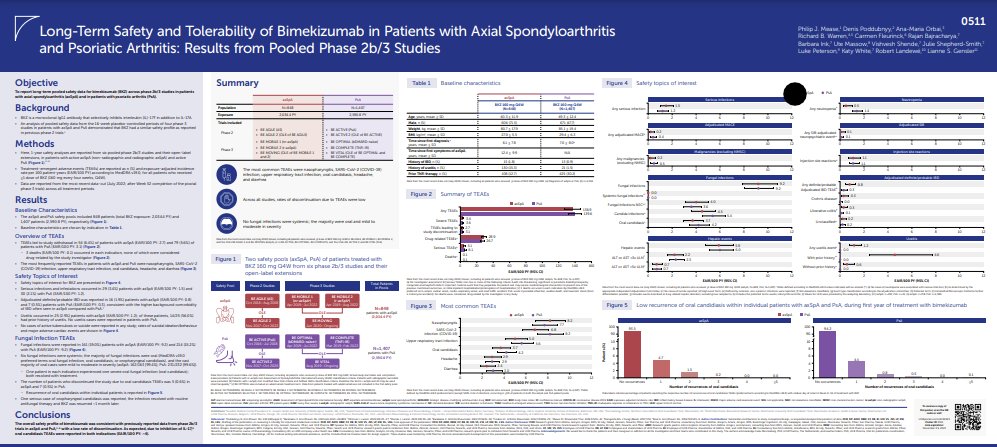
All News
IV Secukinumab for AxSpa
The FDA recently announced approval for intravenous secukinumab (Cosentyx) for adults with psoriatic arthritis, ankylosing spondylitis, and non-radiographic spondylitis. Data for this new approval will be presented at ACR Convergence on November 14th by Dr. Atul Deodhar.
Read ArticleBimekizumab is Coming… in Third Place
Of the many exciting advancements for patients with psoriatic arthritis at ACR Convergence, one upcoming therapy stood out: the dual IL-17A/F inhibitor bimekizumab. Nearly two dozen bimekizumab abstracts will be featured at this year’s meeting and it recently received authorization in Europe and the U.S. for psoriasis. That makes this the year I plan to figure out where this drug will be useful for my patients with PsA.
Read ArticleLate Should Not Be Less (11.3.2023)
Dr. Jack Cush reviews the news, journal reports and regulatory approvals from the past week on RheumNow.com.
Read ArticleMethotrexate in OA (10.20.2023)
Dr. Jack Cush discusses the news, journal articles and regulatory actions. This week we discuss JAKne, DLE and SLE and more.
Read ArticleFDA Approves Bimekizumab for Plaque Psoriasis
UCB announced today that the US Food and Drug Administration (FDA) has approved bimekizumab-bkzx (Bimzelx)) for the treatment of moderate to severe plaque psoriasis in adults who needing systemic therapy or phototherapy.
Read ArticleSleep Apnea Smart Watches (10.13.2023)
Dr. Jack Cush reviews the news, journal reports and regulatory approvals from this past week on RheumNow.com.
Read ArticleFDA Approves IV Secukinumab
On Oct. 6, 2023, Novartis announced the US Food and Drug Administration (FDA) approved an intravenous (IV) formulation of secukinumab (Cosentyx) for use in treating adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS) and non-radiographic axial spondyloarthritis (nr-axSpA).
Read ArticleSustained Efficacy of Bimekizumab in Psoriatic Arthritis
BE OPTIMAL trial tested the benefits of a dual IL-17 A/F inhibitor, bimekizumab (BKZ), in patients with active psoriatic arthritis and showed superior efficacy over placebo, that was sustained beyond the primary endpoint, from week 16 to 52.
Read Article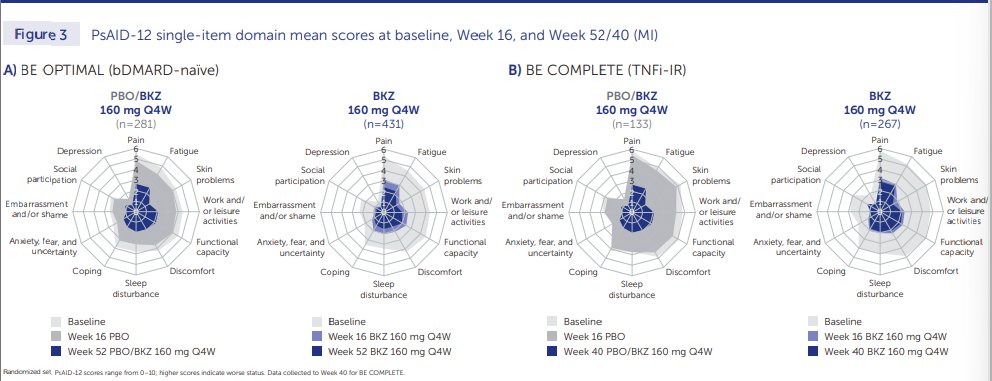

Eric Dein ( View Tweet)
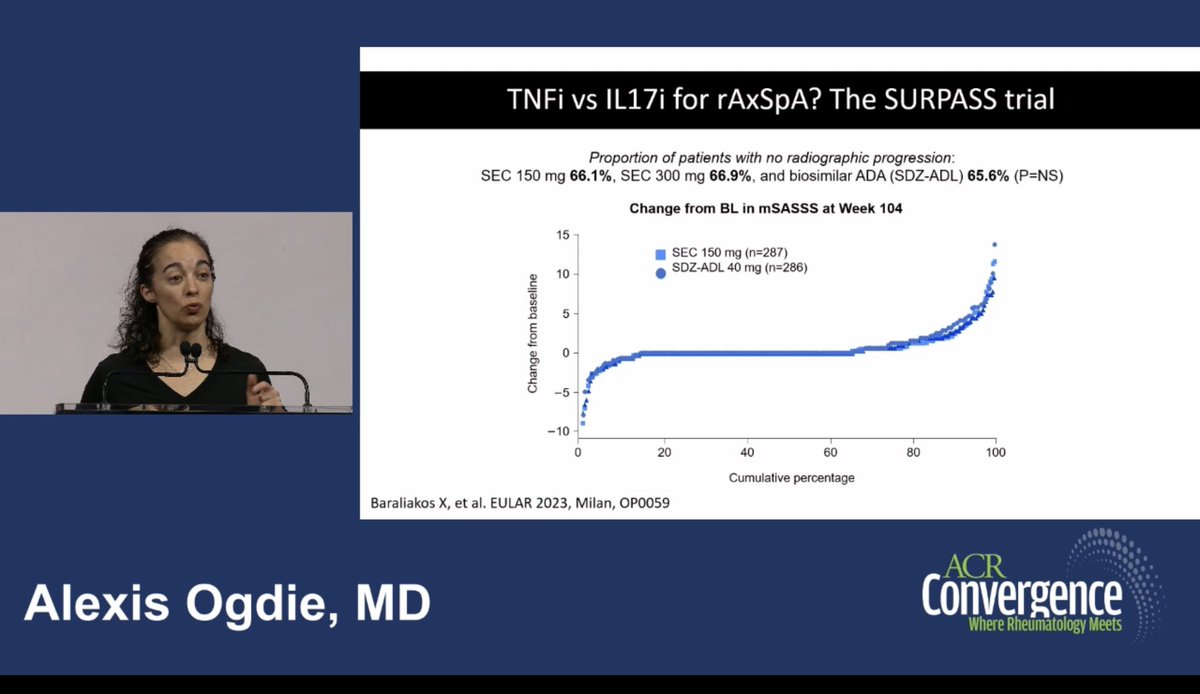

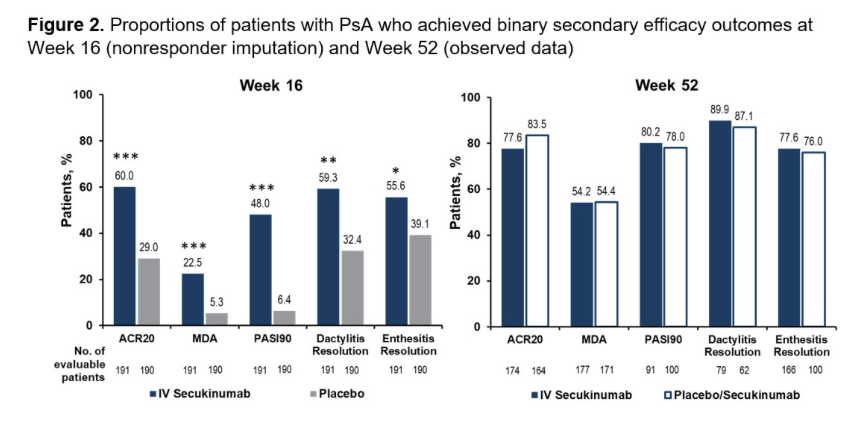

Meral K. El Ramahi, MD MeralElRamahiMD ( View Tweet)

Meral K. El Ramahi, MD MeralElRamahiMD ( View Tweet)