All News
Tofacitinib Scores in TNF inhibitor Resistant Psoriatic Arthritis
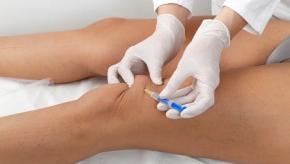
Mease et al has reported in the NEJM the results of a phase 3 trial showing tofacitinib (Tofa) to be significantly more effective than placebo in active psoriatic arthritis (PsA) patients who previously failed to respond to tumor necrosis factor inhibitor (TNFi) therapies.
Read ArticleRheumatologists are Slow to Change DMARDs
The paradigm of rheumatoid arthritis (RA) therapy mandates early diagnosis and aggressive treatment. Yet a recent cohort study has shown that RA patients with moderate to high disease activity (MHDAS) were met with infrequent DMARD adjustments; with median time to DMARD adjustment being 5 months and median time to low disease activity (LDAS) was roughly 10 months.
Read ArticleStelara FDA Approved for Use in Adolescent Psoriasis
Johnson & Johnson has announced that they have received an expanded FDA approval for the use of ustekinumab (Stelara) in treating adolescent patients, aged 12 and over, with moderate to severe plaque psoriais.
Read ArticleHigh Drug Discontinuations and Nonadherence in Fibromyalgia
Compliance and adherence was shown to be quite poor in fibromyalgia (FM) patients, with high rates of drug discontinuation and greater than 60% demonstrated low adherence.
Read ArticleSpinal X-ray Inhibition in Ankylosing Spondylitis Best Achieved by Control of Activity
Although tumour necrosis factor inhibitors (TNFi) are highly effective (on clinical grounds) in ankylosing spondylitis (AS), proof that they may alter the course of spinal radiographic progression has been scant.
Read Article13 October 2017 The RheumNow Week in Review
The RheumNow Week in Review discusses the past week's news, journal articles and highlights from RheumNow.com. In this week's report, Dr. Jack Cush discusses a novel antiviral drug or the use of stem cell therapy for scleroderma, rituximab in HSP, when to withdraw NSAIDS in RA, and congrats to PAs and NPs!
Read ArticleNovel Gel Drug Delivery Developed for Rheumatoid Arthritis
Scientists at the Institute for Basic Science have invented a hydrogel capable of delivering drug at sites of inflammation in disorders such as rheumatoid arthritis.
Read ArticleBSR Guidelines on Lupus Management
In the UK, NICE has accredited the British Society of Rheumatology (BSR) to develop a guidance document on the management of systemic lupus erythematosus in adults. The last published guidelines for lupus were published in 2008 by EULAR and in 2012 by the ACR.
Read ArticleZilretta - a New Drug FDA Approved for Osteoarthritis of the Knee
Flexion Therapeutics announced friday that the U.S. Food and Drug Administration (FDA) has approved its osteoarthritis (OA) of the knee injectable steroid drug Zilretta with the indication of moderate-to-severe knee pain.
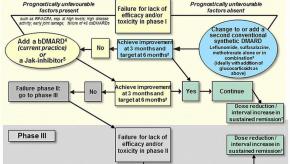
Read Article2016 EULAR Guidelines on RA Management
The management of rheumatoid arthritis (RA) has evolved significantly with time. Nevertheless, there are still some uncertainties - such as when, what and which biologic or novel therapy should be used.
Read ArticleThe RheumNow Week in Review - 6 October 2017
The RheumNow Week in Review discusses the past week's news, journal articles and highlights from RheumNow.com. In this week's report, Dr. Jack Cush discusses when to hold the biologic, lymphoma risk with tofacitinib, early clues to the diagnosis of RA, biologic use in pregnancy, what's killing psoriasis patients and the 2016 top 5, best selling drugs in rheumatology.
Read ArticlePatterns of Biologic Use During Autoimmune Pregnancy
While it is highly desirable to avoid medications during conception and pregnancy, statistics show that >90% of women take at least one drug during pregnancy and nearly half will take 3 or more medications during pregnancy.
Read ArticleAlcohol Related Deaths Increased in Psoriasis
JAMA Dermatology has published a report showing psoriasis carries a 60% greater risk alcohol-related death compared with those without psoriasis. (Citation source: https://buff.ly/2galDwr)
Read ArticleAmgen-Abbvie Settle Humira Biosimilar Patent Dispute
Amgen's biosimilar version of adalimumab (Humira) was FDA-approved in September 2016 and given the trade name Amjevita (generic: adalimumab-atto). This new TNF inhibitor biosimilar has not yet been to market because of legal wranglings over patent issues by Abbvie's Humira.
Read Article29 September 2017 The RheumNow Week in Review
The RheumNow Week in Review discusses the past week's news, journal articles and highlights from RheumNow.com. This week's report discusses regulatory actions by NICE and FDA, higher death rates in RA and psoriasis, increased risk of RA with Asthma, rising numbers for OA, RA, and STDs.
Read ArticlePsoriasis Increases Risk of Major Adverse Cardiovascular Events
A report in the Journal of the American Academy of Dermatology shows that psoriasis duration and inflammation may result in cardiovascular inflammation and an increased risk of major adverse CV events.
Read ArticleSafety of Long-Term PPI Use
The current issue of JAMA reviews the safety of long-term use of proton pump inhibitors (PPI). The following is a collection of the evidence about these purported risks.
Fractures
Read ArticleUAB Researchers Shed Light on Age-Related Osteoporosis
Researchers from the University of Alabama at Birmingham (UAB) have detailed mechanisms leading to age-related bone loss and osteoporosis.
Read ArticleNo Cancer Risk With Biologic Use
A polulation-based study from Sweden has shown that treatment with tocilizumab, abatacept, rituximab, or tumor necrosis factor (TNFi) inhibitors does not affect the risk of malignant neoplasms among patients with rheumatoid arthritis. Specifically, use of a first or second TNFi or biologic DMARDs (bDMARD) does confer a different cancer risk when compared to conventional DMARDS in biologic–naive RA patients.
Read ArticleSirukumab Turned Down by FDA
On Friday the U.S. Food and Drug Administration announced that will not approve Johnson & Johnson’s rheumatoid arthritis drug sirukumab, stating further data and study would be needed to establish its safety.
Read Article