All News
Great Lectures; Great Presentations
Dr. Jack Cush lectures on giving a great presentation; based on 40 years of lecturing in Rheumatology.
Read ArticleCOVID Virus Persistence with DMARD Use
Drugs commonly used to treat systemic autoimmune rheumatic diseases (SARDs) may keep the SARS-CoV-2 circulating after COVID-19 infection in patients with these conditions, with researchers documenting substantially increased viral antigen persistence compared with other post-COVID patients.
Read ArticleReview of GLP-1 Receptor Agonists in Psoriasis
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have transformed cardiometabolic medicine and are now attracting intense interest in inflammatory disorders.
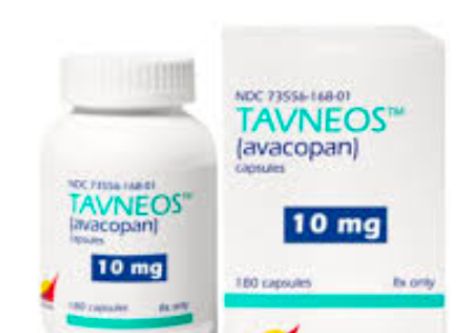
Read ArticleFDA Targeting the Safety of Avacopan
The future of avacopan is up in the air. In April, the FDA issued a drug safety warning, previously requesting that Amgen to pull its rare disease drug Tavneos from the market. Now, there appears more pressure to remove it from the market.
Read ArticleEULAR Recommendations for Physical Activity in Arthritis
EULAR has published a 2025 update for physical activity recommendations for patients with osteoarthritis (OA) and rheumatoid arthritis (RA).
Read ArticleNeoplasia and Autoimmune Disease
Malignancy rivals cardiovascular disease as a leading cause of death in patients with systemic autoimmune diseases. Chronic inflammation and immune dysregulation can drive oncogenesis, while antitumor immune responses can trigger autoimmune phenomena (paraneoplastic syndromes, checkpoint inhibitor-induced rheumatic disease).
Read ArticleSwitch or Cycle - Upadacitinib vs Adalimumab in Refractory RA
After the first tumour necrosis factor inhibitor (TNFi) failure, patients with active rheumatoid arthritis (RA) responded by switching to upadacitinib, compared to cycling to a second TNFi, adalimumab.
Follow the Money (4.23.2026)
Dr. Jack Cush follows the money and all the news that fits the Rheumatology Gab for this past week.
Read ArticleTwofold Mortality in SLE
Despite declining incidence of lupus, mortality for SLE was twice that of controls in this large incident cohort study.
NSAIDs in Inflammatory Bowel Disease?
At least some patients with inflammatory bowel disease (IBD) can safely use common drugs for musculoskeletal aches and pains, a large study of insurance claims data suggested, contradicting a widespread concern that these products can trigger IBD flares.
Read Article

Links:

Links:



Links: