All News
Curbside Consults - January 2017
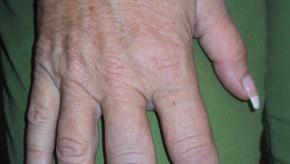
Curbside Consults are cases submitted by our rheumatology colleagues as challenging therapeutic or safety issues. Answers are based on experience, with added evidence from the medical literature and published guidelines.
Read ArticleThe RheumNow Week in Review – 27 January 2017
Dr. Cush reviews highlights from the past week on RheumNow.com.
Read ArticleDSB - Safety Update and Drug Shortages January 2017
RheumNow will periodically report safety issues as Drug Safety Bulletins. These will update you with safety issues, label changes and reports of new, ongoing and resolved drug shortages that affect rheumatology. If you have suggestions or information about specific drug shortages or drug safety issues, please email us at info@rheumnow.com.
Read ArticleFDA Issues Draft Guidance on Biosimilar Interchangeability
Interchangeability, or the substitution of a biosimilar for its innovator biologic, is on the horizon. Will the prescriber chose the biosimilar in place of the originator? Or will this allow pharmacists or healthcare plans to substitute a biosimilar, without intervention from a healthcare provider?
Read ArticleThe RheumNow Week in Review – 20 January 2017
Dr. Jack Cush reviews highlights from the past week on RheumNow.
Read ArticleThe RheumNow Week in Review – 13 January 2017
Dr. Jack Cush reviews highlights from this past week on RheumNow.com:
Read ArticleThe RheumNow Week in Review – 6 January 2017
Dr. Jack Cush reviews highlights from the first week of 2017 on RheumNow.com.
Happy New Year!
Read ArticlePregnancy Outcomes in Psoriatic Arthritis
Disease Activity Pre-pregnancy During Pregnancy Post-pregnancy
Mild 10 6 4
Moderate 2 6 8
Severe 2 2 2
CHMP Recommend Baricitinib for Approval in Europe
Eli Lilly and Company announced last friday that the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) has issued a positive opinion, recommending the approval of baricitinib for the treatment of adults with moderate to severely active rheumatoid arthritis (R
Read ArticleRisk of GI Perforations on Biologics
Gastrointestinal perforation is a rare but serious complication that RA patients may be at particular risk for.
Read ArticleRheumNow Week in Review – 16 December 2016
Dr. Jack Cush reviews highlights from this week on RheumNow.com
Read ArticleEULAR 2016 Recommendations on Early Arthritis
Combe and leading European rheumatologist have published an update to the 2007 European League Against Rheumatism (EULAR) recommendations for management of early arthritis.
Read ArticleSupreme Court Declines Biosimilar Patent Dispute
The U.S. Supreme Court has declined to hear a case over whether companies that make biosimilar drugs must wait six months after federal approval before they bring them to the market.
Read ArticleRandom Drug Levels and Anti-Drug Antibodies Predict Rheumatoid Outcomes
Researchers from Manchester, UK have reported the results of their BRAGGSS study showing that certolizumab (CZP) random drug levels and anti-drug antibody (ADAbs) levels can predict optimal outcomes in CZP treated rheumatoid arthritis patients.
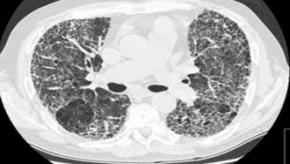
Read ArticleRituximab May Reduce Mortality Risk in Rheumatoid Lung Disease
While it is known that rheumatoid arthritis (RA) patients with interstitial lung involvement is associated with severe seropositive disease and is associated with an increased mortality.
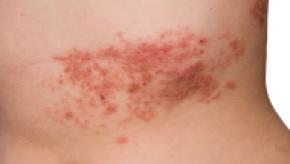
Read ArticleHerpes Zoster and GCA – What’s the Deal?
In November I reported on an abstract presented at ACR by Muratore, et al. who looked for evidence of VZV infection in temporal artery biopsies of GCA patients and found none.
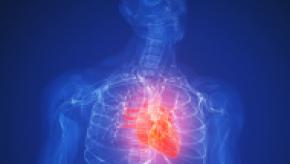
Read ArticleNonsignificant CV Risk with Actemra vs Enbrel
WASHINGTON -- Rheumatoid arthritis is a recognized risk factor for heart attacks and stroke, but a study comparing two leading biologics found only a non-significant increase in relative risk for patients treated with toclizimab (Actemra) vs etanercept (Enbrel), researchers reported here.
Read ArticleKey Lessons from the TNF Inhibitor Head-to-Head EXXELERATE Study
The EXXELERATE study is featured prominently in Lancet this week. In some ways, this represents a landmark negative trial that rheumatologists should review and be aware of.
Read ArticleIs ABT-494 the Next JAK Inhibitor?
Tofacitinib may well be joined by a new once daily JAK inhibitor, baricitinib in the next few months.
Read Article