All News
EGPA Review
A current review of eosinophilic granulomatosis with polyangiitis (EGPA) provides current recommendations on diagnosis and management.
Read ArticleToll-Like Receptor Drugs Effective in Cutaneous Lupus
Enpatoran, an investigational oral drug that inhibits Toll-like receptors (TLR) 7 and 8, was superior to placebo in relieving lupus skin manifestations in a dose-finding phase II trial.
Read ArticleVitamin D in Rheumatic Disease - is it Good Enough?
Breaking the Rules: Dual-Advanced Combinations in Rheumatology
For decades, the standard of care in rheumatology has been combination therapy - pairing methotrexate (MTX) with an advanced biologic or synthetic agent to achieve optimal outcomes.
Read ArticleGreat Lectures; Great Presentations
Dr. Jack Cush lectures on giving a great presentation; based on 40 years of lecturing in Rheumatology.
Read ArticleFollow the Money (4.23.2026)
Dr. Jack Cush follows the money and all the news that fits the Rheumatology Gab for this past week.
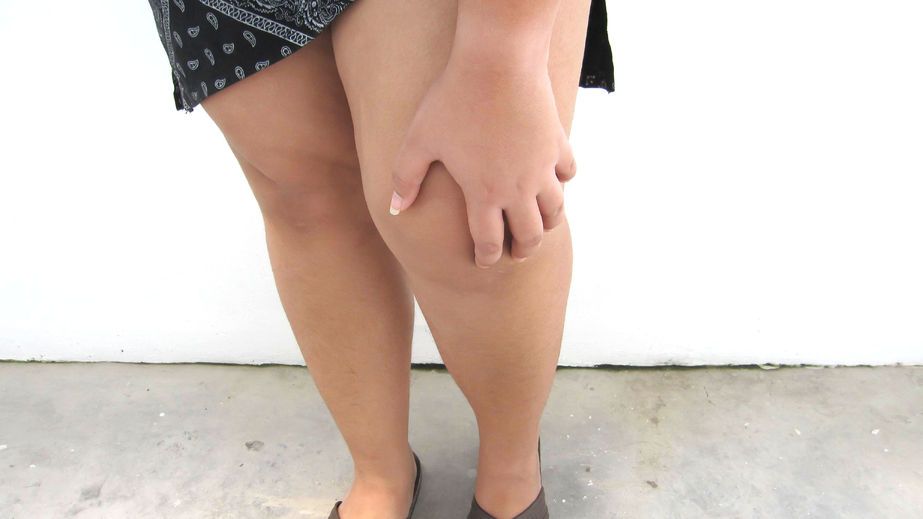
Read ArticleWeight-loss drugs may affect joints directly
A new study from Aarhus University shows that the hormone GLP-1, which is mimicked in medication for diabetes and weight loss, is present in joint fluid. In the long term, this may open up new treatments for arthritis.







Links:

Links: