News
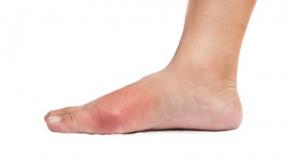
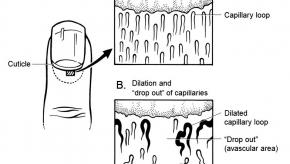
Nailfold Capillaroscopy Excludes Scleroderma in Raynaud’s
Nailfold capillaroscopy test had impressive 90% negative predictive value for Systemic Sclerosis (SSc)RheumNow Week in Review – 26 August 2016
Dr. Jack Cush reviews news and journal reports from the past week on RheumNow.com.
Prior Cancer Patients May Safely Receive TNF Inhibitors or Rituximab
Current guidelines regarding the use of biologics in patients who have previously had a malignancy are based more on opinion than evidence.
Adalimumab Controls Non-Infectious Uveitis in VISUAL II Study
It has long been observed that patients receiving monoclonal antibody based anti-TNF (TNFi) therapies for spondylitis or psoriatic disease have less inflammatory eye complications, such as uveitis. These agents have become common interventions based on these observational results.
Drug Use Compounds Risk - Before and After Fragility Fractures
Is the occurrence of a fragility fracture a missed opportunity to reduce exposure to prescription drugs associated with fracture risk?
Risk of Sepsis or Death Lower with Biologics in Rheumatoid Arthritis
Researchers from Germany have analyzed their large prospective rheumatoid arthritis (RA) registry to address whether being on biologic therapies leads to poorer outcomes - namely sepsis or death, following a serious infection (SIE).
Knuckle Cracking Good News
Researchers from the radiology department at University of California, Davis Health System have studied the phenomenon of knuckle cracking by simultaneously recording audio and ultrasound imaging from 17 women and 23 men.
RheumNow Week in Review – 19 August 2016
Dr. Cush reviews the RheumNow.com highlights in rheumatology for the week ending 19 August 2016.ARCTIC Study Shows No Benefit to Ultrasound Remission in RA
The goal in rheumatoid arthritis (RA) treatment is remission. But what level or method of remission is best?
Remicade Patent Infringements Denied: Opens Door for New Biosimilars
A U.S. court has affirmed its decision that the patent for Johnson & Johnson's drug Remicade is invalid. This new ruling favors the intent of Celltrion and Pfizer, who are seeking to bring their biosimilar versions of infliximab to market in the near future.