All News
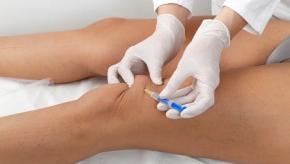
EULAR Guidelines on Intraarticular Therapy
EULAR has published evidence-based recommendations on the use of intra-articular therapies (IAT) based on the literature review and recommendations of a multidisciplinary international task force. These IAT recommendations apply to adult patients with peripheral arthropathies.
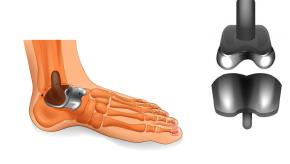
Read ArticleTotal Ankle Arthroplasty vs. Arthodeisis?
The Journal of Bone & Joint Surgery reports the findings of a 4-year trial comparing outcomes of total ankle arthroplasty (TAA) versus ankle arthrodesis (AA) in patients with severe ankle arthritis; finding that while both groups improved, TAA was associated with less pain and better function after 4 years.
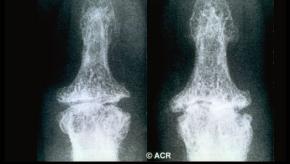
Read ArticleSeverity Associated with Erosive Hand Osteoarthritis
Erosive hand osteoarthritis (EHOA) was studied as part of the Osteoarthritis Initiative, and shown to increase with advanced age and female sex, with developmental findings to suggest that EHOA as a disorder of skeletal frailty.
Read ArticleNSAID Safety with COVID-19 Infection
NSAIDs do not affect COVID outcomes; here we are nearly 16 months into the pandemic, and this is the common sense conclusion of a recent trial published in Lancet Rheumatology.
Read ArticleRheumNow Podcast – Uppers and Downers
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com.
Read ArticleLittle Effect with Multimodal Treatment of CMC1 Osteoarthritis
A randomized clinical trial of patients with CMC1 osteoarthritis shows modest benefits when a combination of conservative treatments (education, splinting, hand exercises, 1% diclofenac sodium gel) was used compared to education alone. While there were small to medium benefits with regard to hand function, there was no benefit in pain outcomes.
Read ArticleRheumNow Podcast – Coconspirators: RA and Lung Disease
Dr. Jack Cush reviews and discusses more than a dozen news and journal articles from the past week on RheumNow.com.
Read ArticleNICE Guidelines on Chronic Pain Management
In the United Kingdom, where it is estimated that chronic pain affects one‑third to one‑half of the population, NICE (National Institute for Health and Care Excellence) has recently released an updated guideline on the management of chronic pain for people aged 16 years and over.
Read ArticleCost Efficacy of Knee Replacement in Obese Osteoarthritis
Total knee replacement (TKR) in obese patients with end-stage knee osteoarthritis appears to be a beneficial and cost-effective strategy for treating. The only potential limitation is a greater risk for adverse events in those with a body mass index (BMI) of 40 kg/m2 or greater.
Read ArticleAortic Calcification and Lumbar Fracture Risk in Older Men
Fractures of the spine and abdominal aortic calcification (AAC) can both be visualized with spinal imaging; a new report shows that finding both radiographic vertebral fracture and AAC on the same lateral spine images may predict the risk of future incident major osteoporotic fracture i
Read Article
Links:
Dr. John Cush RheumNow ( View Tweet)
Links:

Links:

Links:

Links:

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)
Links:

Links:
Dr. John Cush RheumNow ( View Tweet)
Links: