All News
Psoriatic Disease Management - RNL 2021 Highlights
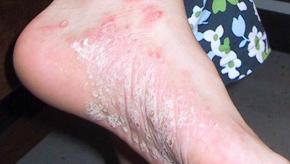
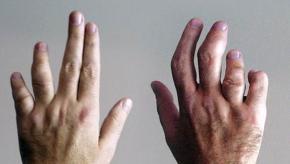
This year’s coverage of psoriatic arthritis was once again, both exciting and informative. Topics included an overview of the latest therapeutic options, an excellent discussion on psoriasis specifically, and the epidemiology of psoriatic arthritis.
Read ArticleRheumNow Podcast – Infection in ANCA-Associated Vasculitis (3.26.2021)
Dr. Jack Cush reviews the news, FDA Denials and journal articles from the past week on RheumNow.com.
Read ArticleRheumNow Podcast – Pounds of Prevention (3.5.2021)
Dr. Jack Cush reviews and discusses nearly a dozen news reports and journal articles from the past week on RheumNow.com.
Read ArticleBaricitinib Therapy in Covid-19 Pneumonia
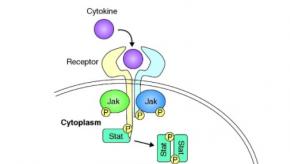
The NEJM has published the results of the ACCT-2 Study, a double-blind, randomized, placebo-controlled trial evaluating baricitinib, an inhibitor of Janus kinase 1 (JAK1) and JAK2, plus remdesivir in hospitalized adults with Covid-19. The primary outcome was the time to recovery.
Read ArticleRheumNow Podcast – Tofacitinib Safety Concerns (2.5.2021)
Dr. Jack Cush reviews and discusses the news and journal reports from the past week on RheumNow.com.
Read ArticleCardiac and Cancer Signals Tofacitinib Safety Alert from FDA
The FDA has notified healthcare professionals of a safety alert concerning tofacitinib (Xeljanz), noting that preliminary results from a long-term safety clinical trial show an increased risk of serious heart-related problems and cancer with tofacitinib (compared to adalimumab) when given to
Read ArticleCalculating Serious Infection Risk in IMID Patients
Predictive modeling of data derived from the DANBIO registry (of treated IMID patients) revealed a 4 fold increased risk of serious infection (SIE) in those starting biologic DMARD (bDMARD) treatment. From this large dataset, researchers developed a simple prediction model to estimate future infection risk that may inform shared decision-making in individual patients.
Read ArticleJAK Inhibitor Misses Endpoint in Safety Study. Now What?
Pfizer announced results Wednesday from its FDA-mandated postmarketing safety study of tofacitinib (Xeljanz), and they don't bode well for the drug and possibly others in its class.
Read Article2020 Rheumatology Year in Review
2020 was historic, memorable and game-changing. Under the cloud of COVID-19, there were many significant and memorable advances and setbacks for the rheumatology world. In our accounting of most read articles for 2020, (not surprisingly) 17/20 were COVID-related.
Read ArticleFilgotinib Bows Out of the RA Race in the USA
Gilead Sciences Inc announced yesterday that it has decided not to pursue the an rheumatoid arthritis (RA) indicatin for its once daily oral JAK inhibitor, filgotinib (also named Jyseleca).
Read ArticleConsensus Recommendations on JAK Inhibitor Use
A steering committee and task force was convened to develop recommendations for the use of Janus kinase (JAK) inhibitors in the treatment of immune-mediated inflammatory diseases (IMID).
Read ArticleUpadacitinib Effective in Biologic Refractory Psoriatic Arthritis
The SELECT-PsA 2 trial was presented at the ACR meeting and recently published in Annals of Rheumatic Disease, shows that psoriatic arthritis (PsA) patients who have failed at least one biologic DMARD, did respond well to upadacitinib.
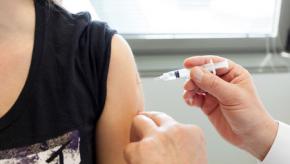
Read ArticleVaccination with Shingrix: Does it work for patients on JAKi?
Dr. Jeffrey Curtis examines abstracts #1997 and #0452 presented at the ACR 2020 annual meeting.
Read Article
Links:


Links:
Dr. John Cush RheumNow ( View Tweet)

Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Dr. John Cush RheumNow ( View Tweet)

Links: