Review of Toll-like Receptor Research Save
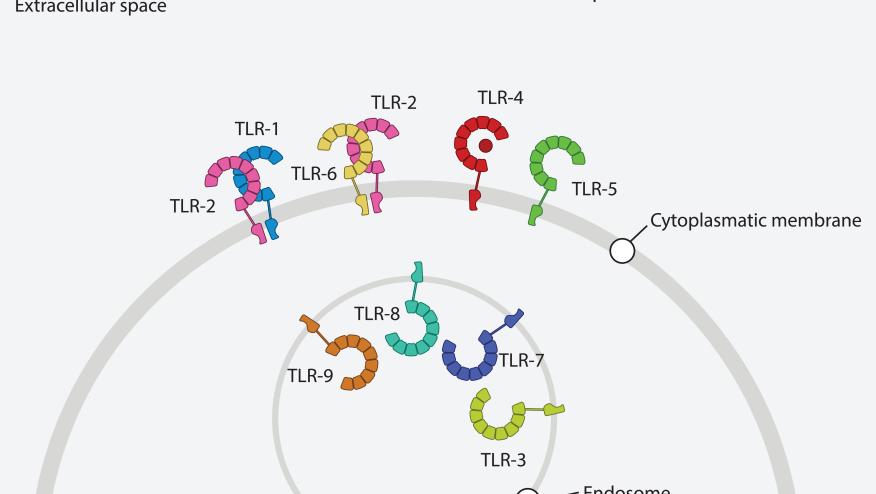
A new review on Toll-like receptors (TLRs) systematically summarizes forty years of discoveries in the TLR field, from the initial identification of Toll in Drosophila embryonic development to the current understanding of multilayered regulatory mechanisms that govern TLR signaling.
The ability of the innate immune system to detect pathogens while maintaining self-tolerance has been a central question in immunology. The discovery of Toll-like receptors as pattern recognition receptors that sense pathogen-associated and damage-associated molecular patterns revolutionized this field, earning the 2011 Nobel Prize in Physiology or Medicine. However, research over the past decade has revealed that TLR signaling is far more nuanced than initially appreciated, the host has evolved multiple layers of regulatory mechanisms operating in parallel and cross-talk.
Post-translational modifications such as phosphorylation, ubiquitination, methylation, acetylation, SUMOylation, succinylation, and nitrosylation, each fine-tuning TLR activation intensity and signal duration. Epigenetic mechanisms—including DNA methylation, histone modifications, chromatin remodeling, and RNA modifications—shape the chromatin landscape of TLR responses over longer time scales. Metabolic reprogramming reveals bidirectional crosstalk between energy metabolism, fatty acid oxidation, lipid metabolism, amino acid metabolism, and TLR signaling, forming flexible feedback loops.
Of particular interest, the identification of biomolecular condensates driven by phase separation represents an entirely new dimension of TLR regulation, offering fresh perspectives on the spatiotemporal dynamics of innate immune signal transduction.
The review further delineates five fundamental modes of cross-regulation that enable signal integration: complex interactions among multiple post-translational modifications triggered by the same pathogen stimulus; convergence of post-translational modifications, metabolic regulation, and epigenetic modifications to form a stable yet flexible response system; synergistic amplification when different TLRs are activated by distinct ligands; mutual regulation among effector mechanisms downstream of different TLRs; and extensive crosstalk between TLRs and other pattern recognition receptor pathways. Together, these mechanisms ensure precise and balanced immune responses.
Beyond intracellular molecular events, TLR signaling is deeply integrated with diverse extrinsic signals from the tissue microenvironment. Cell death signals, cytokine networks, oxygen gradients, nutrient availability, neuroimmune regulation, mechanical forces, and pH fluctuations all intersect with the TLR network to shape the direction of inflammatory responses. This integrative capacity allows TLRs to generate context-appropriate immune outcomes across a wide range of pathophysiological settings, including infection, tissue injury, autoimmunity, cancer, and inflammatory aging.
In parallel, pathogens have evolved sophisticated evasion strategies, including mask of PAMPs, degradation or hijacking of TLR signaling molecules, exploitation of host immune dysregulation, and manipulation of phase separation mechanisms. “These evasion tactics, in turn, provide natural functional annotations of critical nodes within the TLR pathway,” the authors proposed.
Aberrant TLR signaling is closely linked to multiple human diseases. In infectious diseases, TLR polymorphisms significantly influence host susceptibility. In autoimmune conditions such as systemic lupus erythematosus, rheumatoid arthritis, psoriasis, and inflammatory bowel disease, overactivation or dysregulation of endosomal TLRs (TLR7, TLR8, TLR9) is a core driver of pathogenesis. In cancer, TLR signaling exhibits a complex duality, capable of both activating anti-tumor immunity and promoting tumor initiation and metastasis. In immunosenescence and inflammaging, dysregulated TLR signaling serves as a key hub connecting aging with diverse age-related diseases. These profound links between fundamental mechanisms and human pathology underscore the therapeutic potential of targeting TLR pathways.
Pharmacological strategies targeting TLRs are rapidly evolving. Approved agents such as MPLA, imiquimod, and CpG-1018 have demonstrated clinical value as vaccine adjuvants or in topical immunotherapy. However, systemic TLR agonists face major translational hurdles, with late-stage failures of compounds like eritoran and PF-3512676 highlighting the need for precise timing, patient stratification, and optimized delivery strategies.
“Future directions are shifting from broad immune activation to disease-specific, precision modulation of signaling”, the authors pointed out, “including biased ligands or allosteric modulators for pathway-directed regulation, smart delivery systems such as nanoparticles for spatiotemporal precision, biomarker-driven patient stratification, and integration of host-microbiome interactions into therapeutic design.”
The story of TLR research is far from finished. This major review not only synthesizes the intrinsic regulatory networks, tissue microenvironment interactions, and diverse disease roles of TLR signaling but also builds a bridge between established mechanistic knowledge and unmet clinical needs. With continued advances in systems immunology, structural biology, artificial intelligence, and precision delivery technologies, the precise modulation of TLR pathways is expected to play an increasingly important role in future immunotherapy for infectious diseases, autoimmune diseases, cancer, neurodegeneration, and transplant rejection.









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.