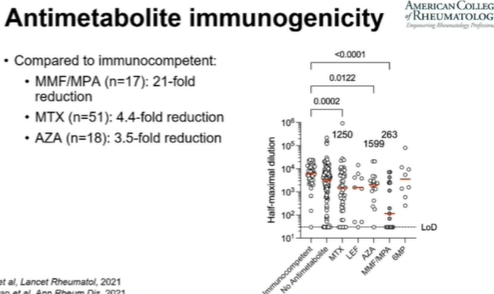
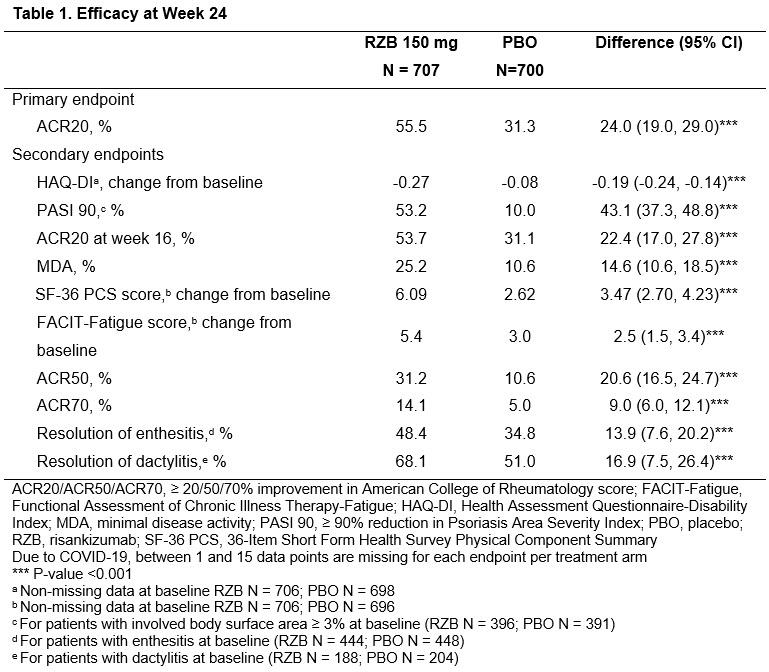


TheDaoIndex KDAO2011 ( View Tweet)


TheDaoIndex KDAO2011 ( View Tweet)