All News
AstraZeneca Pushes Forward on Brodalumab Development
Despite Amgen's withdrawal as a co-developer of brodalumab, the anti-IL17A monoclonal antibody, AstraZeneca has announced it will push its plan to develop the drug. Brodalumab is currently in phase III trials for use in psoriasis, psoriatic arthritis and ankylosing spondylitis.
Read ArticleACTEMRA Gets "Breakthrough" Status from the FDA for Scleroderma
The FASSCINATE study presented at EULAR 2015 was instrumental in tocilizumab (TCZ) getting a nod from the FDA to develop TCZ for scleroderma.
Read ArticleLegionella Outbreak in NYC
An outbreak of Legionnaires' disease has killed four people and sickened 65 in the Bronx section of New York City since July 10, according to New York City health officials. This Legionnaires' outbreak is now more than five times the number of cases recorded in the last outbre
Read ArticleB cell Inhibition with Epratuzumab Fails in Two Phase III Lupus Trials
Targeting B cells in systemic lupus erythematosus, the prototypic autoimmune disorder, has long been an attractive target for researchers. Despite negative trials with rituximab and the limited success of belimumab, efforts to inhibit B cell activity have continued.
Read ArticleMen and Blacks Undertreated for Osteoporosis
Using claims data from 36,965 patients in an Indiana database, researchers found women (OR 1.86) and non-black patients (OR 1.52) were more likely to be treated (p<0.001) for osteoporosis.
Read Article2015 Guidelines for Idiopathic Pulmonary Fibrosis
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, fibrosing form of interstitial pneumonia, with poor survival rates of nearly 50% at 3 years.
Read ArticleOutcry Over the Cost of Drugs
Several news articles have addressed the rising concern by patients and physicians over the cost of newer drugs. This issue comes to a head as more than 100 oncologists from top cancer hospitals around the U.S.
Read ArticleDSB: Drug Shortages July 2015
Several drugs continue to be in short supply, posing significant problems for patients. Foremost on this list is leflunomide (Arava) which is backordered for both 10 mg and 20 mg tablets.
Leflunomide Shortage
Read ArticleRituximab Induced Serum Sickness
Karmacharya and colleages have reviewed 33 cases of rituximab-induced serum sickness. Half of these patients had the classic triad of SS fever, rash, and arthralgia and a self-limiting course. The time to onset after infusion was greater with the first dose of rituximab c
Read ArticleIL-1 Inhibition May Be an Alternative Treatment for Behcet's
Behcet's disease is difficult to manage, especially if the goal is to avoid corticosteroids.
Read ArticleDSB Reports & Updates – July 2015
FDA Scours the Internet for Safety Signals. A Bloomberg News report shows how the FDA is using Google or Yahoo keyword searches to identify new safety signals or drug interactions, often more than a year before they were brought to light by the FDA based on prior methodologies de
Read ArticleThe Use of Non-TNF Biologic Treatments in Uveitis Reviewed
A systematic review of treatments used in autoimmune chronic uveitis after failure of DMARDs and at least one TNF inhibitor identified few patients (12 children and 34 adults), and few trials examining rituximab (3), abatacept (3), tocilizumab (3), and 1 each with alemtuzumab and anakin
Read ArticleXeljanz Warnings on Shingles Updated by FDA
The MedWatch June 2015 safety labeling changes were published yesterday, and updated the warnings for Xeljanz (tofacitinib). "The risk of herpes zoster is increased in patients treated with Xeljanz and appears to be higher in patients treated with Xeljanz in Japan."
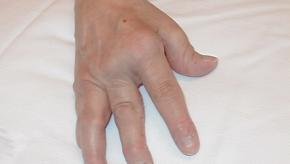
Read ArticleGuselkumab Tops Adalimumab in Psoriasis Trial
NEJM reports a 52 week, phase II trial in 293 adult patients with moderate-to-severe psoriasis (>10 BSA) who were randomized to receive either placebo, adalimumab or guselkumab - a novel IL-23 inhibitor.
Read ArticleBrodalumab Backlash
On May 26, RheumNow.com reported that Amgen and Astra-Zenica suspended drug development for the IL-17 inhibitor brodalumab.
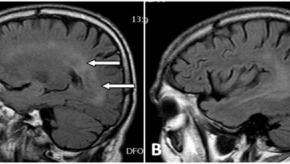
Read ArticleNeurologic Events with TNF Inhibitor Therapy
Demyelinating disorders are an uncommon complication of TNF inhibition and have been described with all TNF inhibitors but also with other biologics.
Read ArticleRheumatology Leads the List in Drug Company Engagements
Based on recently released data, ProPublica.org has compiled a list of pharmaceutical payments made to 606,000 US physicians in the last year. Their analysis turned up big differences between the different specialties.
Read ArticleAdalimumab Drug Levels Predict Efficacy
In a prospective study of 311 rheumatoid arthritis patients starting either adalimumab (ADA) or etanercept, investigators sought to correlate drug levels and/or anti-drug antibodies with clinical outcomes (citation source http://url.ie/z237).
Read ArticleThe Unproven Use of Stem Cell Therapy in OA of the Knee
Regenerative medicine and the use of bone marrow stromal cells (or mesenchymal stem cells - MSC) is controversial in many areas of medicine, including osteoarthritis. MSC use is at a very early stage in orthopedic research, but has been investigated in osteoarthritis of the kn
Read ArticleTNF inhibitors in Early Axial SpA Spares NSAID Use
In a 2 year follow-up of the DESIR cohort of patients with early inflammatory back pain (627 patients), TNF inhibitor use was associated with a significantly greater decrease in the median NSAID intake (from 54.9 to 1.9 in TNFi pts versus from 41.9 to 22.3 in non-TNFi pts, p0.05).
Read Article