All News
Count Your Telangiectasias (5.29.2026)
Dr. Jack Cush reviews the news, journal articles and missed quiz questions.
Read ArticleBE BOLD Week 16 data - Bimekizumab vs. Risankizumab in PsA
UCB has released the Week 16 results from BE BOLD trial that will be presented at EULAR 2026 in London on Saturday, June 6.
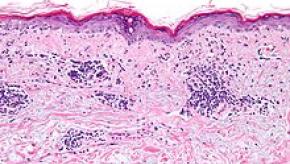
Read ArticleA Role for Plasmacytoid Dendritic Cells in Lupus
Plasmacytoid dendritic cells (pDCs) have been strongly implicated in the pathogenesis of lupus, as their primary product, type I interferon, drive systemic lupus erythematosus and cutaneous lupus erythematosus. A recent comprehensive review synthesizes the available data on pDCs, and, importantly, what we still don't know.
Read ArticleInfectious Rheumatology (5.15.2026)
Dr. Jack Cush discusses his favorite journal articles from the past week on RheumNow.com.
Read ArticleToll-Like Receptor Drugs Effective in Cutaneous Lupus
Enpatoran, an investigational oral drug that inhibits Toll-like receptors (TLR) 7 and 8, was superior to placebo in relieving lupus skin manifestations in a dose-finding phase II trial.
Read ArticleReview of GLP-1 Receptor Agonists in Psoriasis
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have transformed cardiometabolic medicine and are now attracting intense interest in inflammatory disorders.
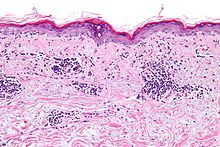
Read ArticleLitifilimab in Cutaneous Lupus Erythematosus
Biogen has announced positive results of their Phase 2 study, evaluating litifilimab in patients with active cutaneous lupus erythematosus (CLE), presented at the recent American Academy of Dermatology (AAD) Annual Meeting.
Read ArticleTop Four and More (4.10.2026)
Dr. Jack Cush reviews the news and journal reports from RheumNow.com this week - including his top four favorite subjects.
Read Article

Links:

Links:





Links: