All News
Are we really treating Psoriatic disease if we only address active disease domains and T2T?
It’s widely accepted that Psoriatic disease has multi-system, multi-domain potential that can lead to life altering complications if left untreated or undertreated. Rheumatologists have shifted the way we discuss treatment options and disease state expectations with patients. Two #ACR22 abstracts stood out to me regarding the Psoriatic disease patient experience and will, ultimately, add to what I do in clinic.
Read ArticleDon’t be ‘ticked’: TYK2 is not a JAK!
Recently the FDA approved deucravacitinib, a highly selective TYK2 inhibitor for psoriasis. Trials are positive in psoriatic arthritis and a phase II study in SLE. What about the effects? Presentations from the ACR22 meeting may provide answers.
Read Article“Hey PRESTO!” – Early identification of psoriatic arthritis
Despite a wealth of treatment options, a number of notable unmet needs remain in PsA, most notably the significant diagnostic delay that many patients endure. We are all aware that those with psoriatic skin disease are the most at risk cohort for PsA. Identifying patients with psoriasis who have, or are at increased risk of PsA, appears to be the low hanging fruit that we just can’t fully seem to grasp.
Read ArticleIndustry Abstract Previews of ACR 2022
The pharmaceutical companies have will showcase their featured clinical trials and abstracts at ACR 2022. These are their best studies for you to review and evaluate as part of your to-do list.
Read Article2022 EULAR Recommendations for Screening and Prophylaxis of Chronic and Opportunistic Infections
A EULAR Task force has developed recommendations for screening and prophylaxis of chronic and opportunistic infections in patients with autoimmune inflammatory rheumatic diseases (AIIRD).
Read ArticleWomen with Rheumatic Disease (11.4.2022)
Dr. Jack Cush reviews the news and journal articles from this past week on RheumNow.com. New regulatory decisions about JAK inhibitors and why do women with rheumatic diseases have unfair outcomes?
Read ArticleEMA Crack Down on JAK Inhibitors
The European Medicines Agency’s (EMA's) safety committee (PRAC) has issued a recommendation that the use of oral JAK inhibitors (tofacitinib, baricitinib, upadacitinib and filgotinib) be limited to certain at-risk patients unless no other suitable treatment alternatives are available.
Read Article
Patricia Harkins DrTrishHarkins ( View Tweet)

Catherine Sims, MD DrCassySims ( View Tweet)
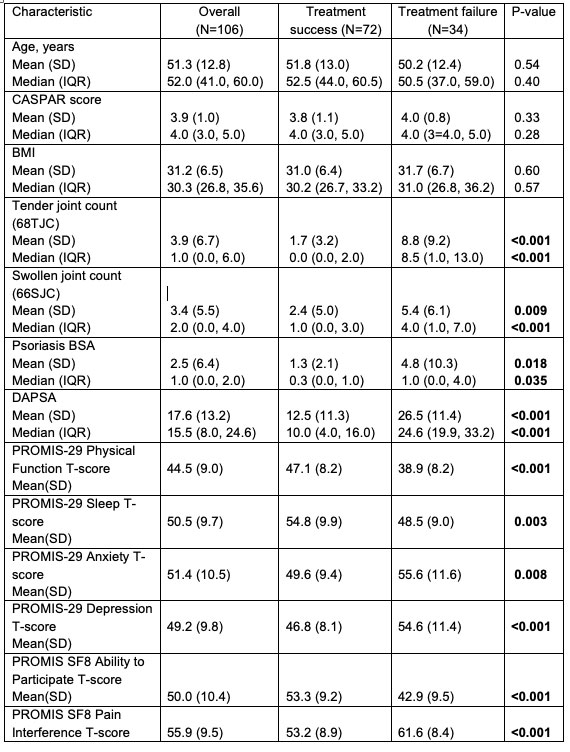
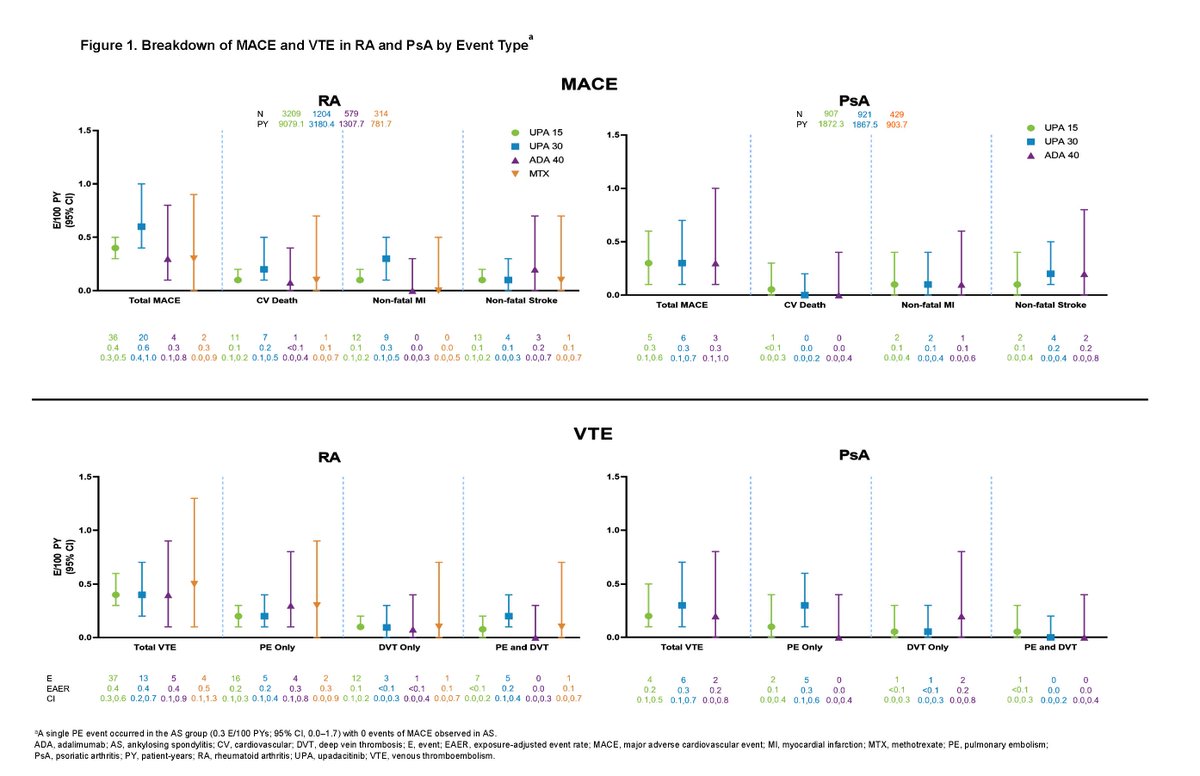
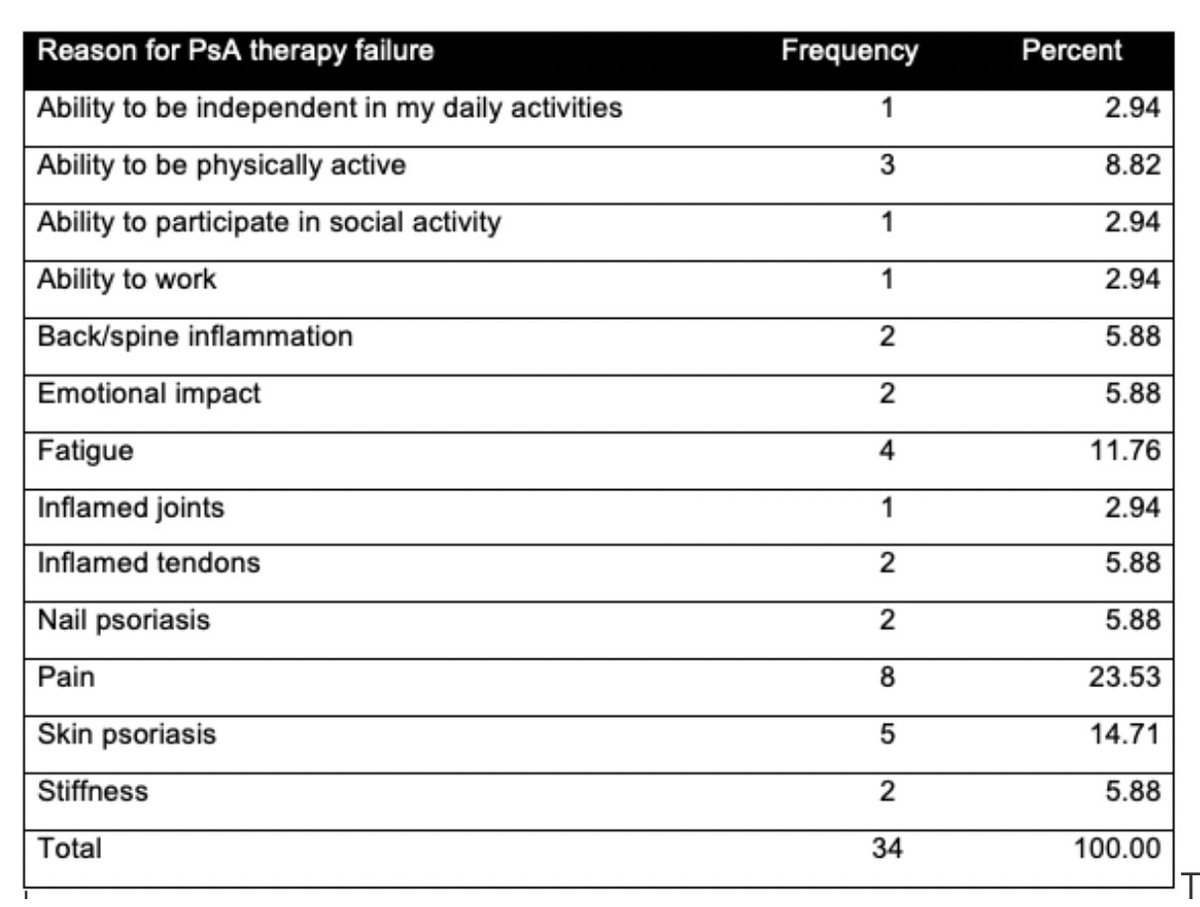
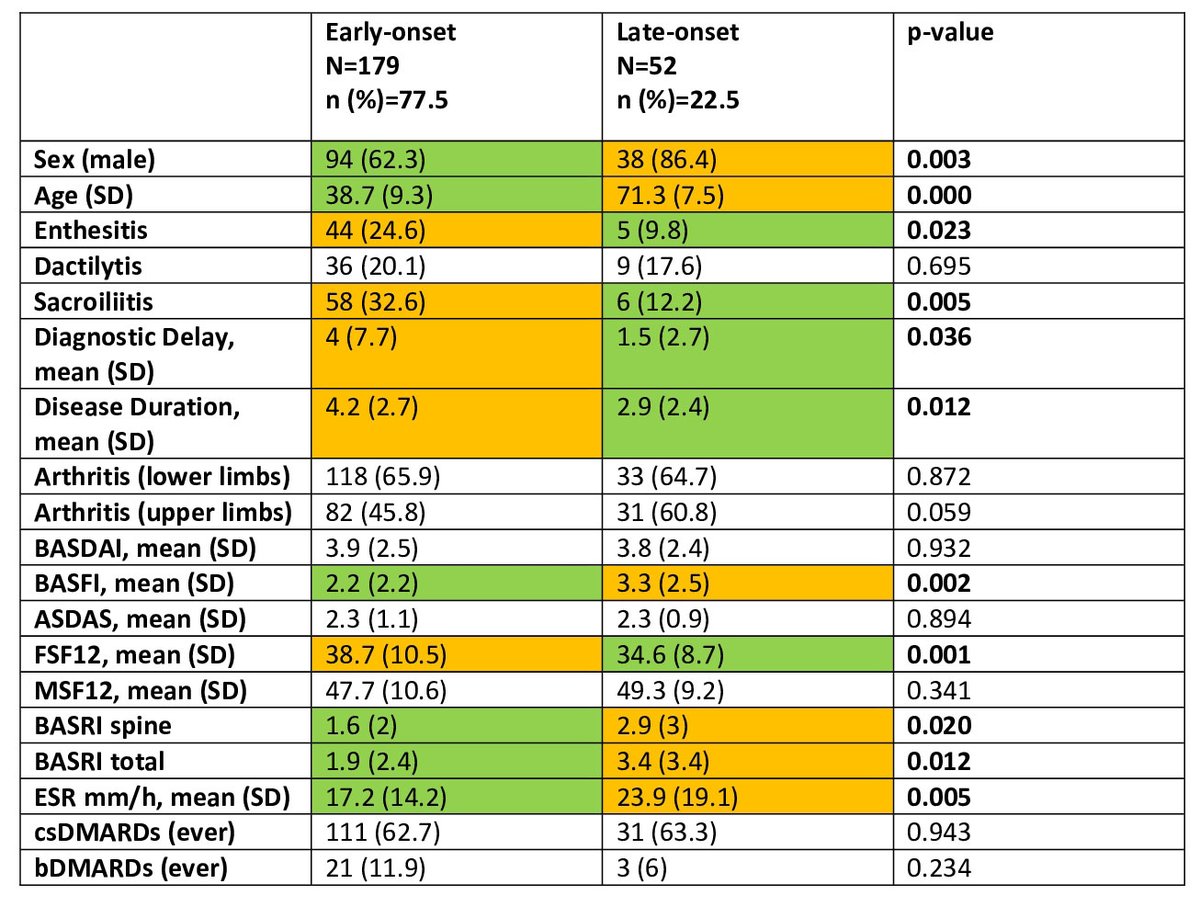
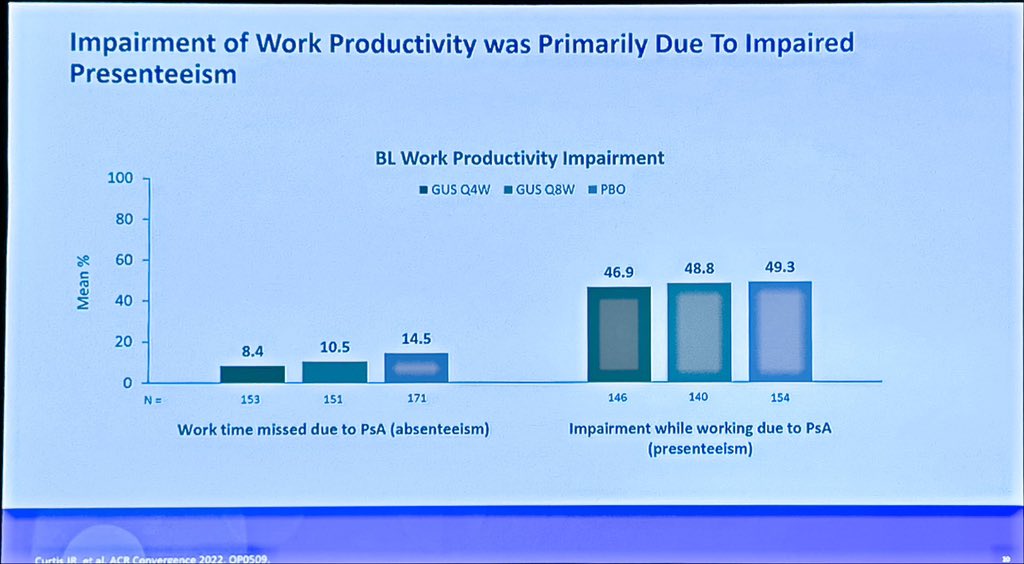

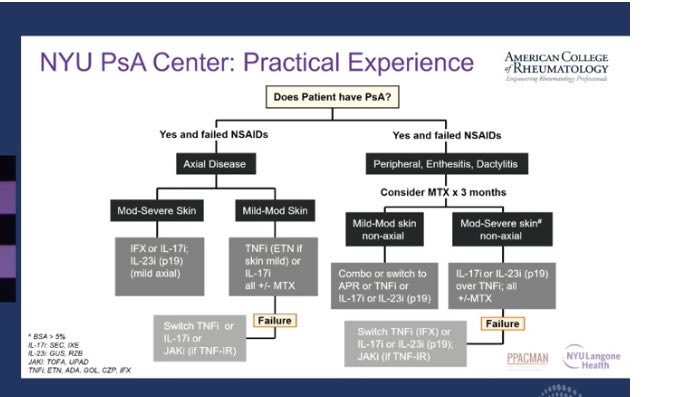

Catherine Sims, MD DrCassySims ( View Tweet)
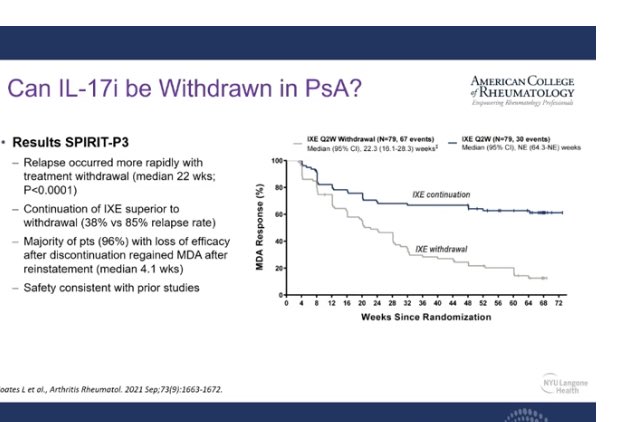
Links: